Abstract
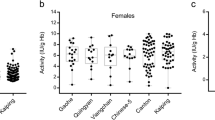
A systematic study on the structure and function of Glucose-6-phosphate dehydrogenase (G6PD) variations was carried out in China. A total of 155,879 participants were screened for G6PD deficiency by the G6PD/6PGD ratio method and 6,683 cases have been found. The prevalence of G6PD deficiency ranged from 0 to 17.4%. With informed consent, 1,004 cases from 11 ethnic-based groups were subjected to molecular analysis. Our results showed the followings: (1) The G6PD variants are consistent across traditional ethnic boundaries, but vary in frequencies across ethnic-based groups in Chinese population, (2) The G6PD variants in Chinese population are different from those in African, European, and Indian populations, (3) A novel G6PD-deficiency mutation, 274C→T, has been found, and (4) Denaturing high performance liquid chromatography is of great advantage to detecting G6PD-deficient mutations for diagnosis and genetic counseling. Moreover, functional analysis of the human G6PD variants showed the following: (1) The charge property, polarity, pK-radical and side-chain radical of the substituting amino acid have an effect on G6PD activity, (2) The G6PDArg459 and Arg463 play important roles in anchoring NADP+ to the catalytic domain to maintain the enzymatic activity, and (3) The sequence from codon 459 to the carboxyl terminal is essential for the enzymatic function.






Similar content being viewed by others
References
Ainoon O, Joyce J, Boo NY, Cheong SK, Zainal ZA, Hamidah NH (1999) Glucose-6-phosphate dehydrogenase (G6PD) variants in Malaysian Chinese. Hum Mutat 14:352
Au SW, Gover S, Lam VM, Adams MJ (2000) Human glucose-6-phosphate dehydrogenase: the crystal structure reveals a structural NADP(+) molecule and provides insights into enzyme deficiency. Structure Fold Des 8:293–303
Beutler E (1993) Study of glucose-6-phosphate dehydrogenase: History and molecular biology. Am J Hematol 42:53–56
Beutler E (1994) G6PD deficiency. Blood 84:3613–3636
Beutler E, Blume KG, Kaplan JC, Lohr GW, Ramot B, Valentine WN (1977) International Committee for Standardization in Haematology: recommended methods for redcell enzyme analysis. Br J Haematol 35:331–340
Beutler E, Blume KG, Kaplan JC, Lohr GW, Ramot B, Valentine WN (1979) International Committee for Standardizationin Haematology: recommended screening test for glucose-6-phosphate dehydrogenase (G6PD) deficiency. Br J Haematol 43:465–467
Calabro V, Mason PJ, Filosa S, Civitelli D, Cittadella R, Tagarelli A, Martini G, Brancati C, Luzzatto L (1993) Genetic heterogeneity of glucose-6-phosphate dehydrogenase deficiency revealed by single-strand conformation and sequence analysis. Am J Hum Genet. 5:527–536
Cai W, Cai L, Zhou D, Kuang Y, Zhou Y, Stefania F, Giuseppe M (2000) 1376 G→T mutation of G6PD gene in Han and Li nationalities in Hainan, China. Zhonghua Yi Xue Yi Chuan Xue Za Zhi 17:326–328
Cai W, Filosa S, Martini G, Zhou Y, Zhou D, Cai L, Kuang Y (2001) Molecular characterization of glucose6-phosphate dehydrogenase deficiency in the Han and Li nationalities in Hainan, China and identification of a new mutation in human G6PDgene. Zhonghua Yi Xue Yi Chuan Xue Za Zhi 18:105–109
Chau TN, Lai ST, Lai JY, Yuen H (1997) Haemolysis complicating acute viral hepatitis in patients with normal or deficient glucose-6-phosphate dehydrogenase activity. Scand J Infect Dis 29:551–553
Chen HL, Huang MJ, Huang CS, Tang TK (1996) G6PD NanKang (517 T→C; 173 Phe→Leu): a new Chinese G6PD variant associated with neonatal jaundice. Hum Hered 46:201–204
Chen HL, Huang MJ, Huang CS, Tang TK (1997) Two novel glucose 6-phosphate dehydrogenase deficiency mutations and association of such mutations with F8C/G6PD haplotype in Chinese. J Formos Med Assoc 96:948–954
Chiang SH, Wu SJ, Wu KF, Hsiao KJ (1999) Neonatal screening for glucose-6-phosphate dehydrogenase deficiency in Taiwan. Southeast Asian J Trop Med Public Health 2:72–274
