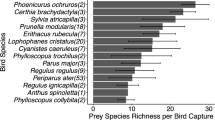
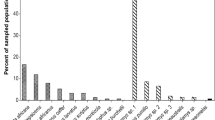
Abstract
Riparian habitats are characterized by substantial flows of emergent aquatic insects that cross the stream-forest interface and provide an important source of prey for insectivorous birds. The increased availability of prey arising from aquatic subsidies attracts high densities of Neotropical migratory songbirds that are thought to exploit emergent aquatic insects as a nestling food resource; however, the prey preferences and diets of birds in these communities are only broadly understood. In this study, we utilized DNA metabarcoding to investigate the extent to which three syntopic species of migratory songbirds—Acadian Flycatcher, Louisiana Waterthrush, and Wood Thrush—breeding in Appalachian riparian habitats (Pennsylvania, USA) exploit and partition aquatic prey subsidies as a nestling food resource. Despite substantial differences in adult foraging strategies, nearly every nestling in this study consumed aquatic taxa, suggesting that aquatic subsidies are an important prey resource for Neotropical migrants nesting in riparian habitats. While our results revealed significant interspecific dietary niche divergence, the diets of Acadian Flycatcher and Wood Thrush nestlings were strikingly similar and exhibited significantly more overlap than expected. These results suggest that the dietary niches of Neotropical migrants with divergent foraging strategies may converge due to the opportunistic provisioning of non-limiting prey resources in riparian habitats. In addition to providing the first application of DNA metabarcoding to investigate diet in a community of Neotropical migrants, this study emphasizes the importance of aquatic subsidies in supporting breeding songbirds and improves our understanding of how anthropogenic disturbances to riparian habitats may negatively impact long-term avian conservation.





Similar content being viewed by others
References
Anderson MJ (2001) A new method for non-parametric multivariate analysis of variance. Austral Ecol 26:32–46
Anderson MJ (2006) Distance-based tests for homogeneity of multivariate dispersions. Biometrics 62:245–253
Arcese P, Smith JN (1988) Effects of population density and supplemental food on reproduction in song sparrows. J Anim Ecol 57:119–136
Barbour MT, Gerritsen J, Snyder BD, Stribling JB (1999) Rapid bioassessment protocols for use in streams and wadeable rivers: periphyton, benthic macroinvertebrates and fish, second edition. EPA 841-B-99-002. U.S. Environmental Protection Agency Office of Water, Washington, DC, USA
Baxter CV, Fausch KD, Carl Saunders W (2005) Tangled webs: reciprocal flows of invertebrate prey link streams and riparian zones. Freshw Biol 50:201–220. https://doi.org/10.1111/j.1365-2427.2004.01328.x
Biermann GC, Sealy SG (1982) Parental feeding of nestling Yellow Warblers in relation to brood size and prey availability. Auk 99:332–341
Blankenberg D et al (2010) Galaxy: a web-based genome analysis tool for experimentalists. Curr Protocols Mol Biol Supplement 19.10.1–19.10.21. Chapter 19. https://doi.org/10.1002/0471142727.mb1910s89
Bowser AK, Diamond AW, Addison JA (2013) From puffins to plankton: a DNA-based analysis of a seabird food chain in the northern Gulf of Maine. PLoS One 8:e83152. https://doi.org/10.1371/journal.pone.0083152
Busby DG, Sealy SG (1979) Feeding ecology of a population of nesting Yellow Warblers. Can J Zool 57:1670–1681
Caporaso JG et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336
Chao A et al (2014) Rarefaction and extrapolation with Hill numbers: a framework for sampling and estimation in species diversity studies. Ecol Monogr 84:45–67
Clare EL (2014) Molecular detection of trophic interactions: emerging trends, distinct advantages, significant considerations and conservation applications. Evol Appl 7:1144–1157
Clare EL, Barber BR, Sweeney BW, Hebert PD, Fenton MB (2011) Eating local: influences of habitat on the diet of little brown bats (Myotis lucifugus). Mol Ecol 20:1772–1780. https://doi.org/10.1111/j.1365-294X.2011.05040.x
Cody ML (1968) On the methods of resource division in grassland bird communities. Am Nat 102:107–147
Crisol-Martínez E, Moreno-Moyano LT, Wormington KR, Brown PH, Stanley D (2016) Using next-generation sequencing to contrast the diet and explore pest-reduction services of sympatric bird species in macadamia orchards in Australia. PLoS One 11:e0150159
