Abstract
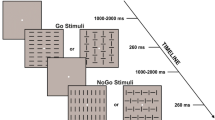
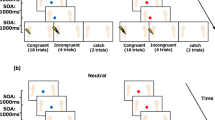
The present work follows recent evidences of studies showing that visual stimuli evoke two early prefrontal event-related potentials (ERP) concomitant to the canonical occipital activities, but originating within the anterior insula (the pN1 and the pP1 components). To clarify the exogenous/endogenous nature of these components, we performed two experiments in which stimulus physical features (Experiment 1) and motor demands of the task (Experiment 2) were considered. In a simple response task (SRT), low-visibility stimuli evoked larger pN1 over the prefrontal areas (Experiment 1) with respect to high-visibility stimuli; in contrast, the occipital P1 component (concomitant to the pN1) had reduced amplitude in the low-visibility condition as expected. Furthermore, the latency of the P1, pN1 and pP1 was slower in the low-visibility condition (from 8 to 18 ms), and the motor response was slowed down as well (on average 14 ms). Pre-stimulus analysis showed that low-visibility stimuli were preceded by greater motor readiness. On the other hand, Experiment 2 showed that, compared with the SRT, the request to passively view the same stimuli was associated with smaller pP1. ERP source analysis confirmed the anterior insula source of the prefrontal ERPs; we interpreted these activities as the correlate of two top-down perceptual processing: the sensory awareness (the pN1) and the awareness of the sensory–motor integration (the pP1), associated with the subjective experience of the visual perception and the conscious experience of the sensory–motor coupling, respectively.









Similar content being viewed by others
References
Allman JM, Watson KK, Tetreault NA, Hakeem AY (2005) Intuition and autism: a possible role for Von Economo neurons. Trends Cogn Sci 9(8):367–373
Band GP, Ridderinkhof KR, van der Molen MW (2003) Speed-accuracy modulation in case of conflict: the roles of activation and inhibition. Psychol Res 67(4):266–279
Barbas H (1988) Anatomic organization of basoventral and mediodorsal visual recipient prefrontal regions in the rhesus monkey. J Comp Neurol 276(3):313–342
Barbas H (1993) Organization of cortical afferent input to orbitofrontal areas in the rhesus monkey. Neuroscience 56(4):841–864
Barbas H, Mesulam MM (1981) Organization of afferent input to subdivisions of area 8 in the rhesus monkey. J Comp Neurol 200(3):407–431
Berchicci M, Spinelli D, Di Russo F (2016) New insights into old waves. Matching stimulus- and response-locked ERPs on the same time-window. Biol Psychol 117:202–215
Bianco V, Di Russo F, Perri RL, Berchicci M (2017a) Different proactive and reactive action control in Fencers’ and Boxers’ brain. Neuroscience 343:260–268
Bianco V, Berchicci M, Perri RL, Quinzi F, Di Russo F (2017b) Exercise-related cognitive effects on sensory–motor control in athletes and drummers compared to non-athletes and other musicians. Neuroscience 360:39–47
Bianco V, Berchicci M, Perri RL, Spinelli D, Russo D, F (2017c) The proactive self-control of actions: time-course of underlying brain activities. Neuroimage 156:388–393
Bobak P, Bodis-Wollner I, Guillory S (1987) The effect of blur and contrast of VEP latency: comparison between check and sinusoidal grating patterns. Electroencephalogr Clin Neurophysiol Evoked Potentials Sect 68(4):247–255
Böcker KB, Brunia CH, van den Berg-Lenssen MM (1994) A spatiotemporal dipole model of the stimulus preceding negativity (SPN) prior to feedback stimuli. Brain Topogr 7(1):71–88
Bullier J, Hupé JM, James A, Girard P (1996) Functional interactions between areas V1 and V2 in the monkey. J Physiol Paris 90(3–4):217–220
