Abstract
Purpose
Glioblastomas represent the most common primary malignant tumor of the nervous system and the most frequent type of astrocytic tumors. Despite improved therapeutic options, prognosis has remained exceptionally poor over the last two decades. Therefore, new treatment approaches are urgently needed. An overexpression of somatostatin (SST) as well as chemokine CXCR4 and endothelin A (ETA) receptors has been shown for many types of cancer. Respective expression data for astrocytic brain tumors, however, are scarce and contradictory.
Methods
SST subtype, CXCR4 and ETA expression was comparatively evaluated in a total of 57 grade I–IV astrocytic tumor samples by immunohistochemistry using well-characterized monoclonal antibodies.
Results
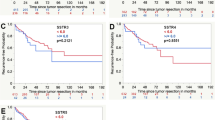
Overall, receptor expression on the tumor cells was only very low. SST5 was the most prominently expressed receptor, followed by SST3, ETA, SST2 and CXCR4. In contrast, tumor capillaries displayed strong SST2, SST3, SST5, CXCR4 and ETA expression. Presence of SST5, CXCR4 and ETA on tumor cells and of SST3, CXCR4 and ETA on microvessels gradually increased from grade II to grade IV tumors. Ki-67 values correlated significantly with CXCR4 expression on tumor cells and with vascular SST3, CXCR4 or ETA positivity. SST5 or CXCR4 positivity of tumor cells and vascular SST3 or CXCR4 expression negatively correlated with patient outcome.
Conclusions
Though having some prognostic value, SST, CXCR4 or ETA expression on astrocytic tumor cells is clearly of no therapeutic relevance. Indirect targeting of these highly vascularized tumors via SST3, SST5, CXCR4 or ETA on the microvessels, in contrast, may represent a promising additional therapeutic strategy.




Similar content being viewed by others
Abbreviations
- CXCR4:
-
C–X–C motif chemokine receptor 4
- ETA:
-
Endothelin receptor A
- SST:
-
Somatostatin receptor
References
Ambroise MM, Khosla C, Ghosh M, Mallikarjuna VS, Annapurneswari S (2011) Practical value of MIB-1 index in predicting behavior of astrocytomas. Indian J Pathol Microbiol 54:520–525
Bianco AM, Uno M, Oba-Shinjo SM, Clara CA, de Almeida Galatro TF, Rosemberg S, Teixeira MJ, Nagahashi Marie SK (2015) CXCR7 and CXCR4 expressions in infiltrative astrocytomas and their interactions with HIF1a expression and IDH1 mutation. Pathol Oncol Res 21:229–240
Buck AK, Stolzenburg A, Hänscheid H, Schirbel A, Lückerath K, Schottelius M, Wester HJ, Lapa C (2017) Chemokine receptor—directed imaging and therapy. Methods 130:63–71
Caplin ME, Pavel M, Ćwikła JB, Phan AT, Raderer M, Sedláčková E, Cadiot G, Wolin EM, Capdevila J, Wall L, Rindi G, Langley A, Martinez S, Blumberg J, Ruszniewski P, for the CLARINETInvestigators (2014) Lanreotide in metastatic enteropancreatic neuroendocrine tumors. New Engl J Med 371:224–233
Cervera P, Videau C, Viollet C, Petrucci C, Lacombe J, Winsky-Sommerer R, Csaba Z, Helboe L, Daumas-Duport C, Reubi JC, Epelbaum J (2002) Comparison of somatostatin receptor expression in human gliomas and medulloblastomas. J Neuroendocrinol 14:458–471
Chen WJ, He DS, Tang RX, Ren FH, Chen G (2015) Ki-67 is a valuable prognostic factor in gliomas: evidence from a systematic review and meta-analysis. Asian Pac J Cancer Prev 16:411–420
Chinezu L, Vasiljevic A, Jouanneau E, François P, Borda A, Trouillas J, Raverot G (2014) Expression of somatostatin receptors, SSTR2A and SSTR5, in 108 endocrine pituitary tumors using immunohistochemical detection with new specific monoclonal antibodies. Hum Pathol 45:71–77
Cloughesy TF, Cavenee WK, Mischel PS (2014) Glioblastoma: from molecular pathology to targeted treatment. Annu Rev Pathol Mech Dis 9:1–25
Cohen A, Holmen S, Colman H (2013) IDH1 and IDH2 mutations in gliomas. Curr Neurol Neurosci Rep 13:345
Domanska UM, Kruizinga RC, Nagengast WB, Timmer-Bosscha H, Huls G, de Vries EGE, Walenkamp AME (2013) A review on CXCR4/CXCL12 axis in oncology: no place to hide. Eur J Cancer 49:219–230
