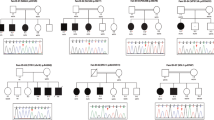
Abstract
Intellectual disability (ID) is a common morbid condition with a wide range of etiologies. The list of monogenic forms of ID has increased rapidly in recent years thanks to the implementation of genomic sequencing techniques. In this study, we describe the phenotypic and genetic findings of 68 families (105 patients) all with novel ID-related variants. In addition to established ID genes, including ones for which we describe unusual mutational mechanism, some of these variants represent the first confirmatory disease–gene links following previous reports (TRAK1, GTF3C3, SPTBN4 and NKX6-2), some of which were based on single families. Furthermore, we describe novel variants in 14 genes that we propose as novel candidates (ANKHD1, ASTN2, ATP13A1, FMO4, MADD, MFSD11, NCKAP1, NFASC, PCDHGA10, PPP1R21, SLC12A2, SLK, STK32C and ZFAT). We highlight MADD and PCDHGA10 as particularly compelling candidates in which we identified biallelic likely deleterious variants in two independent ID families each. We also highlight NCKAP1 as another compelling candidate in a large family with autosomal dominant mild intellectual disability that fully segregates with a heterozygous truncating variant. The candidacy of NCKAP1 is further supported by its biological function, and our demonstration of relevant expression in human brain. Our study expands the locus and allelic heterogeneity of ID and demonstrates the power of positional mapping to reveal unusual mutational mechanisms.




Similar content being viewed by others
Change history
29 December 2017
Variant nomenclature discrepancy was identified in the article.
References
Abouelhoda M, Faquih T, El-Kalioby M, Alkuraya FS (2016) Revisiting the morbid genome of Mendelian disorders. Genome Biol 17:235
Alazami AM, Patel N, Shamseldin HE, Anazi S, Al-Dosari MS, Alzahrani F, Hijazi H, Alshammari M, Aldahmesh MA, Salih MA (2015) Accelerating novel candidate gene discovery in neurogenetic disorders via whole-exome sequencing of prescreened multiplex consanguineous families. Cell Rep 10:148–161
Aldahmesh MA, Khan AO, Mohamed J, Alkuraya FS (2011) Novel recessive BFSP2 and PITX3 mutations: insights into mutational mechanisms from consanguineous populations. Genet Med 13:978–981
Aldahmesh MA, Mohammed JY, Al-Hazzaa S, Alkuraya FS (2012) Homozygous null mutation in ODZ3 causes microphthalmia in humans. Genet Med 14:900–904
Anazi S, Alshammari M, Moneis D, Abouelhoda M, Ibrahim N, Alkuraya FS (2016) Confirming the candidacy of THOC6 in the etiology of intellectual disability. Am J Med Genet A 170A(5):1367–1369
Anazi S, Maddirevula S, Faqeih E, Alsedairy H, Alzahrani F, Shamseldin H, Patel N, Hashem M, Ibrahim N, Abdulwahab F (2017) Clinical genomics expands the morbid genome of intellectual disability and offers a high diagnostic yield. Mol Psychiatry 22(4):615–624
Arnold K, Bordoli L, Kopp J, Schwede T (2006) The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22:195–201
Barel O, Malicdan CV, Ben-Zeev B, Kandel J, Pri-Chen H, Stephen J, Castro IG, Metz J, Atawa O, Moshkovitz S (2017) Deleterious variants in TRAK1 disrupt mitochondrial movement and cause fatal encephalopathy. Brain 140:568–581
Bhalla K, Luo Y, Buchan T, Beachem MA, Guzauskas GF, Ladd S, Bratcher SJ, Schroer RJ, Balsamo J, DuPont BR (2008) Alterations in CDH15 and KIRREL3 in patients with mild to severe intellectual disability. Am J Hum Genet 83:703–713
Bhasin TK, Brocksen S, Avchen RN, Braun KVN (2006) Prevalence of four developmental disabilities among children aged 8 years: Metropolitan Atlanta Developmental Disabilities Surveillance Program, 1996 and 2000. US Department of Health and Human Services, Centers for Disease Control and Prevention
Boycott KM, Rath A, Chong JX, Hartley T, Alkuraya FS, Baynam G, Brookes AJ, Brudno M, Carracedo A, den Dunnen JT (2017) International cooperation to enable the diagnosis of all rare genetic diseases. Am J Hum Genet 100:695–705
Callicott JH, Feighery EL, Mattay VS, White MG, Chen Q, Baranger DA, Berman KF, Lu B, Song H, Ming G-L (2013) DISC1 and SLC12A2 interaction affects human hippocampal function and connectivity. J Clin Investig 123:2961–2964