Busch NA, Debener S, Kranczioch C, Engel AK, Herrmann CS (2004) Size matters: effects of stimulus size, duration and eccentricity on the visual gamma-band response. Clin Neurophysiol 115(8):1810–1820
Campbell FW, Kulikowski JJ (1972) The visual evoked potential as a function of contrast of a grating pattern. J Physiol 222(2):345
Carretié L, Albert J, López-Martín S, Tapia M (2009) Negative brain: an integrative review on the neural processes activated by unpleasant stimuli. Int J Psychophysiol 71(1):57–63
Cavada C, Compañy T, Tejedor J, Cruz-Rizzolo RJ, Reinoso-Suárez F (2000) The anatomical connections of the macaque monkey orbitofrontal cortex. A review. Cereb Cortex 10(3):220–242
Chakor H, Bertone A, McKerral M, Faubert J, Lachapelle P (2005) Visual evoked potentials and reaction time measurements to motion-reversal luminance- and texture-defined stimuli. Doc Ophthalmol 110(2–3):163–172
Clascá F, Llamas A, Reinoso-Suárez F (1997) Insular cortex and neighboring fields in the cat: a redefinition based on cortical microarchitecture and connections with the thalamus. J Comp Neurol 384(3):456–482
Corbetta M, Patel G, Shulman GL (2008) The reorienting system of the human brain: from environment to theory of mind. Neuron 58(3):306–324
Craig AD (2009a) How do you feel—now? the anterior insula and human awareness. Nat Rev Neurosci 10:1
Craig AD (2009b) Emotional moments across time: a possible neural basis for time perception in the anterior insula. Philos Trans Biol Sci 364:1933–1942
Craig AD, Bushnell MC, Zhang ET, Blomqvist A (1994) A thalamic nucleus specific for pain and temperature sensation. Nature 372(6508):770–773
Dehaene S, Naccache L, Cohen L, Le Bihan D, Mangin JF, Poline JB, Rivière D (2001) Cerebral mechanisms of word masking and unconscious repetition priming. Nat Neurosci 4(7):752–758
Desikan RS, Ségonne F, Fischl B, Quinn BT, Dickerson BC, Blacker D, Albert MS (2006) An automated labeling system for subdividing the human cerebral cortex on MRI scans into gyral based regions of interest. Neuroimage 31(3):968–980
Di Russo F, Lucci G, Sulpizio V, Berchicci M, Spinelli D, Pitzalis S, Galati G (2016) Spatiotemporal brain mapping during preparation, perception, and action. Neuroimage 126:1–14
Di Russo F, Berchicci M, Bozzacchi C, Perri RL, Pitzalis S, Spinelli D (2017) Beyond the “Bereitschaftspotential”: action preparation behind cognitive functions. Neurosci Biobehav Rev 78:57–81
Downar J, Crawley AP, Mikulis DJ, Davis KD (2000) A multimodal cortical network for the detection of changes in the sensory environment. Nat Neurosci 3(3):277–283
Flynn FG (1999) Anatomy of the insula functional and clinical correlates. Aphasiology 13(1):55–78
Gonçalves OF, Rego G, Conde T, Leite J, Carvalho S, Lapenta OM, Boggio PS (2018) Mind wandering and task-focused attention: ERP correlates. Sci Rep 8(1):7608
Hilgetag CC, Medalla M, Beul SF, Barbas H (2016) The primate connectome in context: principles of connections of the cortical visual system. Neuroimage 134:685–702
Ho TC, Brown S, Serences JT (2009) Domain general mechanisms of perceptual decision making in human cortex. J Neurosci 29(27):8675–8687
Ivanoff J, Branning P, Marois R (2009) Mapping the pathways of information processing from sensation to action in four distinct sensorimotor tasks. Hum Brain Mapp 30(12):4167–4186
Johannes S, Münte TF, Heinze HJ, Mangun GR (1995) Luminance and spatial attention effects on early visual processing. Cogn Brain Res 2(3):189–205
Joyce MKP, Barbas H (2018) Cortical connections position primate area 25 as a keystone for interoception, emotion, and memory. J Neurosci 38(7):1677–1698
Jung TP, Makeig S, Humphries C, Lee TW, Mckeown MJ, Iragui V, Sejnowski TJ (2000) Removing electroencephalographic artifacts by blind source separation. Psychophysiology 37(02):163–178