Costa E, Cabeda JM, Vieira E, Pinto R, Pereira SA, Ferraz L, Santos R, Barbot J (2000) Glucose-6-phosphate dehydrogenase aveiro: a de novo mutation associated with chronic nonspherocytic hemolytic anemia. Blood 95:1499–1501
Drent M, Gorgels AP, Bast A (2003) Cardiac failure associated with G6PD deficiency. Circ Res 93:e75
Du C, He Y (1997) A case of nt 1004C→ A G6PD gene mutation in Yunnan Han people. Zhonghua Xue Ye Xue Za Zhi 18:535–537
Du CS, Ren X, Chen L, Jiang W, He Y, Yang M (1999) Detection the most common G6PDgene mutation in Chinese using amplication refractory mutation system. Hum Hered 49:133–138
Du CS, Xu YK, Hu XY (1987) Favism. People’s Medical Publishing House, Bejing, China, pp 189
Filosa S, Fico A, Paglialunga F, Balestrieri M, Crooke A, Verde P, Abrescia P, Bautista JM, Martini G (2003) Failure to increase glucose consumption through the pentose-phosphate pathway results in the death of glucose-6-phosphate dehydrogenase gene-deleted mouse embryonic stem cells subjected to oxidative stress. Biochem J 370:(3):935–943
Fouts D, Ganguly R, Gutierrez AG, Lucchesi JC, Manning JE (1988) Nucleotide sequence of the Drosophila glucose-6-phosphate dehydrogenase gene and comparison with the homologous human gene. Gene 31:261–275
Ho YS, Howard AJ, Crapo JD (1988) Cloning and sequence of a cDNA encoding rat glucose-6-phosphate dehydrogenase. Nucleic Acids Res 16:7746–7751
Iwai K, Hirono A, Matsuoka H, Kawamoto F, Horie T, Lin K, Tantular IS, Dachlan YP, Notopuro H, Hidayaho NI, Abdul MA, Fujii SH, Miwa S, Ishii A (2001) Distribution of glucose 6 phosphate dehydrogenase mutations in Southeast Asia. Hum Genet 108:445–449
Jain M, Brenner DA, Cui L, Lim CC, Wang B, Pimentel DR, Koh S, Sawyer DB, Leopold JA, Handy DE, Loscalzo J, Apstein CS, Liao R (2003) Glucose-6-phosphate ehydrogenased modulates cytosolic redox status and contractile phenotype in adult cardiomyocytes. Circ Res 93:e9–e16
Jiang W, Du C, Chen L (1999) Study on G487A mutation of the glucose -6-phosphate dehydrogenase gene. Zhonghua Xue Ye Xue Za Zhi 20:518–520
Jiang W, Du C, Duan S, Shan MLD, Chen L, Lin Q, Liwen Y (1999) Molecular characterization of glucose-6-phosphate dehydrogenase variants in four ethnic groups in Yunnan province of China. Zhong hua Yi Xue Yi Chuan Xue Za Zhi 16:149–152
Kneller DG, Cohen FE, Langridge R (1990) Improvements in protein secondary structure prediction by an enhanced neural network. J Mol Biol 214:171–182
Kotaka M, Gover S, Vandeputte-Rutten L, Au SW, Lam VM, Adams MJ (2005) Structural studies of glucose-6-phosphate and NADP+ binding to human glucose-6-phosphate dehydrogenase. Acta Crystallogr D Biol Crystallogr 61:495–504
Kwiatkowski DP (2005) How malaria has affected the human genome and what human genetics can teach us about malaria. Am J Hum Genet 77:171–190
Lo YS, Lu CC, Chiou SS, Chen BH, Chang TT, Chang JG (1994) Molecular characterization of glucose-6phosphate dehydrogenase deficiency in Chinese infants with or without severe neonatal hyperbilirubinaemia. Br J Haematol 86:858–862
Matsuoka H, Nguon C, Kanbe T, Jalloh A, Sato H, Yoshida S, Hirai M, Arai M, Socheat D, Kawamoto F (2005) Glucose-6-phosphate dehydrogenase (G6PD) mutations in Cambodia: G6PD Viangchan (871G→A) is the most common variant in the Cambodian population. J Hum Genet 50:468–472
Mohanty D, Mukherjee MB, Colah RB (2004) Glucose-6-phosphate dehydrogenase deficiency in India. Indian J Pediatr 71:525–529
Nikitin EV, Pak SG (1990) The functioning of the glutathione system in patients with acute viral hepatitis. 62:76–79
Ren X, He Y, Du C, Jiang W, Chen L, Lin Q (2001) A novel mis-sense mutation (G1381A) in the G6PD gene identified in a Chinese man. Chin Med J (Engl) 114:399–401
Rodrigues MO, Pereira JD, Gaspar G, Olim G, Martins MD, Monteiro C (2004) Novel point mutation in exon 12 of the glucose-6- phosphate dehydrogenase gene: G6PD FLORES. J Clin Lab Anal 18:129–131
