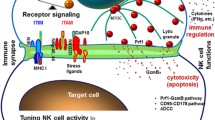
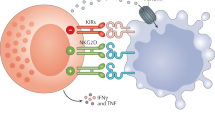
Abstract
Purpose
Natural killer (NK) cells can kill transformed cells and represent anti-tumor activities for improving the immunotherapy of cancer. In previous works, we established human interleukin-15 (hIL-15) gene-modified NKL cells (NKL-IL15) and demonstrated their efficiency against human hepatocarcinoma cells (HCCs) in vitro and in vivo. To further assess the applicability of NKL-IL15 cells in adoptive cellular immunotherapy for human leukemia, here we report their natural cytotoxicity against leukemia in vitro and in vivo.
Methods
Flow cytometry, ELISA and MTT methods were performed for molecular expression, cell proliferation and cytotoxicity assays. Leukemia xenograft NOD/SCID mice were established by subcutaneous injection with K562 cells, and then treated with irradiated NKL cells.
Results
We found NKL-IL15 cells displayed a significant high cytolysis activity against both human leukemia cell lines and primary leukemia cells from patients, accompanied with up-regulated expression of molecules related to NK cell cytotoxicity such as perforin, granzyme B and NKp80. Moreover, cytokines secreted by NKL-IL15 cells, including TNF-α and IFN-γ, could induce the expression of NKG2D ligands on target cells, which increased the susceptibility of leukemia cells to NK cell-mediated cytolysis. Encouragingly, NKL-IL15 cells significantly inhibited the growth of leukemia cells in xenografted NOD/SCID mice and prolonged the survival of tumor-bearing mice dramatically. Furthermore, NKL-IL15 cells displayed stimulatory effects on hPBMCs, indicating the immunesuppressive status of leukemia patients could be improved by NKL-IL15 cell treatment.
Conclusions
These results provided evidence that IL-15 gene-modification could augment NK cell-mediated anti-human leukemia function, which would improve primary NK cell-based immunotherapy for leukemia in future.






Similar content being viewed by others
References
Bray SM, Vujanovic L, Butterfield LH (2011) Dendritic cell-based vaccines positively impact natural killer and regulatory T cells in hepatocellular carcinoma patients. Clin Dev Immunol 2011:249–281. https://doi.org/10.1155/2011/249281
Cheng M, Chen Y, Xiao W et al (2013) NK cell-based immunotherapy for malignant diseases. Cell Mol Immunol 10:230–252 https://doi.org/10.1038/cmi.2013.10.
Crucitti L, Crocchiolo R, Toffalori C et al (2015) Incidence, risk factors and clinical outcome of leukemia relapses with loss of the mismatched HLA after partially incompatible hematopoietic stem cell transplantation. Leukemia 29:1143–1152. https://doi.org/10.1038/leu.2014.314
de Thé H, Pandolfi PP, Chen Z (2017) Acute promyelocytic leukemia: a paradigm for oncoprotein-targeted cure. Cancer Cell 32(5):552–560. https://doi.org/10.1016/j.ccell.2017.10.002
Dubois S, Patel HJ, Zhang M et al (2008) Preassociation of IL-15 with IL-15R alpha-IgG1-Fc enhances its activity on proliferation of NK and CD8+/CD44high T cells and its antitumor action. J Immunol 180(4):2099–2106. https://doi.org/10.4049/jimmunol.180.4.2099
He YG, Mayhew E, Mellon J et al (2004) Expression and possible function of IL-2 and IL-15 receptors on human uveal melanoma cells. Investig Ophthalmol Vis Sci 45:4240–4246. https://doi.org/10.1167/iovs.04-0599
Jiang W, Zhang J, Tian Z (2008) Functional characterization of interleukin-15 gene transduction into the human natural killer cell line NKL. Cytotherapy 10:265–274. https://doi.org/10.1080/14653240801965156
Jiang W, Zhang C, Tian Z et al (2014) hIL-15 gene-modified human natural killer cells (NKL-IL15) augments the anti-human hepatocellular carcinoma effect in vivo. Immunobiology 219:547–553 https://doi.org/10.1016/j.imbio.2014.03.007
Joyce S (2001) CD1d and natural T cells: how their properties jump-start the immune system. Cell Mol Life Sci 58:442–469. https://doi.org/10.1007/PL00000869
Kennedy MK, Glaccum M, Brown SN et al (2000) Reversible defects in natural killer and memory CD8 T cell lineages in IL-15 deficient mice. J Exp Med 191:771–780. https://doi.org/10.1084/jem.191.5.771
Lion E, Willemen Y, Berneman ZN et al (2012) Natural killer cell immune escape in acute myeloid leukemia. Leukemia 26:2019–2026. https://doi.org/10.1038/leu.2012.87
Ljunggren HG, Malmberg KJ (2007) Prospects for the use of NK cells in immunotherapy of human cancer. Nat Rev Immunol 7:329–339. https://doi.org/10.1038/nri2073
