Abstract
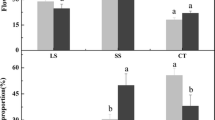
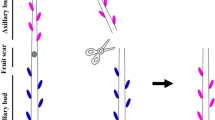
Although gibberellin (GA) has been reported to control branching, little is known about how GA mediates signals regulating the outgrowth of axillary buds (ABs). In the current study, the effect of the exogenous application of 5.0 mM GA3 on ABs outgrowth on 1-year-old ‘Nagafu No. 2’/T337/M. robusta Rehd. apple trees was investigated and compared to the bud-activating treatments, 5 mM BA or decapitation. Additionally, the expression of genes related to bud-regulating signals and sucrose levels in ABs was examined. Results indicated that GA3 did not promote ABs’ outgrowth, nor down-regulate the expression of branching repressors [MdTCP40, MdTCP33, and MdTCP16 (homologs of BRANCHED1 and BRC2)], which were significantly inhibited by the BA and decapitation treatments. MdSBP12 and MdSBP18, the putative transcriptional activators of these genes, which are expressed at lower levels in BA-treated and decapitated buds, were up-regulated in the GA3 treatment in comparison to the BA treatment. Additionally, GA3 did not up-regulate the expression of CK response- and auxin transport-related genes, which were immediately induced by the BA treatment. In addition, GA3 also up-regulated the expression of several Tre6P biosynthesis genes and reduced sucrose levels in ABs. Sucrose levels, however, were still higher than what was observed in BA-treated buds, indicating that sucrose may not be limiting in GA3-controlled AB outgrowth. Although GA3 promoted cell division, it was not sufficient to induce AB outgrowth. Conclusively, some branching-inhibiting genes and bud-regulating hormones are associated with the inability of GA3 to activate AB outgrowth.









Similar content being viewed by others
References
Agharkar M, Lomba P, Altpeter F, Zhang H, Kenworthy K, Lange T (2007) Stable expression of AtGA2ox1 in a low-input turfgrass (Paspalum notatum Flugge) reduces bioactive gibberellin levels and improves turf quality under field conditions. Plant Biotechnol J 5:791–801
Aguilarmartínez JA, Pozacarrión C, Cubas P (2007) Arabidopsis BRANCHED1 Acts as an integrator of branching signals within axillary buds. Plant Cell 19:458
Agusti J, Herold S, Schwarz M, Sanchez P, Ljung K, Dun EA, Brewer PB, Beveridge CA, Sieberer T, Sehr EM (2011) Strigolactone signaling is required for auxin-dependent stimulation of secondary growth in plants. Proc Natl Acad Sci USA 108:20242–20247
Argyros RD, Mathews DE, Chiang YH, Palmer CM, Thibault DM, Etheridge N, Argyros DA, Mason MG, Kieber JJ, Schaller GE (2008) Type B response regulators of Arabidopsis play key roles in cytokinin signaling and plant development. Plant Cell 20:2102–2116
Arite T, Iwata H, Ohshima K, Maekawa M, Nakajima M, Kojima M, Sakakibara H, Kyozuka J (2007) DWARF10, an RMS1/MAX4/DAD1 ortholog, controls lateral bud outgrowth in rice. Plant J 51:1019–1029
Atay E, Koyuncu F (2013) A new approach for augmenting branching of nursery trees and its comparison with other methods. Sci Hortic 160:345–350
Balla J, Kalousek P, Reinöhl V, Friml J, Procházka S (2011) Competitive canalization of PIN-dependent auxin flow from axillary buds controls pea bud outgrowth. Plant J Cell Mol Biol 65:571–577
Bennett T, Sieberer T, Willett B, Booker J, Luschnig C, Leyser O (2006) The Arabidopsis MAX pathway controls shoot branching by regulating auxin transport. Curr Biol 16:553–563
Bennett T, Hines G, Leyser O (2014) Canalization: what the flux? Trends Genet 30:41–48
Bergmann C, Wegmann K, Frischmuth K, Samson E, Kranz A, Weigelt D, Koll P, Welzel P (1993) Stimulation of Orobanche crenata seed germination by (+)-strigol and structural analogues dependence on constitution and configuration of the germinatio stimulants. J Plant Physiol 142:338–342
