Abstract
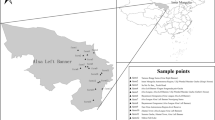
Cryptosporidium is an opportunistic protozoan parasite that can inhabit in the gastrointestinal tract of various hosts. Cryptosporidium infection in black-boned goats and black-boned sheep may pose a threat to the survival and productivity, causing considerable economic losses to the livestock industry. However, it is yet to know whether black-boned goats and black-boned sheep in China are infected with Cryptosporidium. Thus, the objective of the present study was to investigate the prevalence and associated risk factors of Cryptosporidium infection in black-boned goats and black-boned sheep in Yunnan province, China. A total of 590 fecal samples were obtained from black-boned goats and black-boned sheep from five counties in Yunnan province, and the prevalence and species distribution of Cryptosporidium were determined by amplification of the 18S rDNA fragment using the nested PCR. The overall Cryptosporidium prevalence was 13.2% (78/590), with 18.0% (55/305) in black-boned goats and 8.1% (23/285) in black-boned sheep. The age and sampling site were identified as main factors that result in significant differences in Cryptosporidium prevalence. Three species, namely C. muris, C. xiaoi, and C. ubiquitum, were identified in black-boned goats and black-boned sheep in the present study, with C. muris (46/78) as the predominant species. This is the first report of Cryptosporidium infection in black-boned goats and black-boned sheep in China, and the findings will facilitate better understanding, prevention, and control of Cryptosporidium infection in black-boned goats and black-boned sheep in China.
Similar content being viewed by others
References
Adamu H, Petros B, Zhang G, Kassa H, Amer S, Ye J, Feng Y, Xiao L (2014) Distribution and clinical manifestations of Cryptosporidium species and subtypes in HIV/AIDS patients in Ethiopia. PLoS Negl Trop Dis 8:e2831
Ajjampur SS, Sankaran P, Kang G (2008) Cryptosporidium species in HIV-infected individuals in India: an overview. Natl Med J India 21:178–184
Baroudi D, Hakem A, Adamu H, Amer S, Khelef D, Adjou K, Dahmani H, Chen X, Roellig D, Feng Y, Xiao L (2018) Zoonotic Cryptosporidium species and subtypes in lambs and goat kids in Algeria. Parasit Vectors 11:582
Borowski H, Thompson RC, Armstrong T, Clode PL (2010) Morphological characterization of Cryptosporidium parvum life-cycle stages in an in vitro model system. Parasitology 137:13–26
Bouzid M, Hunter PR, Chalmers RM, Tyler KM (2013) Cryptosporidium pathogenicity and virulence. Clin Microbiol Rev 26:115–134
Cama VA, Bern C, Roberts J, Cabrera L, Sterling CR, Ortega Y, Gilman RH, Xiao L (2008) Cryptosporidium species and subtypes and clinical manifestations in children, Peru. Emerg Infect Dis 14:1567–1574
Chen XM, Keithly JS, Paya CV, La Russo NF (2002) Cryptosporidiosis. N Engl J Med 346:1723–1731
Deng WD, Yang SL, Huo YQ, Gou X, Shi XW, Mao HM (2006) Physiological and genetic characteristics of black-boned sheep (Ovis aries). Anim Genet 37:586–588
Diaz P, Navarro E, Prieto A, Perez-Creo A, Vina M, Diaz-Cao JM, Lopez CM, Panadero R, Fernandez G, Diez-Banos P, Morrondo P (2018) Cryptosporidium species in post-weaned and adult sheep and goats from N.W. Spain: public and animal health significance. Vet Parasitol 254:1–5
