Abstract
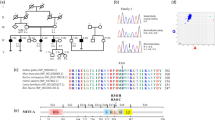
Hearing loss (HL) is a major public health issue. It is clinically and genetically heterogeneous.The identification of the causal mutation is important for early diagnosis, clinical follow-up, and genetic counseling. HL due to mutations in COL11A2, encoding collagen type XI alpha-2, can be non-syndromic autosomal-dominant or autosomal-recessive, and also syndromic as in Otospondylomegaepiphyseal Dysplasia, Stickler syndrome type III, and Weissenbacher–Zweymuller syndrome. However, thus far only one mutation co-segregating with autosomal recessive non-syndromic hearing loss (ARNSHL) in a single family has been reported. In this study, whole exome sequencing of two consanguineous families with ARNSHL from Tunisia and Turkey revealed two novel causative COL11A2 mutations, c.109G > T (p.Ala37Ser) and c.2662C > A (p.Pro888Thr). The variants identified co-segregated with deafness in both families. All homozygous individuals in those families had early onset profound hearing loss across all frequencies without syndromic findings. The variants are predicted to be damaging the protein function. The p.Pro888Thr mutation affects a -Gly-X–Y- triplet repeat motif. The novel p.Ala37Ser is the first missense mutation located in the NC4 domain of the COL11A2 protein. Structural model suggests that this mutation will likely obliterate, or at least partially compromise, the ability of NC4 domain to interact with its cognate ligands. In conclusion, we confirm that COL11A2 mutations cause ARNSHL and broaden the mutation spectrum that may shed new light on genotype–phenotype correlation for the associated phenotypes and clinical follow-up.




Similar content being viewed by others
References
Carson M (1991) Ribbons 2.0. J Appl Crystallogr 24:958–961
Chakchouk I, Ben Said M, Jbeli F et al (2015) NADf chip, a two-color microarray for simultaneous screening of multigene mutations associated with hearing impairment in North African Mediterranean countries. J Mol Diagn. doi:10.1016/j.jmoldx.2014.11.003
Chen W, Kahrizi K, Meyer NC et al (2005) Mutation of COL11A2 causes autosomal recessive non-syndromic hearing loss at the DFNB53 locus. J Med Genet 42:e61
Diaz-Horta O, Duman D, Foster J et al (2012) Whole-exome sequencing efficiently detects rare mutations in autosomal recessive nonsyndromic hearing loss. PLoS ONE 7:e50628
Kimura T, Cheah KS, Chan SD et al (1989) The human α2(XI) collagen (COL11A2) chain. Molecular cloning of cDNA and genomic DNA reveals characteristics of a fibrillar collagen with differences in genomic organization. J Biol Chem 264:13910–13916
Koradi R, Billeter M, Wuthrich K (1996) MOLMOL: a program for display and analysis of macromolecular structures. J Mol Graph 14:51–55
Larkin MA, Blackshields G, Brown NP et al (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948
Leppanen VM, Tossavainen H, Permi P et al (2007) Crystal structure of the N-terminal NC4 domain of collagen IX, a zinc binding member of the laminin–neurexin-sex hormone binding globulin (LNS) domain family. J Biol Chem 282:23219–23230
Lo Conte L, Ailey B, Hubbard TJ et al (2000) SCOP: a structural classification of proteins database. Nucleic Acids Res 28:257–259
Martí-Renom MA, Stuart AC, Fiser A et al (2000) Comparative protein structure modeling of genes and genomes. Annu Rev Biophys Biomol Struct 29:291–325
McGuirt WT, Prasad SD, Griffith AJ et al (1999) Mutations in COL11A2 cause non-syndromic hearing loss (DFNA13). Nat Genet 23:413–419
Morell RJ, Kim HJ, Hood LJ et al (1998) Mutations in the connexin 26 gene (GJB2) among Ashkenazi Jews with nonsyndromic recessive deafness. N Engl J Med 339:1500–1505
Murzin AG, Brenner SE, Hubbard T, Chothia CA (1995) Structural classification of protein database for the investigation of sequences and structures. J Mol Biol 247:536–540
Pihlajamaa T, Lankinen H, Ylostalo J et al (2004) Characterization of recombinant amino-terminal NC4 domain of human collagen IX: interaction with glycosaminoglycans and cartilage oligomeric matrix protein. J Biol Chem 279:24265–24273
Tokgöz-Yılmaz S, Sahlı S, Fitoz S et al (2011) Audiological findings in otospondylomegaepiphyseal dysplasia (OSMED) associated with a novel mutation in COL11A2. Int J Pediatr Otorhinolaryngol 75:433–437
Vuoristo MM, Pihlajamaa T, Vandenberg P et al (1996) Complete structure of the human COL11A2 gene: the exon sizes and other features indicate the gene has not evolved with genes for other fibriller collagens. Ann NY Acad Sci 785:343–344
Wu JJ, Eyre DR (1995) Structural analysis of cross-linking domains in cartilage type XI collagen. Insights on polymeric assembly. J Biol Chem 270:18865–18870
Yan D, Tekin M, Blanton SH, Liu XZ (2013) Next-generation sequencing in genetic hearing loss. Genet Test Mol Bioma 17:581–587
Zhidkova NI, Justice SK, Mayne R (1995) Alternative mRNA processing occurs in the variable region of the pro-alpha 1(XI) and pro-alpha 2(XI) collagen chains. J Biol Chem 270:9486–9493
Acknowledgments
We gratefully thank all the subjects in this study for their collaboration. This study was supported by the National Institutes of Health grants R01DC005575, R01DC012115 to XZL, R01DC009645 to MT, R01GM083897 to AF and the Sylvester Comprehensive Cancer Center to AF. This work was also supported by funds from the ICGEB (International Centre for Genetic Engineering and Biotechnology) and the Ministry of Higher Education and Research of Tunisia to SM.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by S. Hohmann.
The authors I. Chakchouk, M. Grati, and G. Bademci have contributed equally.
Rights and permissions
About this article
Cite this article
Chakchouk, I., Grati, M., Bademci, G. et al. Novel mutations confirm that COL11A2 is responsible for autosomal recessive non-syndromic hearing loss DFNB53. Mol Genet Genomics 290, 1327–1334 (2015). https://doi.org/10.1007/s00438-015-0995-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-015-0995-9