Bostan M (2010) Influence of crown formation method on development of the apple trees in the nursery. J Am Soc Hortic Sci 7:1193–1198
Boucheron E, Healy JC, Sauvanet A, Rembur J, Noin M, Sekine M, Riou KC, Murray JA, Van OH, Chriqui D (2005) Ectopic expression of Arabidopsis CYCD2 and CYCD3 in tobacco has distinct effects on the structural organization of the shoot apical meristem. J Exp Bot 56:123–134
Braun N, Germain ADS, Pillot JP, Boutet-Mercey S, Dalmais M, Antoniadi I, Xin L, Maia-Grondard A, Signor CL, Bouteiller N (2012) The pea TCP transcription factor PsBRC1 acts downstream of strigolactones to control shoot branching. Plant Physiol 158:225–238
Brewer PB, Beveridge CA (2009) Strigolactone acts downstream of auxin to regulate bud outgrowth in pea and Arabidopsis. Plant Physiol 150:482–493
Cheng H, Qin L, Lee S, Fu X, Richards DE, Cao D, Luo D, Harberd NP, Peng J (2004) Gibberellin regulates Arabidopsis floral development via suppression of DELLA protein function. Development 131:1055
Choubane D, Rabot A, Mortreau E, Legourrierec J, Péron T, Foucher F, Ahcène Y, Pelleschi-Travier S, Leduc N, Hamama L (2012) Photocontrol of bud burst involves gibberellin biosynthesis in Rosa sp. J Plant Physiol 169:1271–1280
Crawford S, Shinohara NT, Williamson L, George G, Hepworth J, Muller D, Domagalska MA, Leyser O (2010) Strigolactones enhance competition between shoot branches by dampening auxin transport. Development 137:2905
Daviere JM, Wild M, Regnault T, Baumberger N, Eisler H, Genschik P, Achard P (2014) Class I TCP-DELLA interactions in inflorescence shoot apex determine plant height. Curr Biol 24:1923–1928
Davière JM, Achard P (2013) Gibberellin signaling in plants. Development 140:1147–1151
De SGA, Ligerot Y, Dun EA, Pillot JP, Ross JJ, Beveridge CA, Rameau C (2013) Strigolactones stimulate internode elongation independently of gibberellins. Plant Physiol 163:1012–1025
Domagalska MA, Leyser O (2011) Signal integration in the control of shoot branching. Nat Rev Mol Cell Biol 12:211
Du L, Qi S, Ma J, Xing L, Fan S, Zhang S, Li Y, Shen Y, Zhang D, Han M (2017) Identification of TPS family members in apple (Malus × domestica Borkh.) and the effect of sucrose sprays on TPS expression and floral induction. Plant Physiol Biochem 120:10–23
Dun EA, Germain ADS, Rameau C, Beveridge CA (2012) Antagonistic action of strigolactone and cytokinin in bud outgrowth control. Plant Physiol 158:487
Dun EA, Germain ADS, Rameau C, Beveridge CA (2013) Dynamics of strigolactone function and shoot branching responses in Pisum sativum. Mol Plant 6:128
Elfving DC (2010) Plant bioregulators in the deciduous fruit tree nursery. XI Int Symp Plant Bioregul Fruit Prod 884:159–166
Elfving D, Visser D, Henry J (2011) Gibberellins stimulate lateral branch development in young sweet cherry trees in the orchard. Int J Fruit Sci 11:41–54
Fan S, Zhang D, Gao C, Zhao M, Wu H, Li Y, Shen Y, Han M (2017) Identification, classification, and expression analysis of GRAS gene family in Malus domestica. Front Physiol 8:253
Fichtner F, Barbier FF, Feil R, Watanabe M, Annunziata MG, Chabikwa TG, Hofgen R, Stitt M, Beveridge CA, Lunn JE (2017) Trehalose 6-phosphate is involved in triggering axillary bud outgrowth in garden pea (Pisum sativum L.). Plant J 92:611–623
Finlayson SA (2007) Arabidopsis teosinte Branched1-like 1 regulates axillary bud outgrowth and is homologous to monocot teosinte Branched1. Plant Cell Physiol 48:667
Foster T, Kirk C, Jones WT, Allan AC, Espley R, Karunairetnam S, Rakonjac J (2006) Characterisation of the DELLA subfamily in apple (Malus × domestica Borkh.). Tree Genet Genomes 3:187–197