Drohan PJ, Brittingham M, Bishop J, Yoder K (2012) Early trends in landcover change and forest fragmentation due to shale-gas development in Pennsylvania: a potential outcome for the northcentral Appalachians. Environ Manage 49:1061–1075. https://doi.org/10.1007/s00267-012-9841-6
Dudgeon D et al (2006) Freshwater biodiversity: importance, threats, status and conservation challenges. Biol Rev 81:163–182
Esling P, Lejzerowicz F, Pawlowski J (2015) Accurate multiplexing and filtering for high-throughput amplicon-sequencing. Nucl Acids Res 43:2513–2524
Evans M, Gow E, Roth RR, Johnson MS, Underwood TJ (2011) Wood Thrush (Hylocichla mustelina). In: Rodewald PG (ed) The Birds of North America. Cornell Lab of Ornithology, Ithaca
Giardine B et al (2005) Galaxy: a platform for interactive large-scale genome analysis. Genome Res 15:1451–1455
Goecks J, Nekrutenko A, Taylor J (2010) Galaxy: a comprehensive approach for supporting accessible, reproducible, and transparent computational research in the life sciences. Genome Biol 11:R86
Gotelli NJ, Hart EM, Ellison AM (2015) EcoSimR: Null model analysis for ecological data. R package version 0.1.0. http://github.com/gotellilab/EcoSimR
Gray LT (1993) Response of insectivorous birds to emerging aquatic insects in riparian habitats of a tallgrass prairie stream. Am Midl Nat 129:288–300
Hodges MF, Krementz DG (1996) Neotropical migratory breeding bird communities in riparian forests of different widths along the Altamaha River, Georgia. Wilson Bull 108:496–506
Holmes R, Black C, Sherry T (1979a) Comparative population bioenergetics of three insectivorous passerines in a deciduous forest. Condor 81:9–20
Holmes RT, Schultz JC, Nothnagle P (1979b) Bird predation on forest insects: an exclosure experiment. Science 206:462–463
Hsieh TC, Ma KH, Chao A (2016) iNEXT: iNterpolation and EXTrapolation for species diversity. R package version 2.0.12. http://chao.stat.nthu.edu.tw/blog/software-download/
Hurlbert SH (1978) The measurement of niche overlap and some relatives. Ecology 59:67–77
Jedlicka JA, Vo ATE, Almeida RP (2016) Molecular scatology and high-throughput sequencing reveal predominately herbivorous insects in the diets of adult and nestling Western Bluebirds (Sialia mexicana) in California vineyards. Auk 134:116–127
Jetz W, Thomas GH, Joy JB, Hartmann K, Mooers AO (2012) The global diversity of birds in space and time. Nature 491:444–448
Knopf FL, Johnson RR, Rich T, Samson FB, Szaro RC (1988) Conservation of riparian ecosystems in the United States. Wilson Bull 100:272–284
Knopf FL, Samson FB (1994) Scale perspectives on avian diversity in western riparian ecosystems. Conserv Biol 8:669–676
Krüger F, Clare EL, Greif S, Siemers BM, Symondson WOC, Sommer RS (2014) An integrative approach to detect subtle trophic niche differentiation in the sympatric trawling bat species Myotis dasycneme and Myotis daubentonii. Mol Ecol 23:3657–3671
Kruskal JB (1964) Multidimensional scaling by optimizing goodness of fit to a nonmetric hypothesis. Psychometrika 29:1–27
Levins R (1968) Evolution in changing environments. Princeton University Press, Princeton
MacArthur R (1958) Population ecology of some warblers of northeastern coniferous forests. Ecology 39:599–619
Marshall MR, Cooper RJ, DeCecco JA, Strazanac J, Butler L (2002) Effects of experimentally reduced prey abundance on the breeding ecology of the Red-eyed Vireo. Ecol Appl 12:261–280
Martin TE (1987) Food as a limit on breeding birds: a life-history perspective. Annu Rev Ecol Evol Syst 18:453–487
Martin TE, Scott J, Menge C (2000) Nest predation increases with parental activity: separating nest site and parental activity effects. Proc R Soc Lond B Biol Sci 267:2287–2293
Mattsson BJ, Master TL, Mulvihill RS, Robinson DW (2009) Louisiana Waterthrush (Parkesia motacilla). In: Rodewald PG (ed) The Birds of North America Online. Cornell Lab of Ornithology, Ithaca
Merritt RW, Cummins KW (2008) An introduction to the aquatic insects of North America, 4th edn. Kendall/Hunt, Dubuque
Mulvihill RS, Latta SC, Newell FL (2009) Temporal constraints on the incidence of double brooding in the Louisiana Waterthrush. Condor 111:341–348. https://doi.org/10.1525/cond.2009.080037
