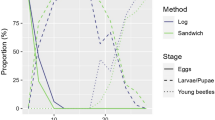
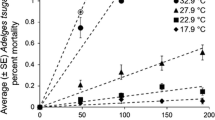
Abstract
Phenological synchrony can promote population growth in species with positive density dependence. Variation among life stages in the thermal thresholds for development can foster phenological synchrony under thermal regimes that include frequent occurrence of temperatures between developmental thresholds. The southern pine beetle is an insect with positive density dependence that has recently undergone important shifts in population abundance at the northern extremes of their distribution. We evaluated the hypothesis that cooler winter temperatures in their northern range cause a convergence of the population life stage structure that leads to synchrony in spring flight phenology. We used a combination of approaches. First, in situ laboratory experiments demonstrated a threshold temperature for pupation that was greater than was required for larval development; rearing larvae at lower temperatures increased the pooling of individuals at the end stage of larval development and synchrony in adult emergence. Second, a development rate model showed a similar convergence of the majority of the population at the end stage of larval development when brood experienced the cooler temperatures of the northern region, but not with temperatures from the southern region, or as a null model. Finally, field trapping of wild beetles showed greater synchrony in the pine forests of New Jersey than in the warmer, historically occupied forests of Georgia and Mississippi. Given these results, pine-dominated forests in the northern edge of the southern pine beetle’s range may experience more frequent occurrence of outbreaks, due to the positive feedbacks associated with a synchronous spring emergence of this insect.




Similar content being viewed by others
References
Angilletta MJ, Dunham AE (2003) The temperature-size rule in ectotherms: simple evolutionary explanations may not be general. Am Nat 162:332–342
Aoki CF (2017) Forest risk and irruptive insect pests: ecology for management in changing times. PhD dissertation. Dept. of Biological Sciences, Dartmouth College, Hanover, NH USA
Ayres MP, Maclean SF (1987) Molt as a component of insect development: Galerucella sagittariae (Chrysomelidae) and Epirrita autulmnata (Geometridae). Oikos 48:273–279
Ayres MP, Scriber JM (1994) Local adaptation to regional climates in Papilio canadensis (Lepidoptera: Papilionidae). Ecol Monogr 64:465–482
Ayres MP, Martinson SJ, Friedenberg NA (2011) Southern pine beetle ecology: populations within stands. In: Coulson RN (eds) Southern pine beetle II (general technical report SRS 140). USDA Forest Service Southern Research Station, Asheville
Bentz BJ, Logan JA, Amman GD (1991) Temperature-dependent development of the mountain pine beetle (Coleoptera: Scolytidae) and simulation of its phenology. Can Entomol 123:1083–1094
Bentz BJ, Vandygriff J, Jensen C, Coleman T, Maloney P, Smith S, Grady A, Schen-Langenheim G (2014) Mountain pine beetle voltinism and life history characteristics across latitudinal and elevational gradients in the western United States. For Sci 60:343–449
Billings RF, Upton WW (2010) A methodology for assessing annual risk of southern pine beetle outbreaks across the southern region using pheromone traps. In: Pye JM, Rauscher HM, Sands Y, Lee DC, Beatty JS, Tech (eds) Advances in threat assessment and their application to forest and rangeland management. Gen. Tech. Rep. PNW-GTR-802, vol 2. U.S. Department of Agriculture, Forest Service, Pacific Northwest and Southern Research Stations, Portland pp 708
Bjørnstad ON, Nelson WA, Tobin PC (2016) Developmental synchrony in multivoltine insects: generation separation versus smearing. Popul Ecol 58:479–491
Boucher DH (1985) Lotka-Volterra models of mutualism and positive density-dependence. Ecol Model 27:251–270
Briere JF, Pracros P, Le Roux AY, Pierre JS (1999) A novel rate model of temperature-dependent development for arthropods. Environ Entomol 28:22–29