Klados MA, Papadelis CL, Bamidis PD (2009) REG-ICA: a new hybrid method for eog artifact rejection. In: Information technology and applications in biomedicine, 2009. ITAB 2009. 9th international conference on IEEE, pp 1–4
Lamichhane B, Adhikari BM, Dhamala M (2016) The activity in the anterior insulae is modulated by perceptual decision-making difficulty. Neuroscience 327:79–94
Lamme VA (2000) Neural mechanisms of visual awareness: a linking proposition. Brain Mind 1(3):385–406
Lee JH, van Donkelaar P (2002) Dorsal and ventral visual stream contributions to perception–action interactions during pointing. Exp Brain Res 143(4):440–446
Lewis JW, Beauchamp MS, DeYoe EA (2000) A comparison of visual and auditory motion processing in human cerebral cortex. Cereb Cortex 10(9):873–888
Luck SJ, Heinze HJ, Mangun GR, Hillyard SA (1990) Visual event-related potentials index focused attention within bilateral stimulus arrays. II. Functional dissociation of P1 and N1 components. Electroencephalogr Clin Neurophysiol 75(6):528–542
Lumer ED, Rees G (1999) Covariation of activity in visual and prefrontal cortex associated with subjective visual perception. Proc Natl Acad Sci 96(4):1669–1673
Martinez A, Anllo-Vento L, Sereno MI, Frank LR, Buxton RB, Dubowitz DJ, Hillyard SA (1999) Involvement of striate and extrastriate visual cortical areas in spatial attention. Nat Neurosci 2(4):364–369
Menon V (2011) Large-scale brain networks and psychopathology: a unifying triple network model. Trends Cogn Sci 15(10):483–506
Menon V, Uddin LQ (2010) Saliency, switching, attention and control: a network model of insula function. Brain Struct Funct 214(5–6):655–667
Milner AD, Goodale MA (2008) Two visual systems re-viewed. Neuropsychologia 46(3):774–785
Miniussi C, Rao A, Nobre AC (2002) Watching where you look: modulation of visual processing of foveal stimuli by spatial attention. Neuropsychologia 40(13):2448–2460
Mufson EJ, Mesulam MM (1982) Insula of the old world monkey. II: afferent cortical input and comments on the claustrum. J Comp Neurol 212(1):23–37
Mufson EJ, Mesulam M (1984) Thalamic connections of the insula in the rhesus monkey and comments on the paralimbic connectivity of the medial pulvinar nucleus. J Comp Neurol 227(1):109–120
Müller TJ, Federspiel A, Horn H, Lövblad K, Lehmann C, Dierks T, Strik WK (2005) The neurophysiological time pattern of illusionary visual perceptual transitions: a simultaneous EEG and fMRI study. Int J Psychophysiol 55(3):299–312
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 104:199–206
Perri RL, Di Russo F (2017) Executive functions and performance variability measured by event-related potentials to understand the neural bases of perceptual decision-making. Front Hum Neurosci 11:556
Perri RL, Berchicci M, Spinelli D, Di Russo F (2014a) Individual differences in response speed and accuracy are associated to specific brain activities of two interacting systems. Front Behav Neurosci 8:251
Perri RL, Berchicci M, Lucci G, Cimmino RL, Bello A, Di Russo F (2014b) Getting ready for an emotion: specific premotor brain activities for self-administered emotional pictures. Front Behav Neurosci 8:197
Perri RL, Berchicci M, Lucci G, Spinelli D, Di Russo F (2015a) Why do we make mistakes? Neurocognitive processes during the preparation–perception–action cycle and error-detection. Neuroimage 113:320–328
Perri RL, Berchicci M, Lucci G, Spinelli D, Di Russo F (2015b) The premotor role of the prefrontal cortex in response consistency. Neuropsychology 29(5):767–775
Perri RL, Berchicci M, Lucci G, Spinelli D, Di Russo F (2016) How the brain prevents a second error in a perceptual decisionmaking task. Sci Rep 6:32058
Perri RL, Spinelli D, Di Russo F (2017) Missing the target: the neural processing underlying the omission error. Brain Topogr 30(3):352–363