Mulvihill RS, Newell FL, Latta SC (2008) Effects of acidification on the breeding ecology of a stream-dependent songbird, the Louisiana waterthrush (Seiurus motacilla). Freshw Biol 53:2158–2169. https://doi.org/10.1111/j.1365-2427.2008.02032.x
Nagy LR, Holmes RT (2005) Food limits annual fecundity of a migratory songbird: an experimental study. Ecology 86:675–681
Nagy LR, Smith KG (1997) Effects of insecticide-induced reduction in lepidopteran larvae on reproductive success of Hooded Warblers. Auk 114:619–627
Nakano S, Miyasaka H, Kuhara N (1999) Terrestrial-aquatic linkages: riparian arthropod inputs alter trophic cascades in a stream food web. Ecology 80:2435–2441
Nakano S, Murakami M (2001) Reciprocal subsidies: dynamic interdependence between terrestrial and aquatic food webs. Proc Natl Acad Sci 98:166–170
Newton I (2004) Population limitation in migrants. Ibis 146:197–226. https://doi.org/10.1111/j.1474-919X.2004.00293.x
Nuttle T, Yerger EH, Stoleson SH, Ristau TE (2011) Legacy of top-down herbivore pressure ricochets back up multiple trophic levels in forest canopies over 30 years. Ecosphere 2:1–11
O’Halloran J, Gribbin SD, Tyler SJ, Ormerod SJ (1990) The ecology of dippers Cinclus cinclus (L.) in relation to stream acidity in upland Wales: time-activity budgets and energy expenditure. Oecologia 85:271–280
Oksanen J et al. (2017) vegan: Community Ecology Package. R package version 2.4.2. https://cran.r-project.org/package=vegan
Ormerod SJ, O’Halloran J, Gribbin SD, Tyler SJ (1991) The ecology of dippers Cinclus cinclus in relation to stream acidity in upland Wales: breeding performance, calcium physiology and nestling growth. J Appl Ecol 28:419–433
Pianka ER (1973) The structure of lizard communities. Annu Rev Ecol Syst 4:53–74
Polis GA, Anderson WB, Holt RD (1997) Toward an integration of landscape and food web ecology: the dynamics of spatially subsidized food webs. Annu Rev Ecol Syst 28:289–316
Pompanon F, Deagle BE, Symondson WO, Brown DS, Jarman SN, Taberlet P (2012) Who is eating what: diet assessment using next generation sequencing. Mol Ecol 21:1931–1950. https://doi.org/10.1111/j.1365-294X.2011.05403.x
Raley CM, Anderson SH (1990) Availability and use of arthropod food resources by Wilson’s Warblers and Lincoln’s Sparrows in southeastern Wyoming. Condor 92:141–150
Rappole JH, McDonald MV (1994) Cause and effect in population declines of migratory birds. Auk 111:652–660
Ratnasingham S, Hebert PDN (2007) BOLD: the barcode of life data system (www. barcodinglife.org). Mol Ecol Notes 7:355–364. https://doi.org/10.1111/j.1471-8286.2006.01678.x
Razgour O et al (2011) High-throughput sequencing offers insight into mechanisms of resource partitioning in cryptic bat species. Ecology Evol 1:556–570. https://doi.org/10.1002/ece3.49
Robbins CS, Sauer JR, Greenberg RS, Droege S (1989) Population declines in North American birds that migrate to the Neotropics. Proc Natl Acad Sci 86:7658–7662
Rodenhouse NL, Holmes RT (1992) Results of experimental and natural food reductions for breeding Black-throated Blue Warblers. Ecology 73:357–372
Rosenberg KV, Cooper RJ (1990) Approaches to avian diet analysis. Stud Avian Biol 80–90
Rosenberg KV, Ohmart RD, Anderson BW (1982) Community organization of riparian breeding birds: response to an annual resource peak. Auk 99:260–274
Rotenberry JT (1980) Dietary relationships among shrubsteppe passerine birds: competition or opportunism in a variable environment. Ecol Monogr 50:93–110
Saab V (1999) Importance of spatial scale to habitat use by breeding birds in riparian forests: a hierarchical analysis. Ecol Appl 9:135–151
Sauer JR, Hines JE, Fallon JE, Pardieck KL, Ziolkowski DJ, Link WA (2014) The North American Breeding Bird Survey, results and analysis 1966–2015. Version 2.07.2017, 01.30.2015 edn. USGS Patuxent Wildlife Research Center, Laurel
Sauer JR, Link WA (2011) Analysis of the North American Breeding Bird Survey using hierarchical models. Auk 128:87–98. https://doi.org/10.1525/auk.2010.09220
Schoener TW (1974) Resource partitioning in ecological communities. Science 185:27–39
Strong AM (2000) Divergent foraging strategies of two neotropical migrant warblers: implications for winter habitat use. Auk 117:381–392
Symondson WOC, Harwood JD (2014) Special issue on molecular detection of trophic interactions: unpicking the tangled bank. Mol Ecol 23:3601–3604