Dutour A, Kumar U, Panetta R, Ouafik L, Fina F, Sasi R, Patel YC (1998) Expression of somatostatin receptor subtypes in human brain tumors. Int J Cancer 76:620–627
Egidy G, Eberl LP, Valdenaire O, Irmler M, Majdi R, Diserens AC, Fontana A, Janzer RC, Pinet F, Juillert-Jeanneret (2000) The endothelin system in human glioblastoma. Lab Invest 80:1681–1689
Feindt J, Becker I, Blömer U, Hugo HH, Mehdorn HM, Krisch B, Mentlein R (1995) Expression of somatostatin receptor subtypes in cultured astrocytes and gliomas. J Neurochem 65:1997–2005
Fischer T, Doll C, Jacobs S, Kolodziej A, Stumm R, Schulz S (2008a) Reassessment of SST2 somatostatin receptor expression in human normal and neoplastic tissues using the novel rabbit monoclonal antibody UMB-1. J Clin Endocrinol Metab 93:4519–4524
Fischer T, Nagel F, Jacobs S, Stumm R, Schulz S (2008b) Reassessment of CXCR4 chemokine receptor expression in human normal and neoplastic tissues using the novel rabbit monoclonal antibody UMB-2. PLoS One 3:e4069
Furusato B, Rhim JS (2010) CXCR4 and cancer. Pathol Internat 7:497–505
Gravina GL, Mancini A, Colapietro A, Vitale F, Vetuschi A, Pompili S, Rossi G, Marampon F, Richardson PJ, Patient L, Patient L, Burbidge S, Festuccia C (2017) The novel CXCR4 antagonist, PRX177561, reduces tumor cell proliferation and accelerates cancer stem cell differentiation in glioblastoma preclinical models. Tumour Biol. https://doi.org/10.1177/1010428317695528
Hattermann K, Held-Feindt J, Lucius R, Müerköster SS, Penfold MET, Schall T, Mentlein R (2010) The chemokine receptor CXCR7 is highly expressed in human glioma cells and mediates antiapoptotic effects. Cancer Res 70:3299–3308
Heute D, Kostron H, von Guggenberg E, Ingorokva S, Gabriel M, Dobrozemsky G, Stockhammer G, Virgolini I (2010) Response of recurrent high-grade glioma to treatment with 90Y-DOTATOC. J Nucl Med 51:397–400
Irani S, Salajegheh A, Smith RA, Lam AKY (2014) A review of the profile of endothelin axis in cancer and its management. Crit Rev Oncol Hematol 89:314–321
Johannessen AL, Thorp SH (2006) The clinical value of Ki-67/MIB-1 labeling index in human astrocytomas. Pathol Onc Res 12:143–147
Kaemmerer D, Träger T, Hoffmeister M, Sipos B, Hommann M, Sänger J, Schulz S, Lupp A (2015a) Inverse expression of somatostatin and CXCR4 chemokine receptors in gastroenteropancreatic neuroendocrine neoplasms of different malignancy. Oncotarget 6:27566–27579
Kaemmerer D, Reimann C, Specht E, Wirtz RM, Sayeg M, Baum RP, Schulz S, Lupp A (2015b) Differential expression and prognostic value of the chemokine receptor CXCR4 in bronchopulmonary neuroendocrine neoplasm. Oncotarget 6:3346–3358
Kim SJ, Lee HJ, Kim MS, Choi HJ, He J, Wu Q, Aldape K, Weinberg JS, Yung WKA, Conrad CA, Langley RR, Lehembre F, Regenass U, Fidler IJ (2015) Macitentan, a dual endothelin receptor antagonist, in combination with temozolomide leads to glioblastoma regression and long-term survival in mice. Clin Cancer Res 21:4630–4641
Kiviniemi A, Gardberg M, Frantzén J, Pesola M, Vuorinen V, Parkkola R, Tolvanen T, Suilamo S, Johansson J, Luoto P, Kemppainen J, Roivainen A, Minn H (2015) Somatostatin receptor subtype 2 in high-grade gliomas: PET/CT with 68Ga-DOTA-peptides, correlation to prognostic markers, and implications for targeted radiotherapy. EJNMMI Res 5:25
Kiviniemi A, Gardberg M, Kivinen K, Posti JP, Vuorinen V, Sipilä J, Rahi M, Sankinen M, Minn H (2017) Somatostatin receptor 2A in gliomas: association with oligodendrogliomas and favourable outcome. Oncotarget 8:49123–49132
Lambertini C, Barzaghi-Rinaudo P, D’Amato L, Schulz S, Nuciforo P, Schmid HA (2013) Evaluation of somatostatin receptor subtype expression in human neuroendocrine tumors using two sets of new monoclonal antibodies. Regul Pept 187:35–41
Lamszus K, Meyerhof W, Westphal M (1997) Somatostatin and somatostatin receptors in the diagnosis and treatment of gliomas. J Neuro-Oncol 35:353–364
