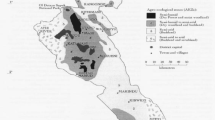
Abstract
This study investigated the prevalence, production losses, spatial clustering, and predictive risk mapping in different climate zones in five states of Mexico. The bulk tank milk samples obtained between January and April 2015 were analyzed for antibodies against Ostertagia ostertagi using the Svanovir ELISA. A total of 1204 farm owners or managers answered the questionnaire. The overall herd prevalence and mean optical density ratio (ODR) of parasite were 61.96% and 0.55, respectively. Overall, the production loss was approximately 0.542 kg of milk per parasited cow per day (mean ODR = 0.92, 142 farms, 11.79%). The spatial disease cluster analysis using SatScan software indicated that two high-risk clusters were observed. In the multivariable analysis, three models were tested for potential association with the ELISA results supported by climatic, environmental, and management factors. The final logistic regression model based on both climatic/environmental and management variables included the factors rainfall, elevation, land surface temperature (LST) day, and parasite control program that were significantly associated with an increased risk of infection. Geostatistical kriging was applied to generate a risk map for the presence of parasite in dairy cattle herds in Mexico. The results indicate that climatic and meteorological factors had a higher potential impact on the spatial distribution of O. ostertagi than the management factors.



Similar content being viewed by others
References
Beck MA, Colwell DD, Goater CP, Kienzle SW (2015) Where’s the risk? Landscape epidemiology of gastrointestinal parasitism in Alberta beef cattle. Parasit Vectors 8(1):434
Bennema S, Vercruysse J, Claerebout E, Schnieder T, Strube C, Ducheyne E, Hendrickx G, Charlier J (2009) The use of bulk-tank milk ELISAs to assess the spatial distribution of Fasciola hepatica, Ostertagia ostertagi and Dictyocaulus viviparus in dairy cattle in Flanders (Belgium). Vet Parasitol 165(1–2):51–57. https://doi.org/10.1016/j.vetpar.2009.07.006
Bennema SC, Vercruysse J, Morgan E, Stafford K, Hoglund J, Demeler J, von Samson-Himmelstjerna G, Charlier J (2010) Epidemiology and risk factors for exposure to gastrointestinal nematodes in dairy herds in northwestern Europe. Vet Parasitol 173(3–4):247–254. https://doi.org/10.1016/j.vetpar.2010.07.002
Charlier J, Claerebout E, Duchateau L, Vercruysse J (2005) A survey to determine relationships between bulk tank milk antibodies against Ostertagia ostertagi and milk production parameters. Vet Parasitol 129(1–2):67–75. https://doi.org/10.1016/j.vetpar.2004.11.024
Charlier J, Duchateau L, Claerebout E, Vercruysse J (2007) Predicting milk-production responses after an autumn treatment of pastured dairy herds with eprinomectin. Vet Parasitol 143(3–4):322–328. https://doi.org/10.1016/j.vetpar.2006.08.015
Charlier J, Ghebretinsae AH, Levecke B, Ducheyne E, Claerebout E, Vercruysse J (2016) Climate-driven longitudinal trends in pasture-borne helminth infections of dairy cattle. Int J Parasitol 46(13–14):881–888. https://doi.org/10.1016/j.ijpara.2016.09.001
Charlier J, Hoglund J, von Samson-Himmelstjerna G, Dorny P, Vercruysse J (2009) Gastrointestinal nematode infections in adult dairy cattle: impact on production, diagnosis and control. Vet Parasitol 164(1):70–79. https://doi.org/10.1016/j.vetpar.2009.04.012
Charlier J, Vercruysse J, Morgan E, Van Dijk J, Williams D (2014) Recent advances in the diagnosis, impact on production and prediction of Fasciola hepatica in cattle. Parasitology 141(03):326–335
Fanke J, Charlier J, Steppin T, von Samson-Himmelstjerna G, Vercruysse J, Demeler J (2017) Economic assessment of Ostertagia ostertagi and Fasciola hepatica infections in dairy cattle herds in Germany using Paracalc(R). Vet Parasitol 240:39–48. https://doi.org/10.1016/j.vetpar.2017.03.018
Forbes AB, Vercruysse J, Charlier J (2008) A survey of the exposure to Ostertagia ostertagi in dairy cow herds in Europe through the measurement of antibodies in milk samples from the bulk tank. Vet Parasitol 157(1–2):100–107. https://doi.org/10.1016/j.vetpar.2008.06.023
Guitian FJ, Dohoo IR, Markham RJ, Conboy G, Keefe GP (2000) Relationships between bulk-tank antibodies to Ostertagia ostertagi and herd-management practices and measures of milk production in Nova Scotia dairy herds. Prev Vet Med 47(1–2):79–89