Briscoe NJ, Porter WP, Sunnucks P, Kearney MR (2012) Stage-dependent physiological responses in a butterfly cause non-additive effects on phenology. Oikos 212:1464–1472
Cesaraccio C, Spano D, Duce P, Snyder RL (2001) An improved model for determining degree-day values from daily temperature data. Int J Biometeorol 45:161–169
Coulson RN, Flamm RO, Pulley PE, Payne TL, Rykiel EJ, Wagner TL (1986) Response of the southern pine bark beetle guild (Coleoptera: Scolytidae) to host disturbance. Environ Entomol 15:850–858
Bale JS, Masters GJ, Hodkinson ID, Awmack C, Bezemer TM, Brown VK, Butterfield J, Buse A, Coulson JC, Farrar J, Good JEG, Harrington R, Hartley S, Jones TH, Lindroth RL, Press MC, Symrnioudis I, Watt AD, Whittaker JB (2002) Herbivory in global climate change research: direct effects of rising temperatures on insect herbivores. Glob Change Biol 8:1–16
Cronin JT, Reeve JD, Wilkens R, Turchin P (2000) The pattern and range of movement of a checkered beetle predator relative to its bark beetle prey. Oikos 90:127–138
Davidowitz G, D’Amico LJ, Nijhout HF (2003) Critical weight in the development of insect body size. Evol Dev 5:188–197
De Moed GH, Kruitwagen CLJJ, De Jong G, Scharloo W (1999) Critical weight for the induction of pupariation in Drosophila melanogaster: genetic and environmental variation. J Evol Biol 12:85–858
Friedenberg NA, Powell JA, Ayres MP (2007) Synchrony’s double edge: transient dynamics and the Allee effect in stage structured populations. Ecol Lett 10:564–573
Gara RI (1967) Studies on the attack behavior of the southern pine beetle. I. The spreading and collapse of outbreaks. Contrib Boyce Thompson Inst 23:349–354
Hodges JD, Pickard LS (1971) Lightning in the ecology of southern pine beetle, Dendroctonus frontalis (Coleoptera: Scolytidae). Can Entomol 103:44–51
Jenkins JL, Powell JA, Logan JA, Bentz BJ (2001) Low seasonal temperatures promote life cycle synchronization. B Math Biol 63:573–595
Kingsolver JG, Woods HA, Buckley LB, Potter KA, MacLean HJ, Higgins JK (2011) Complex life cycles and the response of insects to climate change. Integr Comp Biol 51:719–732
Lesk C, Coffel E, D’Amato AW, Dodds K, Horton R (2017) Threats to North American forests from southern pine beetle with warming winters. Nat Clim Change 7:713–717
Liebhold AM, Tobin PC (2008) Population ecology of insect invasions and their management. Annu Rev Entomol 53:387–408
Logan JA, Bentz BJ (1999) Model analysis of mountain pine beetle (Coleoptera: Scolytidae) seasonality. Environ Entomol 28:924–934
Logan JA, Wollkind DJ, Hoyt SC, Tanigoshi LK (1976) Analytic model for description of temperature-dependent rate phenomena in Arthropods. Environ Entomol 5:1133–1140
Lombardero MJ, Ayres MP, Ayres BD, Reeve JD (2000) Cold tolerance of four species of bark beetle (Coleoptera: Scolytidae) in North America. Environ Entomol 29:421–432
Martinson SJ, Ylioja T, Sullivan BT, Billings RF, Ayres MP (2013) Alternate attractors in the population dynamics of a tree-killing bark beetle. Popul Ecol 55:95–106
Mawby WD, Hain FP, Doggett CA (1989) Endemic and epidemic populations of southern pine beetle: implications of the 2-phase model for forest managers. For Sci 35:1075–1087
May RM (1977) Thresholds and breakpoints in ecosystems with a multiplicity of stable states. Nature 269:471–477
Parmesan C (2007) Influences of species, latitudes and methodologies on estimates of phenological response to global warming. Glob Change Biol 13:1860–1872
Peacher V (2011) Southern pine beetle information system (SPBIS). Southern Pine Beetle II. USDA Forest Service General Technical Report, Southern Research Station 140:25–50. http://www.treesearch.fs.fed.us/pubs/download/39081.pdf. Accessed 13 Apr 2017
Pincebourde S, Casas J (2015) Warming tolerance across insect ontogeny: influence of joint shifts in microclimates and thermal limits. Ecology 96:986–987
Powell JA, Jenkins JL, Logan JA, Bentz BJ (2000) Seasonal temperature alone can synchronize life cycles. B Math Biol 62:977–998
Pureswaran DS, Sullivan BT (2012) Semiochemical emission from individual galleries of the southern pine beetle, (Coleoptera: Curculionidae), attacking standing trees. J Econ Entomol 105:140–148
