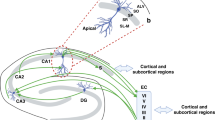
Abstract
Patients with neuropsychiatric and neurological disorders often express limbic circuit abnormalities and deficits in information processing. While these disorders appear to have diverse etiologies, their common features suggest neurodevelopmental origins. Neurodevelopment is a prolonged process of diverse events including neurogenesis/apoptosis, axon pathfinding, synaptogenesis, and pruning, to name a few. The precise timing of the neurodevelopmental insult to these processes likely determines the resulting functional outcome. We used the epilepsy and schizophrenia-related gestational day 17 methylazoxymethanol acetate model to examine the impact of this timed neurodevelopmental insult on principal cell morphology and synaptic network function of the dorsal hippocampus (dHPC) circuit. Our observed structural and functional alterations in dHPC are compartment specific, indicating that adverse global exposure during gestation can produce specific alterations and distort information processing in neural circuits that underlie cognitive abilities.





Similar content being viewed by others
References
Amaral DG, Scharfman HE, Lavenex P (2007) The dentate gyrus: fundamental neuroanatomical organization (dentate gyrus for dummies). Prog Brain Res 163:3–22. https://doi.org/10.1016/S0079-6123(07)63001-5
Bayer SA (1980) Development of the hippocampal region in the rat. I. Neurogenesis examined with 3H-thymidine autoradiography. J Comp Neurol 190(1):87–114. https://doi.org/10.1002/cne.901900107
Brankack J, Stewart M, Fox SE (1993) Current source density analysis of the hippocampal theta rhythm: associated sustained potentials and candidate synaptic generators. Brain Res 615(2):310–327
Cimadevilla JM, Wesierska M, Fenton AA, Bures J (2001) Inactivating one hippocampus impairs avoidance of a stable room-defined place during dissociation of arena cues from room cues by rotation of the arena. Proc Natl Acad Sci USA 98(6):3531–3536. https://doi.org/10.1073/pnas.05162839898/6/3531
Colgin LL, Denninger T, Fyhn M, Hafting T, Bonnevie T, Jensen O, Moser MB, Moser EI (2009) Frequency of gamma oscillations routes flow of information in the hippocampus. Nature 462(7271):353–357. https://doi.org/10.1038/nature08573
Cross-Disorder Group of the Psychiatric Genomics C (2013) Identification of risk loci with shared effects on five major psychiatric disorders: a genome-wide analysis. Lancet 381(9875):1371–1379. https://doi.org/10.1016/S0140-6736(12)62129-1
Danglot L, Triller A, Marty S (2006) The development of hippocampal interneurons in rodents. Hippocampus 16(12):1032–1060. https://doi.org/10.1002/hipo.20225
Diamantaki M, Frey M, Berens P, Preston-Ferrer P, Burgalossi A (2016) Sparse activity of identified dentate granule cells during spatial exploration. Elife. https://doi.org/10.7554/eLife.20252
Fries P (2009) The model- and the data-gamma. Neuron 64(5):601–602. https://doi.org/10.1016/j.neuron.2009.11.024
Gastambide F, Taylor AM, Palmer C, Svard H, Karjalainen M, Janhunen SK, Tricklebank M, Bannerman DM (2015) Alterations in spatial memory and anxiety in the MAM E17 rat model of hippocampal pathology in schizophrenia. Psychopharmacology 232(21–22):4099–4112. https://doi.org/10.1007/s00213-014-3862-1
Gourevitch R, Rocher C, Le Pen G, Krebs MO, Jay TM (2004) Working memory deficits in adult rats after prenatal disruption of neurogenesis. Behav Pharmacol 15(4):287–292
Hernan AE, Mahoney JM, Curry W, Richard G, Lucas MM, Massey A, Holmes GL, Scott RC (2018) Environmental enrichment normalizes hippocampal timing coding in a malformed hippocampus. PLoS One 13(2):e0191488. https://doi.org/10.1371/journal.pone.0191488
Herreras O (1990) Propagating dendritic action potential mediates synaptic transmission in CA1 pyramidal cells in situ. J Neurophysiol 64(5):1429–1441. https://doi.org/10.1152/jn.1990.64.5.1429
Insel TR (2010) Rethinking schizophrenia. Nature 468(7321):187–193. https://doi.org/10.1038/nature09552
Jaaro-Peled H, Hayashi-Takagi A, Seshadri S, Kamiya A, Brandon NJ, Sawa A (2009) Neurodevelopmental mechanisms of schizophrenia: understanding disturbed postnatal brain maturation through neuregulin-1-ErbB4 and DISC1. Trends Neurosci 32(9):485–495. https://doi.org/10.1016/j.tins.2009.05.007