François B, Thomas P, Marion L, Maria-Dolores PG, Quentin B, Jakub R, Stéphanie BM, Sylvie C, Remi L, Benoît P (2015) Sucrose is an early modulator of the key hormonal mechanisms controlling bud outgrowth in Rosa hybrida. J Exp Bot 66:2569
Gambino G, Perrone I, Gribaudo I (2008) A rapid and effective method for RNA extraction from different tissues of grapevine and other woody plants. Phytochem Anal 19:520–525
Ghosh A, Chikara J, Chaudhary DR (2011) Diminution of economic yield as affected by pruning and chemical manipulation of Jatropha curcas L. Biomass Bioenergy 35:1021–1029
Greenboim-Wainberg Y, Weiss D (2005) Cross talk between gibberellin and cytokinin: the Arabidopsis GA response inhibitor SPINDLY plays a positive role in cytokinin signaling. Plant Cell 17:92
Hao L, Wang RX, Qian Q, Yan MX, Meng XB, Fu ZM, Yan CY, Jiang B, Zhen S, Li JY (2009) DWARF27, an iron-containing protein required for the biosynthesis of strigolactones, regulates rice tiller bud outgrowth. Plant Cell 21:1512
Henry C, Rabot A, Laloi M, Mortreau E, Sigogne M, Leduc N, Lemoine R, Sakr S, Vian A, Pelleschitravier S (2011) Regulation of RhSUC2, a sucrose transporter, is correlated with the light control of bud burst in Rosa sp. Plant Cell Environ 34:1776–1789
Ho KM, English S, Bell J (1981) The control of the patterned differentiation of vascular tissues. Adv Bot Res 9:151–262
Holalu SV, Finlayson SA (2017) The ratio of red light to far red light alters Arabidopsis axillary bud growth and abscisic acid signalling before stem auxin changes. J Exp Bot 68:943–952
Itoh H, Ueguchitanaka M, Sato Y, Ashikari M, Matsuoka M (2002) The gibberellin signaling pathway is regulated by the appearance and disappearance of SLENDER RICE1 in nuclei. Plant Cell 14:57
Kebrom TH, Finlayson SA (2006) Phytochrome B represses Teosinte Branched1 expression and induces sorghum axillary bud outgrowth in response to light signals. Plant Physiol 140:1109
Kebrom TH, Mullet JE (2016) Transcriptome profiling of tiller buds provides new insights into PhyB regulation of tillering and indeterminate growth in Sorghum. Plant Physiol 170:2232–2250
Kebrom TH, Spielmeyer W, Finnegan EJ (2013) Grasses provide new insights into regulation of shoot branching. Trends Plant Sci 18:41–48
Kosugi S, Ohashi Y (1997) PCF1 and PCF2 specifically bind to cis elements in the rice proliferating cell nuclear antigen gene. Plant Cell 9:1607
Kviklys D (2006) Induction of feathering of apple planting material. Agron Vestis 9:58–63
Labuschagné IF, Louw JH, Schmidt K, Sadie A (2003) Selection for increased budbreak in apple. J Am Soc Hortic Sci 128:363–373
Leyser O (2009) The control of shoot branching: an example of plant information processing. Plant Cell Environ 32:694–703
Li C-J, Bangerth F (1999) Autoinhibition of indoleacetic acid transport in the shoots of two-branched pea (Pisum sativum) plants and its relationship to correlative dominance. Physiol Plant 106:415–420
Li C, Bangerth F (2003) Stimulatory effect of cytokinins and interaction with IAA on the release of lateral buds of pea plants from apical dominance. J Plant Physiol 160:1059–1063
Li J, Hou H, Li X, Jiang X, Yin X, Gao H, Zheng Y, Bassett CL, Wang X (2013) Genome-wide identification and analysis of the SBP-box family genes in apple (Malus × domestica Borkh.). Plant Physiol Biochem 70:100
Li Y, Zhang D, Zhang L, Zuo X, Fan S, Zhang X, Shalmani A, Han M (2017) Identification and expression analysis of cytokinin response-regulator genes during floral induction in apple (Malus domestica Borkh). Plant Growth Regul:1–10
Liu J, Cheng X, Liu P, Sun J (2017) miR156-targeted SBP-box transcription factors interact with DWARF53 to regulate TEOSINTE BRANCHED1 and BARREN STALK1 expression in bread wheat. Plant Physiol 174:1931