Lapa C, Linsenmann T, Lückerath K, Samnick S, Herrmann K, Stoffer C, Ernestus RI, Buck AK, Löhr M, Monoranu CM (2015) Tumor-associated macrophages in glioblastoma multiforme—a suitable target for somatostatin receptor-based imaging and therapy? PloS One 10:e0122269
Louis DN, Perry A, ·Reifenberger G,·von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820
Lupp A, Danz M, Müller D (2001) Morphology and cytochrome P450 isoforms expression in precision-cut rat liver slices. Toxicology 161:53–66
Lupp A, Hunder A, Petrich A, Nagel F, Doll C, Schulz S (2011) Reassessment of SST5 somatostatin receptor expression in normal and neoplastic human tissues using the novel rabbit monoclonal antibody UMB-4. Neuroendocrinol 94:255–264
Lupp A, Nagel F, Doll C, Röcken C, Evert M, Mawrin C, Saeger W, Schulz S (2012) Reassessment of sst3 somatostatin receptor expression in normal and neoplastic human tissues using the novel rabbit monoclonal antibody UMB-4. Neuroendocrinol 96:301–310
Lupp A, Nagel F, Schulz S (2013) Reevaluation of sst1 somatostatin receptor expression in human normal and neoplastic tissues using the novel rabbit monoclonal antibody UMB-7. Regul Pept 183:1–6
Lupp A, Mann A, Heeb A, Kaemmerer D, Sänger J, Evert M, Evert K, Mawrin C, Schulz S (2015) Reassessment of endothelin receptor A expression in normal and neoplastic human tissues using the novel rabbit monoclonal antibodyUMB-8. Peptides 66:19–25
Ma YH, Mentlein R, Knerlich F, Kruse ML, Mehdorn HM, Held-Feindt J (2007) Expression of stem cell markers in human astrocytomas of different WHO grades. J Neurooncol. https://doi.org/10.1007/s11060-007-9439-7
Mawrin C, Schulz S, Pauli SU, Treuheit T, Diete S, Dietzmann K, Firsching R, Schulz S, Höllt V (2004) Differential expression of sst1, sst2A, and sst3 somatostatin receptor proteins in low-grade and high-grade astrocytomas. J Neuropathol Exp Neurol 63:13–19
Mercurio L, Ajmone-Cat MA, Cecchetti S, Ricci A, Bozzuto G, Molinari A, Manni I, Pollo B, Scala S, Carpinelli G, Minghetti L (2016) Targeting CXCR4 by a selective peptide antagonist modulates tumor microenvironment and microglia reactivity in a human glioblastoma model. J Exp Clin Cancer Res 35:55
Naidoo V, Naidoo S, Mahabeer R, Raidoo DM (2005) Localization of the endothelin system in human diffuse astrocytomas. Cancer 104:1049–1057
Ostrom QT, Gittleman H, Fulop J, Liu M, Blanda R, Kromer C, Wolinsky Y, Kruchko C, Barnholtz-Sloan JS (2015) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2008–2012. Neuro-Oncology 17:iv1–iv62
Razmkhah M, Arabpour F, Taghipour M, Mehrafshan A, Chenari N, Ghaderi A (2014) Expression of chemokines and chemokine receptors in brain tumor tissue derived cells. Asian Pac J Cancer Prev 15:7201–7205
Remmele W, Stegner HE (1987) Recommendation for uniform definition of an immunoreactive score (IRS) for immunohistochemical estrogen receptor detection (ER-ICA) in breast cancer tissue. Pathologe 8:138–140
Rempel S, Dudas S, Ge S, Gutierrez JA (2000) Identification and localization of the cytokine SDF1 and its receptor, CXC chemokine receptor 4, to regions of necrosis and angiogenesis in human glioblastoma. Clin Cancer Res 6:102–111
Rinke A, Müller HH, Schade-Brittinger C, Klose KJ, Barth P, Wied M, Mayer C, Aminossadati B, Pape UF, Bläker M, Harder J, Arnold C, Gress T, Arnold R; PROMID Study Group (2009) Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol 27:4656–4663
Rosano L, Spinella F, Bagnato A (2013) Endothelin 1 in cancer: biological implications and therapeutic opportunities. Nat Rev 13:637–651
Rubin JB, Kung AL, Klein RS, Chan JA, Sun Y, Schmidt K, Kieran MW, Luster AD, Segal R (2003) A small-molecule antagonist of CXCR4 inhibits intracranial growth of primary brain tumors. PNAS 100:13513–13518
Said N, Theodorescu D (2012) Permissive role of endothelin receptors in tumor metastasis. Life Sci 91:522–527
Savelli G, Muni A (2015) Somatostatin receptors in anaplastic oligodendroglioma relapse evidenced by 68Ga DOTANOC PET/CT. Clin Nucl Med 40:e363-e365
Smit Duijzentkunst DA, Kwekkeboom DJ, Bodei L (2017) Somatostatin receptor 2-targeting compounds. J Nucl Med 58:54S-60S