Hoglund J, Dahlstrom F, Engstrom A, Hessle A, Jakubek EB, Schnieder T, Strube C, Sollenberg S (2010) Antibodies to major pasture borne helminth infections in bulk-tank milk samples from organic and nearby conventional dairy herds in south-central Sweden. Vet Parasitol 171(3–4):293–299. https://doi.org/10.1016/j.vetpar.2010.04.002
Kantzoura V, Kouam MK, Demiris N, Feidas H, Theodoropoulos G (2011) Risk factors and geospatial modelling for the presence of Fasciola hepatica infection in sheep and goat farms in the Greek temperate Mediterranean environment. Parasitology 138(7):926–938. https://doi.org/10.1017/S0031182011000436
Kulldorff M, Feuer EJ, Miller BA, Freedman LS (1997) Breast cancer clusters in the northeast United States: a geographic analysis. Am J Epidemiol 146(2):161–170
Kulldorff M, Nagarwalla N (1995) Spatial disease clusters: detection and inference. Stat Med 14(8):799–810
Pablos-Tanarro A, Perez-Cabal MA, Ortega-Mora LM, Ferre I (2013) Presence of Ostertagia ostertagi antibodies in bulk tank milk from cattle herds in northern Spain. Vet Parasitol 197(1–2):388–392. https://doi.org/10.1016/j.vetpar.2013.05.021
Pachauri RK, et al. (2014) Climate change 2014: synthesis report. Contribution of Working Groups I, II and III to the fifth assessment report of the Intergovernmental Panel on Climate Change. IPCC
Rose H, Wang T, van Dijk J, Morgan ER (2015) GLOWORM-FL: a simulation model of the effects of climate and climate change on the free-living stages of gastro-intestinal nematode parasites of ruminants. Ecol Model 297:232–245. https://doi.org/10.1016/j.ecolmodel.2014.11.033
Rodríguez-Vivas RI, Grisi L, Pérez de León AA, Silva VH, Torres-Acosta JFJ, Fragoso SH, Romero SD, Rosario CR, Saldierna F, García CD (2017) Potential economic impact assessment for cattle parasites in Mexico. Rev Mex Cienc Pecu 8(1):61–74
Sanchez J, Dohoo I (2002) A bulk tank milk survey of Ostertagia ostertagi antibodies in dairy herds in Prince Edward Island and their relationship with herd management factors and milk yield. Can Vet Journal = La revue veterinaire canadienne 43(6):454–459
Sanchez J, Markham F, Dohoo I, Sheppard J, Keefe G, Leslie K (2004) Milk antibodies against Ostertagia ostertagi: relationships with milk IgG and production parameters in lactating dairy cattle. Vet Parasitol 120(4):319–330. https://doi.org/10.1016/j.vetpar.2004.01.010
Sandholt I, Rasmussen K, Andersen J (2002) A simple interpretation of the surface temperature/vegetation index space for assessment of surface moisture status. Rem Sens Environ 79(2):213–224. https://doi.org/10.1016/S0034-4257(01)00274-7
Sekiya M, Zintl A, Doherty ML (2013) Bulk milk ELISA and the diagnosis of parasite infections in dairy herds: a review. Ir Vet J 66(1):14. https://doi.org/10.1186/2046-0481-66-14
Van Dijk J, Sargison N, Kenyon F, Skuce P (2010) Climate change and infectious disease: helminthological challenges to farmed ruminants in temperate regions. Animal 4(03):377–392
Vanderstichel R, Dohoo I, Sanchez J, Conboy G (2012) Effects of farm management practices and environmental factors on bulk tank milk antibodies against gastrointestinal nematodes in dairy farms across Canada. Prev Vet Med 104(1–2):53–64. https://doi.org/10.1016/j.prevetmed.2011.09.022
Verschave SH, Charlier J, Rose H, Claerebout E, Morgan ER (2016) Cattle and nematodes under global change: transmission models as an ally. Trends Parasitol 32(9):724–738. https://doi.org/10.1016/j.pt.2016.04.018
Acknowledgements
The authors are grateful to Jesús Valle Juárez for providing excellent technical assistance.
Funding
This study was supported by Benemérita Universidad Autónoma de Puebla (VIEP-VIMA-NAT-17-I).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: AV and CP. Performed the experiments: AV, CP, AC, and AR. Analyzed the data: AV, AC, and AR. Contributed reagents/materials/analysis tools: AV, JO, and AR. Wrote the paper: AV, CP, and JO. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Electronic supplementary material
ESM 1
(PDF 12 kb)
Rights and permissions
About this article
Cite this article
Villa-Mancera, A., Pastelín-Rojas, C., Olivares-Pérez, J. et al. Bulk tank milk prevalence and production losses, spatial analysis, and predictive risk mapping of Ostertagia ostertagi infections in Mexican cattle herds. Parasitol Res 117, 1613–1620 (2018). https://doi.org/10.1007/s00436-018-5845-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-018-5845-3