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-delta delta C(T)) method. Methods 25:402–408
Lo SF, Yang SY, Chen KT, Hsing YL, Zeevaart JAD, Chen LJ, Yu SM (2008) A novel class of gibberellin 2-oxidases control semidwarfism, tillering, and root development in rice. Plant Cell 20:2603–2618
Lu Z, Yu H, Xiong G, Wang J, Jiao Y, Liu G, Jing Y, Meng X, Hu X, Qian Q, Fu X, Wang Y, Li J (2013) Genome-wide binding analysis of the transcription activator ideal plant architecture1 reveals a complex network regulating rice plant architecture. Plant Cell 25:3743–3759
Lunn EJ, Feil R, Hendriks Janneke HM, Gibon Y, Morcuende R, Osuna D, Scheible W, Carillo P, Hajirezaei MR, Stitt M (2006) Sugar-induced increases in trehalose 6-phosphate are correlated with redox activation of ADP glucose pyrophosphorylase and higher rates of starch synthesis in Arabidopsis thaliana. Biochem J 397:139
Marínde lRN, Pfeiffer A, Hill K, Locascio A, Bhalerao RP, Miskolczi P, Grønlund AL, Wanchookohli A, Thomas SG, Bennett MJ (2015) Genome wide binding site analysis reveals transcriptional coactivation of cytokinin-responsive genes by DELLA proteins. Plos Genet 11:e1005337
Martin K, Vera M, Richard W, Brendan D (2011) TCP14 and TCP15 affect internode length and leaf shape in Arabidopsis. Plant J Cell Mol Biol 68:147–158
Martín-Trillo M, Grandío EG, Serra F, Marcel F, Rodríguez-Buey ML, Schmitz G, Theres K, Bendahmane A, Dopazo H, Cubas P (2011) Role of tomato BRANCHED1-like genes in the control of shoot branching. Plant J Cell Mol Biol 67:701
Mason MG, Ross JJ, Babst BA, Wienclaw BN, Beveridge CA (2014) Sugar demand, not auxin, is the initial regulator of apical dominance. Proc Natl Acad Sci USA 111:6092–6097
Mauriat M, Sandberg LG, Moritz T (2011) Proper gibberellin localization in vascular tissue is required to control auxin-dependent leaf development and bud outgrowth in hybrid aspen. Plant J Cell Mol Biol 67:805
Maymon I, Greenboim-Wainberg Y, Sagiv S, Kieber JJ, Moshelion M, Olszewski N, Weiss D (2009) Cytosolic activity of SPINDLY implies the existence of a DELLA-independent gibberellin-response pathway. Plant J Cell Mol Biol 58:979
Minakuchi K, Kameoka H, Yasuno N, Umehara M, Luo L, Kobayashi K, Hanada A, Ueno K, Asami T, Yamaguchi S (2010) FINE CULM1 (FC1) works downstream of strigolactones to inhibit the outgrowth of axillary buds in rice. Plant Cell Physiol 51:1127
Moubayidin L, Perilli S, Ioio RD, Mambro RD, Costantino P, Sabatini S (2010) The rate of cell differentiation controls the Arabidopsis root meristem growth phase. Curr Biol Cb 20:1138
Muhr M, Prüfer N, Paulat M, Teichmann T (2016) Knockdown of strigolactone biosynthesis genes in Populus affects BRANCHED1 expression and shoot architecture. New Phytol 212:613–626
Muller D, Leyser O (2011) Auxin, cytokinin and the control of shoot branching. Ann Bot 107:1203–1212
Muller D, Waldie T, Miyawaki K, To JP, Melnyk CW, Kieber JJ, Kakimoto T, Leyser O (2015) Cytokinin is required for escape but not release from auxin mediated apical dominance. Plant J 82:874–886
Murfet IC, Reid JB, Casey R, Davies DR (1993) Developmentalmutants. In: Casey R, Davies DR (eds) Peas genetics molecular biology and biotechnology Developmental mutants. Peas genetics molecular biology and biotechnology, (Wallingford: CAB), pp 165–216
Nakamura H, Xue YL, Miyakawa T, Hou F, Qin HM, Fukui K, Shi X, Ito E, Ito S, Park SH (2013) Molecular mechanism of strigolactone perception by DWARF14. Nat Commun 4:2613
Naor A, Flaishman M, Stern R, Moshe A, Erez A (2003) Temperature effects on dormancy completion of vegetative buds in apple. J Am Soc Hortic Sci Am Soc Hortic Sci 128:636–641