Chelban V, Patel N, Vandrovcova J, Zanetti MN, Lynch DS, Ryten M, Botía JA, Bello O, Tribollet E, Efthymiou S (2017) Mutations in NKX6-2 cause progressive spastic ataxia and hypomyelination. Am J Hum Genet 100:969–977
Del Villar K, Miller CA (2004) Down-regulation of DENN/MADD, a TNF receptor binding protein, correlates with neuronal cell death in Alzheimer’s disease brain and hippocampal neurons. Proc Natl Acad Sci 101:4210–4215
Dzhala VI, Talos DM, Sdrulla DA, Brumback AC, Mathews GC, Benke TA, Delpire E, Jensen FE, Staley KJ (2005) NKCC1 transporter facilitates seizures in the developing brain. Nat Med 11:1205–1213
Gilissen C, Hehir-Kwa JY, Thung DT, van de Vorst M, van Bon BW, Willemsen MH, Kwint M, Janssen IM, Hoischen A, Schenck A (2014) Genome sequencing identifies major causes of severe intellectual disability. Nature 511(7509):344–347
Group, S.M (2015) Comprehensive gene panels provide advantages over clinical exome sequencing for Mendelian diseases. Genome Biol 16:1–14
Harripaul R, Vasli N, Mikhailov A, Rafiq MA, Mittal K, Windpassinger C, Sheikh T, Noor A, Mahmood H, Downey S et al (2017) Mapping autosomal recessive intellectual disability: combined microarray and exome sequencing identifies 26 novel candidate genes in 192 consanguineous families. Mol Psychiatry. doi:10.1038/mp.2017.60
Hermann R (2014) Regulation of neural progenitor proliferation by ANKHD1. Doctorate Dissertation. doi:10.11588/heidok.00016467
Källberg M, Margaryan G, Wang S, Ma J, Xu J (2014) RaptorX server: a resource for template-based protein structure modeling. In: Kihara D (ed) Protein structure prediction. Methods in molecular biology (Methods and protocols), vol 1137. Humana Press, New York, NY
Knierim E, Gill E, Seifert F, Morales-Gonzalez S, Unudurthi SD, Hund TJ, Stenzel W, Schuelke M (2017) A recessive mutation in beta-IV-spectrin (SPTBN4) associates with congenital myopathy, neuropathy, and central deafness. Hum Genet 136(7):903–910
Leonard H, Wen X (2002) The epidemiology of mental retardation: challenges and opportunities in the new millennium. Mental Retard Dev Disabil Res Rev 8:117–134
Machicoane M, de Frutos CA, Fink J, Rocancourt M, Lombardi Y, Garel S, Piel M, Echard A (2014) SLK-dependent activation of ERMs controls LGN–NuMA localization and spindle orientation. J Cell Biol 205:791–799
Merner ND, Mercado A, Khanna AR, Hodgkinson A, Bruat V, Awadalla P, Gamba G, Rouleau GA, Kahle KT (2016) Gain-of-function missense variant in SLC12A2, encoding the bumetanide-sensitive NKCC1 cotransporter, identified in human schizophrenia. J Psychiatr Res 77:22–26
Miller DT, Adam MP, Aradhya S, Biesecker LG, Brothman AR, Carter NP, Church DM, Crolla JA, Eichler EE, Epstein CJ (2010) Consensus statement: chromosomal microarray is a first-tier clinical diagnostic test for individuals with developmental disabilities or congenital anomalies. Am J Hum Genet 86:749–764
Molumby MJ, Anderson RM, Newbold DJ, Koblesky NK, Garrett AM, Schreiner D, Radley JJ, Weiner JA (2017) γ-protocadherins interact with neuroligin-1 and negatively regulate dendritic spine morphogenesis. Cell Rep 18:2702–2714
Monies D, Abouelhoda M, AlSayed M, Alhassnan Z, Alotaibi M, Kayyali H, Al-Owain M, Shah A, Rahbeeni Z, Al-Muhaizea MA et al (2017a) The landscape of genetic diseases in Saudi Arabia based on the first 1000 diagnostic panels and exomes. Hum Genet 136(8):921–939
Monies D, Maddirevula S, Kurdi W, Alanazy MH, Alkhalidi H, Al-Owain M, Sulaiman RA, Faqeih E, Goljan E, Ibrahim N et al (2017b) Autozygosity reveals recessive mutations and novel mechanisms in dominant genes: implications in variant interpretation. Genet Med. doi:10.1038/glm.2017.22
Nakajima K-I, Miyazaki H, Niisato N, Marunaka Y (2007) Essential role of NKCC1 in NGF-induced neurite outgrowth. Biochem Biophys Res Commun 359:604–610
Patel N, Faqeih E, Anazi S, Alfawareh M, Wakil SM, Colak D, Alkuraya FS (2015) A novel APC mutation defines a second locus for Cenani-Lenz syndrome. J Med Genet 52(5):317–321
Perland E, Lekholm E, Eriksson MM, Bagchi S, Arapi V, Fredriksson R (2016) The putative SLC transporters Mfsd5 and Mfsd11 are abundantly expressed in the mouse brain and have a potential role in energy homeostasis. PLoS One 11:e0156912