Miller JS, Soignier Y, Panoskaltsis-Mortari A (2005) Successful adoptive transfer and in vivo expansion of human haploidentical NK cells in cancer patients. Blood 105:3051–3057. https://doi.org/10.1182/blood-2004-07-2974
Nagashima S, Mailliard R, Kashii Y et al (1998) Stable transduction of the interleukin-2 gene into human natural killer cell lines and their phenotypic and functional characterization in vitro and in vivo. Blood 91:3850–3861
Ohteki T (2002) Critical Role for IL-15 in innate immunity. Curr Mol Med 2:371–380. https://doi.org/10.2174/1566524023362519
Pende D, Spaggiari GM, Marcenaro S et al (2005) Analysis of the receptor-ligand interactions in the natural killer-mediated lysis of freshly isolated myeloid or lymphoblastic leukemias: evidence for the involvement of the Poliovirus receptor (CD155) and Nectin-2 (CD112). Blood 105:2066–2073. https://doi.org/10.1182/blood-2004-09-3548
Pierson BA, Miller JS (1996) CD56+ bright and CD56+ dim natural killer cells in patients with chronic myelogenous leukemia progressively decrease in number, respond less to stimuli that recruit clonogenic natural killer cells, and exhibit decreased proliferation on a per cell basis. Blood 88:2279–2287
Robertson MJ, Cochran KJ, Cameron C et al (1996) Characterization of a cell line, NKL, derived from an aggressive human natural killer cell leukemia. Exp Hematol 24:406–415
Romanski A, Bug G, Becker S et al (2005) Mechanisms of resistance to natural killer cell-mediated cytotoxicity in acute lymphoblastic leukemia. Exp Hematol 33:344–352. https://doi.org/10.1016/j.exphem.2004.11.006
Romee R, Leong JW, Fehniger TA (2014) Utilizing cytokines to function-enable human NK cells for the immunotherapy of cancer. Scientifica (Cairo) 2014:205796. https://doi.org/10.1155/2014/205796
Ruggeri L, Capanni M, Urbani E et al (2002) Effectiveness of donor natural killer cell alloreactivity in mismatched hematopoietic transplants. Science 295:2097–2100. https://doi.org/10.1126/science.1068440
Stanietsky N, Mandelboim O et al (2010) Paired NK cell receptors controlling NK cytotoxicity. FEBS Lett 584:4895–4900 https://doi.org/10.1016/j.febslet.2010.08.047
Sutlu T, Alici E (2009) Natural killer cell-based immunotherapy in cancer: current insights and future prospects. J Intern Med 26:154–181 https://doi.org/10.1111/j.1365-2796.2009.02121.x
Terme M, Ullrich E, Delahaye NF et al (2008) Natural killer cell-directed therapies: moving from unexpected results to successful strategies. Nat Immunol 9:486–494. https://doi.org/10.1038/ni1580
Willard VW, Klosky JL, Li C et al (2017) The impact of childhood cancer: perceptions of adult survivors. Cancer 123(9):1625–1634. https://doi.org/10.1002/cncr.30514
Wodnar-Filipowicz A, Kalberer CP (2006) Function of natural killer cells in immune defence against human leukaemia. Swiss Med Wkly 136:359–364
Xu D, Han Q, Hou Z et al (2016) miR-146a negatively regulates NK cell functions via STAT1 signaling. Cell Mol Immunol 14(8):712–720. https://doi.org/10.1038/cmi.2015.113
Yang Y, Han Q, Hou Z et al (2017) Exosomes mediate hepatitis B virus (HBV) transmission and NK-cell dysfunction. Cell Mol Immunol 14(5):465–475. https://doi.org/10.1038/cmi.2016.24
Zhang J, Sun R, Wei H et al (2004a) Characterization of interleukin-15 gene-modified human natural killer cells: implications for adoptive cellular immunotherapy. Haematologica 89:338–347
Zhang J, Sun R, Wei H et al (2004b) Characterization of stem cell factor gene-modified human natural killer cell line, NK-92 cells: implication in NK cell-based adoptive cellular immunotherapy. Oncol Rep 11:1097–1106. https://doi.org/10.3892/or.11.5.1097
Funding
This work was supported by Grants from National Basic Research Program of China (No. 2013CB531503), National Natural Science Foundation of China (Nos. 81172789, 30972692).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Medical Ethics Committee of Shandong University and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was approved by the Institutional Review Board of Shandong University.
Informed consent
Informed consent was obtained from all individual participants and the parents of minor participant included in the study.
Rights and permissions
About this article
Cite this article
Jiang, W., Zhang, C., Tian, Z. et al. hIL-15-gene modified human natural killer cells (NKL-IL15) exhibit anti-human leukemia functions. J Cancer Res Clin Oncol 144, 1279–1288 (2018). https://doi.org/10.1007/s00432-018-2654-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-018-2654-0