Ni J, Gao CC, Chen MS, Pan BZ, Ye KQ, Xu ZF (2015) Gibberellin promotes shoot branching in the perennial woody plant Jatropha curcas. Plant Cell Physiol 56:1655–1666
Nicolas M, Rodríguez-Buey María L, Franco-Zorrilla José M, Cubas P (2015) A recently evolved alternative splice site in the BRANCHED1a gene controls potato plant architecture. Curr Biol 25:1799–1809
Niwa M, Daimon Y, Kurotani K, Higo A, Pruneda-Paz JL, Breton G, Mitsuda N, Kay SA, Ohme-Takagi M, Endo M (2013) BRANCHED1 interacts with FLOWERING LOCUS T to repress the floral transition of the axillary meristems in Arabidopsis. Plant Cell 25:1228–1242
Nunes C, O’Hara LE, Primavesi LF, Delatte TL, Schluepmann H, Somsen GW, Silva AB, Fevereiro PS, Wingler A, Paul MJ (2013) The trehalose 6-phosphate/SnRK1 signaling pathway primes growth recovery following relief of sink limitation. Plant Physiol 162:1720–1732
Otori K, Tamoi M, Tanabe N, Shigeoka S (2017) Enhancements in sucrose biosynthesis capacity affect shoot branching in Arabidopsis. Biosci Biotechnol Biochem 81:1
Patrick JW, Colyvas K (2014) Crop yield components—photoassimilate supply- or utilisation limited-organ development? Funct Plant Biol 41:893
Peng J, Carol P, Richards DE, King KE, Cowling RJ, Murphy GP, Harberd NP (1997) The Arabidopsis GAI gene defines a signaling pathway that negatively regulates gibberellin responses. Genes Dev 11:3194
Rabot A, Henry C, Ben BK, Mortreau E, Azri W, Lothier J, Hamama L, Boummaza R, Leduc N, Pelleschi-Travier S (2012) Insight into the role of sugars in bud burst under light in the rose. Plant Cell Physiol 53:1068
Rameau C, Bertheloot J, Leduc N, Andrieu B, Foucher F, Sakr S (2015) Multiple pathways regulate shoot branching. Front Plant Sci 5:741
Richter R, Behringer C, Muller IK, Schwechheimer C (2010) The GATA-type transcription factors GNC and GNL/CGA1 repress gibberellin signaling downstream from DELLA proteins and phytochrome-interacting factors. Genes Dev 24:2093–2104
Rinne PL, Paul LK, Vahala J, Kangasjarvi J, van der Schoot C (2016) Axillary buds are dwarfed shoots that tightly regulate GA pathway and GA-inducible 1,3-beta-glucanase genes during branching in hybrid aspen. J Exp Bot 67:5975–5991
Roef L, Onckelen HV (2010) Cytokinin Regulation of the Cell Division Cycle. In: Davies PJ (ed) Plant Hormones. Springer, Dordrecht
Rosa M, Hilal M, González JA, Prado FE (2009) Low-temperature effect on enzyme activities involved in sucrose-starch partitioning in salt-stressed and salt-acclimated cotyledons of quinoa (Chenopodium quinoa Willd.) seedlings. Plant Physiol Biochem 47:300–307
Sakai H, Honma T, Aoyama T, Sato S, Kato T, Tabata S, Oka A (2001) ARR1, a transcription factor for genes immediately responsive to cytokinins. Science 294:1519–1521
Shimizu S, Mori H (1998) Analysis of cycles of dormancy and growth in pea axillary buds based on mRNA accumulation patterns of cell cycle-related genes. Plant Cell Physiol 39:255–262
Silverstone AL, Ciampaglio CN, Sun T (1998) The Arabidopsis RGA gene encodes a transcriptional regulator repressing the gibberellin signal transduction pathway. Plant Cell 10:155
Simons JL, Napoli CA, Janssen BJ, Plummer KM, Snowden KC (2007) Analysis of the decreased apical dominance genes of petunia in the control of axillary branching. Plant Physiol 143:697
Sorefan K, Booker J, Haurogné K, Goussot M, Bainbridge K, Foo E, Chatfield S, Ward S, Beveridge C, Rameau C (2003) MAX4 and RMS1 are orthologous dioxygenase-like genes that regulate shoot branching in Arabidopsis and pea. Genes Dev 17:1469
Steiner E, Weiss D (2012) The Arabidopsis O-linked N-acetylglucosamine transferase SPINDLY interacts with class I TCPs to facilitate cytokinin responses in leaves and flowers. Plant Cell 24:96–108