Stollberg S, Kaemmerer D, Neubauer E, Schulz S, Simonitsch-Klupp I, Kiesewetter B, Raderer B, Lupp A (2016) Differential somatostatin and CXCR4 chemokine receptor expression in MALT-type lymphoma of gastric and extragastric origin. J Cancer Res Clin Oncol 142:2239–2247
Thotakura M, Tirumalasetti N, Krishna R (2014) Role of Ki-67 labeling index as an adjunct to the histopathological diagnosis and grading of astrocytomas. J Can Res Ther 10:641–645
Walenkamp AME, Lapa C, Herrmann K, Wester HJ (2017) CXCR4 ligands. The next big hit? J Nucl Med 58:77S-82S
Wang R, Dashwood RH (2011) Endothelins and their receptors in cancer: identification of therapeutic targets. Pharmacol Res 63:519–524
Woerner BM, Warrington NM, Kung AL, Perry A, Rubin JB (2005) Widespread CXCR4 activation in astrocytomas revealed vy phosphor-CXCR4-specific antibodies. Cancer Res 65:11392–11399
Yan H, Parsons DW, Jin G, McLendon R, Rasheed BA, Yuan W, Kos I, Batinic-Haberle I, Jones S, Riggins GJ, Friemann H, Friedmann A, Reardon D, Herndon J, Kinzler KW, Velculescu VE, Vogelstein B, Bigner DD (2009) IDH1 and IDH2 mutations in gliomas. N Engl J Med 360:765–773
Zhao H, Guo L, Zhao H, Zhao J, Weng H, Zhao B (2015) CXCR4 over-expression and survival in cancer: a system review and meta-analysis. Oncotarget 6:5022–5040
Zhou Y, Larsen PH, Hao C, Yong VW (2002) CXCR4 is a major chemokine receptor on glioma cells and mediates their survival. J Biol Chem 277:4981–4987
Zhou W, Jiang Z, Song X, Liu Y, Wen P, Guo Y, Xu F, Kong L, Zhang P, Han A, Yu J (2008) Promoter hypermethylation-mediated down-regulation of CXCL12 in human astrocytoma. J Neurosci Res 86:3002–3010
Funding
The Theranostic Research Center, Zentralklinik Bad Berka, 99437 Bad Berka, Germany, provided funding for this research.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: Daniel Kaemmerer, Amelie Lupp. Provided the tumor samples: Julianne Behnke-Mursch, Wolfgang Brück. Provided the antibodies: Stefan Schulz. Acquired the clinical data: Daniel Kaemmerer. Performed the experiments: Franziska Lange, Amelie Lupp. Analyzed the data: Franziska Lange, Amelie Lupp. Interpreted the data: Amelie Lupp. Wrote the paper: Amelie Lupp. Revised critically the manuscript: Franziska Lange, Daniel Kaemmerer, Julianne Behnke-Mursch, Wolfgang Brück, Stefan Schulz. Each of the authors has approved the manuscript and acknowledges that he or she participated sufficiently in the work to take public responsibility for its content.
Corresponding author
Ethics declarations
Conflict of interest
Daniel Kaemmerer received funding and support for travelling to meetings by the companies Ipsen and Pfizer. All other authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Permission was gained from the local ethics committee (Ethikkommission der Landesärztekammer Thüringen) for this retrospective analysis. For this type of study formal consent is not required. All data were recorded and analyzed anonymously.
Electronic supplementary material
Below is the link to the electronic supplementary material.
432_2018_2645_MOESM1_ESM.tif
Supplemental Figure S1: Typical examples of positive control immunostainings for somatostatin receptors (SST) and CXCR4. SST2, SST3 and SST5: pancreatic islets; CXCR4: germinal center of a lymph node; ETA: placenta. Immunohistochemistry (red-brown color), counterstaining with hematoxylin; scale bar: 20 µm (TIF 18252 KB)
432_2018_2645_MOESM2_ESM.tif
Supplemental Figure S2: Overall survival of patients with WHO grade I-IV astrocytic brain tumors (log-rank-test: p = 0.003) (TIF 929 KB)
Rights and permissions
About this article
Cite this article
Lange, F., Kaemmerer, D., Behnke-Mursch, J. et al. Differential somatostatin, CXCR4 chemokine and endothelin A receptor expression in WHO grade I–IV astrocytic brain tumors. J Cancer Res Clin Oncol 144, 1227–1237 (2018). https://doi.org/10.1007/s00432-018-2645-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-018-2645-1