Duhain GL, Minnaar A, Buys EM (2012) Effect of chlorine, blanching, freezing, and microwave heating on Cryptosporidium parvum viability inoculated on green peppers. J Food Prot 75:936–941
Fayer R (2004) Cryptosporidium: a water-borne zoonotic parasite. Vet Parasitol 126:37–56
Fayer R, Morgan U, Upton SJ (2000) Epidemiology of Cryptosporidium: transmission, detection and identification. Int J Parasitol 30:1305–1322
Feng Y, Xiao L (2017) Molecular epidemiology of cryptosporidiosis in China. Front Microbiol 8:1701
Feng Y, Ryan UM, Xiao L (2018) Genetic diversity and population structure of Cryptosporidium. Trends Parasitol 34:997–1011
Firoozi Z, Sazmand A, Zahedi A, Astani A, Fattahi-Bafghi A, Kiani-Salmi N, Ebrahimi B, Dehghani-Tafti A, Ryan U, Akrami-Mohajeri F (2019) Prevalence and genotyping identification of Cryptosporidium in adult ruminants in central Iran. Parasit Vectors 12:510
Gatei W, Suputtamongkol Y, Waywa D, Ashford RW, Bailey JW, Greensill J, Beeching NJ, Hart CA (2002) Zoonotic species of Cryptosporidium are as prevalent as the anthroponotic in HIV-infected patients in Thailand. Ann Trop Med Parasitol 96:797–802
Han YG, Liu GQ, Jiang XP, Liang GM, He CB, Wang DW, Wu Y, Xiang XL, Hu J, Peng YQ (2013) Investigation of individual heterozygosity correlated to growth traits in Tongshan black-boned goat. Mol Biol Rep 40:6075–6079
Higuera A, Villamizar X, Herrera G, Giraldo JC, Vasquez-A LR, Urbano P, Villalobos O, Tovar C, Ramirez JD (2020) Molecular detection and genotyping of intestinal protozoa from different biogeographical regions of Colombia. Peer J 8:e8554
Hijjawi NS, Meloni BP, Morgan UM, Thompson RC (2001) Complete development and long-term maintenance of Cryptosporidium parvum human and cattle genotypes in cell culture. Int J Parasitol 31:1048–1055
Kaupke A, Michalski MM, Rzezutka A (2017) Diversity of Cryptosporidium species occurring in sheep and goat breeds reared in Poland. Parasitol Res 116:871–879
Khan A, Shaik JS, Grigg ME (2018) Genomics and molecular epidemiology of Cryptosporidium species. Acta Trop 184:1–14
Koinari M, Lymbery AJ, Ryan UM (2014) Cryptosporidium species in sheep and goats from Papua New Guinea. Exp Parasitol 141:134–137
Li P, Cai J, Cai M, Wu W, Li C, Lei M, Xu H, Feng L, Ma J, Feng Y, Xiao L (2016) Distribution of Cryptosporidium species in Tibetan sheep and yaks in Qinghai, China. Vet Parasitol 215:58–62
Mi R, Wang X, Huang Y, Mu G, Zhang Y, Jia H, Zhang X, Yang H, Wang X, Han X, Chen Z (2018) Sheep as a potential source of zoonotic cryptosporidiosis in China. Appl Environ Microbiol 84:e00868–e008618
Mirhashemi ME, Zintl A, Grant T, Lucy F, Mulcahy G, De Waal T (2016) Molecular epidemiology of Cryptosporidium species in livestock in Ireland. Vet Parasitol 216:18–22
Papanikolopoulou V, Baroudi D, Guo Y, Wang Y, Papadopoulos E, Lafi SQ, Abd El-Tawab MM, Diakou A, Giadinis ND, Feng Y, Xiao L (2018) Genotypes and subtypes of Cryptosporidium spp. in diarrheic lambs and goat kids in northern Greece. Parasitol Int 67:472–475
Peng XQ, Tian GR, Ren GJ, Yu ZQ, Lok JB, Zhang LX, Wang XT, Song JK, Zhao GH (2016) Infection rate of Giardia duodenalis, Cryptosporidium spp. and Enterocytozoon bieneusi in cashmere, dairy and meat goats in China. Infect Genet Evol 41:26–31