Taniguchi M, Sasaki N, Tsuge T, Aoyama T, Oka A (2007) ARR1 directly activates cytokinin response genes that encode proteins with diverse regulatory functions. Plant Cell Physiol 48:263–277
Thimann KV, Skoog F (1934) On the inhibition of bud development and other functions of growth substance in Vicia faba. In: Proceedings of the Royal Society of London series B-containing papers of a biological character, vol 114, pp 317–339
Volz RK, Gibbs HM, Popenoe J (1994) Branch induction on apple nursery trees: effects of growth regulators and defoliation. New Zealand J Crop Hortic Sci 22:277–283
Willige BC, Isono E, Richter R, Zourelidou M, Schwechheimer C (2011) Gibberellin regulates pin-formed abundance and is required for auxin transport-dependent growth and development in Arabidopsis thaliana. Plant Cell 23:2184–2195
Xu R, Sun P, Jia F, Lu L, Li Y, Zhang S, Huang J (2014) Genomewide analysis of TCP transcription factor gene family in Malus domestica. J Genet 93:733–746
Yanai O, Shani E, Dolezal K, Tarkowski P, Sablowski R, Sandberg G, Samach A, Ori N (2005) Arabidopsis KNOXI Proteins Activate Cytokinin Biosynthesis. Current Biology 15(17):1566–1571
Yadav UP, Ivakov A, Feil R, Duan GY, Walther D, Giavalisco P, Piques M, Carillo P, Hubberten HM, Stitt M (2014) The sucrose-trehalose 6-phosphate (Tre6P) nexus: specificity and mechanisms of sucrose signalling by Tre6P. J Exp Bot 65:1051
Yoneyama K, Xie X, Dai K, Sekimoto H, Sugimoto Y, Takeuchi Y, Yoneyama K (2007) Nitrogen deficiency as well as phosphorus deficiency in sorghum promotes the production and exudation of 5-deoxystrigol, the host recognition signal for arbuscular mycorrhizal fungi and root parasites. Planta 227:125–132
Zawaski C, Busov VB (2014) Roles of gibberellin catabolism and signaling in growth and physiological response to drought and short-day photoperiods in Populus trees. Plos One 9:e86217–e86217
Zeng XF, Zhao DG (2016) Expression of IPT in Asakura-sanshoo (Zanthoxylum piperitum (L.) DC. f. inerme Makino) alters tree architecture, delays leaf senescence, and changes leaf essential oil composition. Plant Mol Biol Report 34:649–658
Zhang S, Zhang D, Fan S, Du L, Shen Y, Xing L, Li Y, Ma J, Han M (2016) Effect of exogenous GA3 and its inhibitor paclobutrazol on floral formation, endogenous hormones, and flowering-associated genes in ‘Fuji’ apple (Malus domestica Borkh.). Plant Physiol Biochem 107:178–186
Funding
This work was supported by the National Apple Industry Technology System of Agriculture Ministry of China (CARS-28); Yangling Subsidiary Center Project of National Apple Improvement Center and Collaborative Innovation of Center Shaanxi Fruit Industry Development (C000088); Chinese postdoctoral project (2015M582713); Innovation project of science and technology plan projects of Shaanxi province (2016TZC-N-11-6).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest. Ming Tan declares that she has no conflict of interest. Guofang Li declares that he has no conflict of interest. Xiaojie Liu declares that he has no conflict of interest. Fang Cheng declares that she has no conflict of interest. Juanjuan Ma declares that she has no conflict of interest. Caiping Zhao declares that she has no conflict of interest. Dong Zhang declares that he has no conflict of interest. Mingyu Han declares that he has no conflict of interest.
Additional information
Communicated by S. Hohmann.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tan, M., Li, G., Liu, X. et al. Exogenous application of GA3 inactively regulates axillary bud outgrowth by influencing of branching-inhibitors and bud-regulating hormones in apple (Malus domestica Borkh.). Mol Genet Genomics 293, 1547–1563 (2018). https://doi.org/10.1007/s00438-018-1481-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-018-1481-y