Perri RL, Berchicci M, Bianco V, Spinelli D, Di Russo F (2018) Brain waves from an “isolated” cortex: contribution of the anterior insula to cognitive functions. Brain Struct Funct 223(3):1343–1355
Picton TW, Bentin S, Berg P, Donchin E, Hillyard SA, Johnson R, Taylor MJ (2000) Guidelines for using human event-related potentials to study cognition: recording standards and publication criteria. Psychophysiology 37(2):127–152
Pitzalis S, Strappini F, Bultrini A, Di Russo F (2018) Detailed spatiotemporal brain mapping of chromatic vision combining high-resolution VEP with fMRI and retinotopy. Hum Brain Mapp. https://doi.org/10.1002/hbm.24046 (in press)
Quinzi F, Perri RL, Berchicci M, Bianco V, Pitzalis S, Zeri F, Di Russo F. Weak proactive cognitive/motor brain control accounts for poor children’s behavioral performance in speeded discrimination tasks. Biol Psychol (in press)
Rees G (2007) Neural correlates of the contents of visual awareness in humans. Philos Trans R Soc Lond B Biol Sci 362(1481):877–886
Romanski LM, Giguere M, Bates JF, Goldman-Rakic PS (1997) Topographic organization of medial pulvinar connections with the prefrontal cortex in the rhesus monkey. J Comp Neurol 379(3):313–332
Sanchez-Lopez J, Pedersini CA, Di Russo F, Cardobi N, Fonte C, Varalta V, Marzi CA (2017) Visually evoked responses from the blind field of hemianopic patients. Neuropsychologia
Schadow J, Lenz D, Thaerig S, Busch NA, Fründ I, Rieger JW, Herrmann CS (2007) Stimulus intensity affects early sensory processing: visual contrast modulates evoked gamma-band activity in human EEG. Int J Psychophysiol 66(1):28–36
Seeley WW, Menon V, Schatzberg AF, Keller J, Glover GH, Kenna H, Greicius MD (2007) Dissociable intrinsic connectivity networks for salience processing and executive control. J Neurosci 27(9):2349–2356
Sestokas AK, Lehmkuhle S (1986) Visual response latency of X- and Y-cells in the dorsal lateral geniculate nucleus of the cat. Vis Res 26(7):1041–1054
Shibasaki H, Hallett M (2006) What is the Bereitschaftspotential? Clin Neurophysiol 117(11):2341–2356
Sterzer P, Kleinschmidt A (2010) Anterior insula activations in perceptual paradigms: often observed but barely understood. Brain Struct Funct 214(5–6):611–622
Sulpizio V, Lucci G, Berchicci M, Galati G, Pitzalis S, Di Russo F (2017) Hemispheric asymmetries in the transition from action preparation to execution. Neuroimage 148:390–402
Tadel F, Baillet S, Mosher JC, Pantazis D, Leahy RM (2011) Brainstorm: a user-friendly application for MEG/EEG analysis. Comput Intell Neurosci 2011:8
Treder MS, Blankertz B (2010) C) overt attention and visual speller design in an ERP-based brain–computer interface. Behav Brain Funct 6(1):1
Uddin LQ, Supekar K, Amin H, Rykhlevskaia E, Nguyen DA, Greicius MD, Menon V (2010) Dissociable connectivity within human angular gyrus and intraparietal sulcus: evidence from functional and structural connectivity. Cereb Cortex 20(11):2636–2646
Vassilev A, Stomonyakov V, Manahilov V (1994) Spatial-frequency specific contrast gain and flicker masking of human transient VEP. Vis Res 34(7):863–872
Westwood DA, Chapman CD, Roy EA (2000) Pantomimed actions may be controlled by the ventral visual stream. Exp Brain Res 130(4):545–548
Wright MJ, Johnston A (1982) The effects of contrast and length of gratings on the visual evoked potential. Vis Res 22(11):1389–1399
Yeterian EH, Pandya DN (2010) Fiber pathways and cortical connections of preoccipital areas in rhesus monkeys. J Comp Neurol 518(18):3725–3751
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Perri, R.L., Berchicci, M., Bianco, V. et al. Awareness of perception and sensory–motor integration: ERPs from the anterior insula. Brain Struct Funct 223, 3577–3592 (2018). https://doi.org/10.1007/s00429-018-1709-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-018-1709-y