Qi M, Zhang Z, Zhao A, Jing B, Guan G, Luo J, Zhang L (2019) Distribution and molecular characterization of Cryptosporidium spp., Giardia duodenalis, and Enterocytozoon bieneusi amongst grazing adult sheep in Xinjiang, China. Parasitol Int 71:80–86
Robertson LJ (2009) Giardia and Cryptosporidium infections in sheep and goats: a review of the potential for transmission to human via environmental contamination. Epidemiol Infect 137:913–921
Ryan U, Hijjawi N (2015) New developments in Cryptosporidium research. Int J Parasitol 45:367–373
Ryan U, Xiao L, Read C, Zhou L, Lal AA, Pavlasek I (2003) Identification of novel Cryptosporidium genotypes from the Czech Republic. Appl Environ Microbiol 69:4302–4307
Ryan U, Fayer R, Xiao L (2014) Cryptosporidium species in humans and animals: current understanding and research needs. Parasitology 141:1667–1685
Sahraoui L, Thomas M, Chevillot A, Mammeri M, Polack B, Vallee I, Follet J, Ain-Baaziz H, Adjou KT (2019) Molecular characterization of zoonotic Cryptosporidium spp. and Giardia duodenalis pathogens in Algerian sheep. Vet Parasitol Reg Stud Rep 16:100280
Thompson RC, Olson ME, Zhu G, Enomoto S, Abrahamsen MS, Hijjawi NS (2005) Cryptosporidium and cryptosporidiosis. Adv Parasitol 59:77–158
Uga S, Matsuo J, Kono E, Kimura K, Inoue M, Rai SK, Ono K (2000) Prevalence of Cryptosporidium parvum infection and pattern of oocyst shedding in calves in Japan. Vet Parasitol 94:27–32
Wagenaar JA, Newell DG, Kalupahana RS, Mughini-Gras L (2015) Zoonoses - infections affecting humans and animals. Springer, Netherlands
Widmer G, Sullivan S (2012) Genomics and population biology of Cryptosporidium species. Parasite Immunol 34:61–71
Wu Y, Chang Y, Chen Y, Zhang X, Li D, Zheng S, Wang L, Li J, Ning C, Zhang L (2018) Occurrence and molecular characterization of Cryptosporidium spp., Giardia duodenalis, and Enterocytozoon bieneusi from Tibetan sheep in Gansu, China. Infect Genet Evol 64:46–51
Zhong Z, Tu R, Ou H, Yan G, Dan J, Xiao Q, Wang Y, Cao S, Shen L, Deng J, Zuo Z, Ma X, Zhou Z, Liu H, Yu S, Ren Z, Hu Y, Peng G (2018) Occurrence and genetic characterization of Giardia duodenalis and Cryptosporidium spp. from adult goats in Sichuan Province, China. PLoS One 13:e0199
Funding
Project support was provided, in part, by the Agricultural Science and Technology Innovation Program (ASTIP) (Grant No. CAAS-ASTIP-2016-LVRI-03) and the Promotion Project of ESI Disciplines of Yunnan Agricultural University (Grant No. 2019YNAUESIMS03).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
The present study has been reviewed and approved by the Animal Administration and Ethics Committee of Lanzhou Veterinary Research Institute, Chinese Academy of Agricultural Sciences. Fecal samples were collected from the black-boned sheep and black-boned goats with the permission of the farm owners or managers, and all procedures were performed according to the requirements of Animal Ethics Procedures and Guidelines of the People’s Republic of China.
Additional information
Handling Editor: Una Ryan
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, ZW., Chen, D., Zou, Y. et al. First report of Cryptosporidium spp. infection and risk factors in black-boned goats and black-boned sheep in China. Parasitol Res 119, 2813–2819 (2020). https://doi.org/10.1007/s00436-020-06781-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-020-06781-6