Trevelline BK, Latta SC, Marshall LC, Nuttle T, Porter BA (2016) Molecular analysis of nestling diet in a long-distance Neotropical migrant, the Louisiana Waterthrush (Parkesia motacilla). Auk 133:415–428. https://doi.org/10.1642/auk-15-222.1
Vannote RL, Minshall GW, Cummins KW, Sedell JR, Cushing CE (1980) The river continuum concept. Can J Fish Aquat Sci 37:130–137
Vickery J (1992) The reproductive success of the dipper Cinclus cinclus in relation to the acidity of streams in south-west Scotland. Freshw Biol 28:195–205
Vo AT, Jedlicka JA (2014) Protocols for metagenomic DNA extraction and Illumina amplicon library preparation for faecal and swab samples. Mol Ecol Resour 14:1183–1197. https://doi.org/10.1111/1755-0998.12269
Wallace JB, Eggert SL, Meyer JL, Webster JR (1997) Multiple trophic levels of a forest stream linked to terrestrial litter inputs. Science 277:102–104
Whitaker DM, Carroll AL, Montevecchi WA (2000) Elevated numbers of flying insects and insectivorous birds in riparian buffer strips. Can J Zool 78:740–747
Whitehead DR, Taylor T (2002) Acadian Flycatcher (Empidonax virescens). In: Rodewald PG (ed) The Birds of North America. Cornell Lab of Ornithology, Ithaca
Wiens JA (1977) On competition and variable environments: populations may experience “ecological crunches” in variable climates, nullifying the assumptions of competition theory and limiting the usefulness of short-term studies of population patterns. Am Sci 65:590–597
Wiens JA, Rotenberry JT (1979) Diet niche relationships among North American grassland and shrubsteppe birds. Oecologia 42:253–292
Wiesenborn WD, Heydon SL (2007) Diets of breeding southwestern Willow Flycatchers in different habitats. Wilson J Ornithol 119:547–557
Wood PB, Frantz MW, Becker DA (2016) Louisiana Waterthrush and benthic macroinvertebrate response to shale gas development. J Fish Wildl Manag 7:423–433
Zeale MR, Butlin RK, Barker GL, Lees DC, Jones G (2011) Taxon-specific PCR for DNA barcoding arthropod prey in bat faeces. Mol Ecol Resour 11:236–244. https://doi.org/10.1111/j.1755-0998.2010.02920.x
Acknowledgements
We thank the Carnegie Museum of Natural History, Cokie Lindsay, and Dr. John Wenzel for coordinating access to study sites and providing accommodations at Powdermill Nature Reserve. We thank Eduardo Anaya, Thomas Cordray, Danilo Mejía, Michael Miles, Maria Paulino, and Youstina Seliman for field assistance and the Genomics Facility of the Biotechnology Resource Center at Cornell University (Ithaca, NY) for conducting Illumina sequencing. This research was supported by grants from the American Ornithological Society (formerly the American Ornithologists’ Union), Carnegie Museum of Natural History, National Aviary, and National Science Foundation (DEB-1349870 to Tim Nuttle). We thank the Bayer School of Natural and Environmental Sciences at Duquesne University for supporting undergraduate field technicians through a research fellowship. We thank Powdermill Nature Reserve (Carnegie Museum of Natural History) for supporting B.K.T. through the Rea Research Fellowship and the Bayer School of Natural and Environmental Sciences (Duquesne University) for supporting B.K.T. through a teaching assistantship and the Bayer Graduate Research Fellowship.
Author information
Authors and Affiliations
Contributions
B.K.T., T.N., B.A.P., and S.C.L. collectively conceived and designed this study as an extension of S.C.L.’s long-term research on Louisiana Waterthrush. B.K.T. and B.D.H. conducted the fieldwork (with guidance from T.N., B.A.P., and S.C.L.); B.K.T. developed the field and laboratory protocols, conducted molecular work (with B.D.H.), performed statistical analyses (with N.L.B.), and wrote the manuscript in the laboratory of B.A.P.
Corresponding author
Ethics declarations
Ethical approval
All applicable institutional and/or national guidelines for the care and use of animals were followed.
Additional information
Communicated by Indrikis Krams.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Trevelline, B.K., Nuttle, T., Hoenig, B.D. et al. DNA metabarcoding of nestling feces reveals provisioning of aquatic prey and resource partitioning among Neotropical migratory songbirds in a riparian habitat. Oecologia 187, 85–98 (2018). https://doi.org/10.1007/s00442-018-4136-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-018-4136-0