Sabeti PC, Reich DE, Higgins JM, Levine HZ, Richter DJ, Schaffner SF, Gabriel SB, Platko JV, Patterson NJ, McDonald GJ, Ackerman HC, Campbell SJ, Altshuler D, Cooper R, Kwiatkowski D, Ward R, Lander ES (2002) Detecting recent positive selection in the human genome from haplotype structure. Nature 419:832–837
Saunders MA, Hammer MF, Nachman MW (2002) Nucleotide variability at G6pd and the signature of malarial selection in humans. Genetics 162:1849–1861
Saunders MA, Slatkin M, Garner C, Hammer MF, Nachman MW (2005) The extent of linkage disequilibrium caused by selection on G6PD in humans. Genetics 171:1219–1229
Sabeti PC, Reich DE, Higgins JM, Levine HZ, Richter DJ, Schaffner SF, Gabriel SB, Platko JV, Patterson NJ, McDonald GJ, Ackerman HC, Campbell SJ, Altshuler D, Cooper R, Kwiatkowski D, Ward R, Lander ES (2002) Detecting recent positive selection in the human genome from haplotype structure. Nature 419:832–837
Scopes DA, Bautista JM, Naylor CE, Adams MJ, Mason PJ (1998) Amino acid substitutions at the dimer interface of human glucose-6-phosphate dehydrogenase that increase thermostability and reduce the stabilising effect of NADP. Eur J Biochem 251:382–388
Siwik DA, Pagano PJ, Colucci WS (2001) Oxidative stress regulates collagen synthesis and matrix metalloproteinase activity in cardiac fibroblasts. Am J Physiol Cell Physiol 280:C53–C60
Sobngwi E, Gautier JF, Kevorkian JP, Villette JM, Riveline JP, Zhang S, Vexiau P, Leal SM, Vaisse C, Mauvais-Jarvis F (2005) High prevalence of glucose-6-phosphate dehydrogenase deficiency without gene mutation suggests a novel genetic mechanism predisposing to ketosis-prone diabetes. 90:4446–4451
Sukumar S, Mukherjee MB, Colah RB, Mohanty D (2004) Molecular basis of G6PD deficiency in India. Blood Cells Mol Dis 33:141–145
Verrelli BC, McDonald JH, Argyropoulos G, Destro-Bisol G, Froment A, Drousiotou A, Lefranc G, Helal AN, Loiselet J, Tishkoff SA (2002) Evidence for balancing selection from nucleotide sequence analyses of human G6PD. Am J Hum Genet 71:1112–1128
WHO Scientific Groups (1967) Standardization of procedures for the study of glucose-6-phosphate dehydrogenase. Report of a WHO Scientific Group. World Health Organ Tech Rep Ser 366:1–53
Wajcman H, Galacteros F (2004) Glucose-6-phosphate dehydrogenase deficiency: a protection against malaria and a risk for hemolytic accidents. C R Biol 327:711–720
Wang XT, Au SW, Lam VM, Engel PC (2002) Recombinant human glucose-6-phosphate dehydrogenase. Evidence for a rapid-equilibrium random-order mechanism. Eur J Biochem 269:3417–3424
WHO Scientific Groups (1967) Standardization of procedures for the study of glucose-6-phosphate dehydrogenase. Report of a WHO Scientific Group. World Health Organ Tech Rep Ser 366:1–53
Xiao W, Oefner PJ (2001) Denaturing high-performance liquid chromatography: A review. Hum Mutat 17:439–474
Xu WM, Wang Q, Huan XY (1994) PCR-siungle-strand comformation (SSCP), DNA direct sequencing analysis in detecting mutation in exon 2 of G6PDgene. Zhonbghua Yi Xue Zazhi 74:35–37
Yang Z, Chu J, Ban G (2001) The genotype analysis of glucose-6-phosphate dehydrogenase deficiency in Yunnan province. Zhonghua Yi Xue Yi Chuan Xue Za Zhi 18:259–263
Yu GL, Jiang WY, DU CS, Chen LM, Lin QD, Tian QH, Zeng JB, Li SG (2004) Identification of G6PD gene variants from Hakka population in Guangdong province. Zhonghua Yi Xue Yi Chuan Xue Za Zhi 21:448–451
Acknowledgments
The mutated G6PD by site-directed mutagenesis was a kind gift from Professor Chongyi Zhao, who worked in Furen University, Taiwan. Many thanks to Professor Huishang Au for her guiding while at the Kunming University of Science Technology, China. The project is supported by Chinese National Natural Scientific Grants #30440005 and #30470949, China; Scientific Technology Grant of Guangdong province: #2004B3370107, China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, W., Yu, G., Liu, P. et al. Structure and function of glucose-6-phosphate dehydrogenase-deficient variants in Chinese population. Hum Genet 119, 463–478 (2006). https://doi.org/10.1007/s00439-005-0126-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00439-005-0126-5