Reuter MS, Tawamie H, Buchert R, Gebril OH, Froukh T, Thiel C, Uebe S, Ekici AB, Krumbiegel M, Zweier C (2017) Diagnostic yield and novel candidate genes by exome sequencing in 152 consanguineous families with neurodevelopmental disorders. JAMA Psychiatry 74:293–299
Riazuddin S, Hussain M, Razzaq A, Iqbal Z, Shahzad M, Polla DL, Song Y, van Beusekom E, Khan A, Tomas-Roca L et al (2016) Exome sequencing of Pakistani consanguineous families identifies 30 novel candidate genes for recessive intellectual disability. Mol Psychiatry. doi:10.1038/mp.2016.109
Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, Grody WW, Hegde M, Lyon E, Spector E (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17:405–423
Shamseldin HE, Maddirevula S, Faqeih E, Ibrahim N, Hashem M, Shaheen R, Alkuraya FS (2017) Increasing the sensitivity of clinical exome sequencing through improved filtration strategy. Genet Med 19(5):593–598
Sobreira N, Schiettecatte F, Valle D, Hamosh A (2015) GeneMatcher: a matching tool for connecting investigators with an interest in the same gene. Hum Mutat 36(10):928–930
Strande NT, Riggs ER, Buchanan AH, Ceyhan-Birsoy O, DiStefano M, Dwight SS, Goldstein J, Ghosh R, Seifert BA, Sneddon TP (2017) Evaluating the clinical validity of gene-disease associations: an evidence-based framework developed by the Clinical Genome Resource. Am J Hum Genet 100(6):895–906
Study, T.D.D.D (2015) Large-scale discovery of novel genetic causes of developmental disorders. Nature 519:223–228
Suzuki T, Nishiyama K, Yamamoto A, Inazawa J, Iwaki T, Yamada T, Kanazawa I, Sakaki Y (2000) Molecular cloning of a novel apoptosis-related gene, human Nap1 (NCKAP1), and its possible relation to Alzheimer disease. Genomics 63:246–254
Tabarki B, AlMajhad N, AlHashem A, Shaheen R, Alkuraya FS (2016) Homozygous KCNMA1 mutation as a cause of cerebellar atrophy, developmental delay and seizures. Hum Genet 135:1295–1298
Tanaka M, Miyoshi J, Ishizaki H, Togawa A, Ohnishi K, Endo K, Matsubara K, Mizoguchi A, Nagano T, Sato M (2001) Role of Rab3 GDP/GTP exchange protein in synaptic vesicle trafficking at the mouse neuromuscular junction. Mol Biol Cell 12:1421–1430
Thaxton C, Pillai AM, Pribisko AL, Labasque M, Dupree JL, Faivre-Sarrailh C, Bhat MA (2010) In vivo deletion of immunoglobulin domains 5 and 6 in neurofascin (Nfasc) reveals domain-specific requirements in myelinated axons. J Neurosci 30:4868–4876
Wu X, Bradley MJ, Cai Y, Kümmel D, Enrique M, Barr FA, Reinisch KM (2011) Insights regarding guanine nucleotide exchange from the structure of a DENN-domain protein complexed with its Rab GTPase substrate. Proc Natl Acad Sci 108:18672–18677
Xu D, Zhang Y (2012) Ab initio protein structure assembly using continuous structure fragments and optimized knowledge-based force field. Proteins: structure. Funct Bioinform 80:1715–1735
Acknowledgements
We thank the study families for their enthusiastic participation. This work was supported in part by King Salman Center for Disability Research (FSA). We acknowledge the support of the Saudi Human Genome Program and the Sequencing and Genotyping Core Facilities at KFSRHC. The research by STA reported in this publication was supported by funding from King Abdullah University of Science and Technology (KAUST).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Additional information
A correction to this article is available online at https://doi.org/10.1007/s00439-017-1859-7.
Electronic supplementary material
Below is the link to the electronic supplementary material.
439_2017_1843_MOESM1_ESM.pdf
Figure S1. A) Clinical features of patient with GTF3C3-related ID showing facial asymmetry, bilateral temporal narrowing, epicanthal folds, upslanting palpebral fissures, bulbous nose, and full cheeks. (C) Chromatogram showing skipping of exon10 and partial part of exon11 (PDF 164 kb)
Rights and permissions
About this article
Cite this article
Anazi, S., Maddirevula, S., Salpietro, V. et al. Expanding the genetic heterogeneity of intellectual disability. Hum Genet 136, 1419–1429 (2017). https://doi.org/10.1007/s00439-017-1843-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-017-1843-2