Radchuk V, Turlure C, Schtickzelle N (2012) Each life stage matters: the importance of assessing the response to climate change over the complete life cycle in butterflies. J Anim Ecol 82:275–285
Reeve JD (1997) Predation and bark beetle dynamics. Oecologia 112:48–54
Reeve JD, Turchin P (2002) Evidence for predator-prey cycles in a bark beetle. In: Berryman AA (ed) Population cycles: the case for trophic interactions. Oxford University Press, Oxford
Regniere J, Delisle J, Pureswaran DS, Trudel R (2013) Mate-finding Allee effect in spruce budworm population dynamics. Entomol Exp Appl 146:112–122
Ruel JJ, Ayres MP (1999) Jensen’s inequality predicts effects of environmental variation. Trends Ecol Evol 14:361–366
Schrey NMJ, Schrey AW, Heist EJ, Reeve JD (2011) Genetic heterogeneity in a cyclical forest pest, the southern pine beetle, Dendroctonus frontalis, is differentiated into east and west groups in the southeastern United States. J Insect Sci 11:110
Sharpe PJH, Demichele DW (1977) Reaction-kinetics of poikilotherm development. J Theor Biol 64:649–670
Shelomi M (2012) Where are we now? Bergmann’s Rule sensu lato in insects. Am Nat 180:511–519
Sullivan BT (2011) Southern pine beetle behavior and semiochemistry. Southern Pine Beetle II. USDA For Serv Gen Techn Rep South Res Station 140:25–50
Tauber MJ, Tauber CA, Masaki S (1986) Seasonal adaptations of insects. Oxford University Press, New York
Thatcher RC (1967) Winter brood development of the southern pine beetle in southeast Texas. J Econ Entomol 60:599–600
Thatcher RC, Pickard LS (1964) Seasonal variations in activity of the southern pine beetle in east Texas. J Econ Entomol 57:840–842
Tobin PC, Robinet C, Johnson DM, Whitmire SL, Bjornstad ON, Liebhold AM (2009) The role of Allee effects in gypsy moth, Lymantria dispar (L.), invasions. Popul Ecol 51:373–384
Trân JK, Ylioja T, Billings RF, Régnière J, Ayres MP (2007) Impact of minimum winter temperatures on the population dynamics of Dendroctonus frontalis. Ecol Appl 17:882–899
Ungerer MJ, Ayres MP, Lombardero MJ (1999) Climate and the northern distribution limits of Dendroctonus frontalis Zimmermann (Coleoptera: Scolytidae). J Biogeogr 26:1133–1145
Wagner TL, Gagne JA, Sharpe PJH, Coulson RN (1984) A biophysical model of southern pine-beetle, Dendroctonus frontalis Zimmermann (Coleoptera, Scolytidae), development. Ecol Model 21:125–147
Weed AS, Ayres MP, Hicke J (2013) Consequences of climate change for biotic disturbances in North American forests. Ecol Monogr 83:441–470
Weed AS, Ayres MP, Liebhold AM, Billings RF (2016) Spatio-temporal dynamics of a tree-killing beetle and its predator. Ecography 39:1–14
Acknowledgements
We thank the New Jersey Department of Environmental Protection and officials of both the Oconee and Homochitto National Forests (particularly Lee Dunnam) for help in acquiring resources and providing sites for traps. Mike Gallagher, Melanie Maghirang, Brett Manning, JoAnne Barrett, Matt Cloud, Erich Vallery, and Zachary Oliver provided assistance in both field and lab. Thanks to Jim Meeker and Chris Crowe for SPB trapping data and to Ken Clark and John Dighton for logistical support. Research was financially supported by NRI/AFRI 2009-65104-05731 and a cooperative agreement with the Southern Research Station.
Author information
Authors and Affiliations
Contributions
This research was conceived by MPA and BTS. The experiments were designed by MPA, BTS, JAL, CFA, and ASW. Experiments were conducted by JAL, CFA, and ASW. Data analysis and figures completed by JAL, with input from all authors. JAL wrote the manuscript with considerable editorial contributions from all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Sylvain Pincebourde.
Rights and permissions
About this article
Cite this article
Lombardo, J.A., Weed, A.S., Aoki, C.F. et al. Temperature affects phenological synchrony in a tree-killing bark beetle. Oecologia 188, 117–127 (2018). https://doi.org/10.1007/s00442-018-4164-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-018-4164-9