Jenks KR, Lucas MM, Duffy BA, Robbins AA, Gimi B, Barry JM, Scott RC (2013) Enrichment and training improve cognition in rats with cortical malformations. PLoS One 8(12):e84492. https://doi.org/10.1371/journal.pone.0084492
Kirkpatrick B, Buchanan RW, Ross DE, Carpenter WT Jr (2001) A separate disease within the syndrome of schizophrenia. Arch Gen Psychiatry 58(2):165–171
Le Pen G, Gourevitch R, Hazane F, Hoareau C, Jay TM, Krebs MO (2006) Peri-pubertal maturation after developmental disturbance: a model for psychosis onset in the rat. Neuroscience 143(2):395–405. https://doi.org/10.1016/j.neuroscience.2006.08.004
Lewis DA, Levitt P (2002) Schizophrenia as a disorder of neurodevelopment. Annu Rev Neurosci 25:409–432. https://doi.org/10.1146/annurev.neuro.25.112701.142754
Lieberman JA, Stroup TS, McEvoy JP, Swartz MS, Rosenheck RA, Perkins DO, Keefe RS, Davis SM, Davis CE, Lebowitz BD, Severe J, Hsiao JK, Clinical Antipsychotic Trials of Intervention Effectiveness I (2005) Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med 353(12):1209–1223. https://doi.org/10.1056/NEJMoa051688
Lodge DJ (2013) The MAM rodent model of schizophrenia. Curr Protoc Neurosci Chap 9:43. https://doi.org/10.1002/0471142301.ns0943s63
Lodge DJ, Grace AA (2007) Aberrant hippocampal activity underlies the dopamine dysregulation in an animal model of schizophrenia. J Neurosci 27(42):11424–11430. https://doi.org/10.1523/JNEUROSCI.2847-07.2007
Lodge DJ, Grace AA (2009) Gestational methylazoxymethanol acetate administration: a developmental disruption model of schizophrenia. Behav Brain Res 204(2):306–312. https://doi.org/10.1016/j.bbr.2009.01.031
Lucas MM, Lenck-Santini PP, Holmes GL, Scott RC (2011) Impaired cognition in rats with cortical dysplasia: additional impact of early-life seizures. Brain 134(Pt 6):1684–1693. https://doi.org/10.1093/brain/awr087
Marder E, Bucher D (2007) Understanding circuit dynamics using the stomatogastric nervous system of lobsters and crabs. Annu Rev Physiol 69:291–316. https://doi.org/10.1146/annurev.physiol.69.031905.161516
Matricon J, Bellon A, Frieling H, Kebir O, Le Pen G, Beuvon F, Daumas-Duport C, Jay TM, Krebs MO (2010) Neuropathological and Reelin deficiencies in the hippocampal formation of rats exposed to MAM; differences and similarities with schizophrenia. PLoS One 5(4):e10291. https://doi.org/10.1371/journal.pone.0010291
Mitchell KJ, O’Donnell P, Durstewitz D, Fenton AA, Gingrich JA, Gordan JA, Kelsch W, Moghaddam B, Phillips WA, Sawa A (2013) A framework for the use of models in schizophrenia. In: Silverstein SM, Moghaddam B, Wykes T (eds) Schizophrenia: evaluation and synthesis. MIT, Cambridge, pp 212–226
Moore H, Jentsch JD, Ghajarnia M, Geyer MA, Grace AA (2006) A neurobehavioral systems analysis of adult rats exposed to methylazoxymethanol acetate on E17: implications for the neuropathology of schizophrenia. Biol Psychiatry 60(3):253–264. https://doi.org/10.1016/j.biopsych.2006.01.003
O’Reilly KC, Perica MI, Fenton AA (2016) Memory deficits with intact cognitive control in the methylazoxymethanol acetate (MAM) exposure model of neurodevelopmental insult. Neurobiol Learn Mem. https://doi.org/10.1016/j.nlm.2016.07.034
Olypher AV, Klement D, Fenton AA (2006) Cognitive disorganization in hippocampus: a physiological model of the disorganization in psychosis. J Neurosci 26(1):158–168. https://doi.org/10.1523/JNEUROSCI.2064-05.2006
Park EH, Burghardt NS, Dvorak D, Hen R, Fenton AA (2015) Experience-dependent regulation of dentate gyrus excitability by adult-born granule cells. J Neurosci 35(33):11656–11666. https://doi.org/10.1523/JNEUROSCI.0885-15.2015
Pastalkova E, Serrano P, Pinkhasova D, Wallace E, Fenton AA, Sacktor TC (2006) Storage of spatial information by the maintenance mechanism of LTP. Science 313(5790):1141–1144. https://doi.org/10.1126/science.1128657
Pavlowsky A, Wallace E, Fenton AA, Alarcon JM (2017) Persistent modifications of hippocampal synaptic function during remote spatial memory. Neurobiol Learn Mem 138:182–197. https://doi.org/10.1016/j.nlm.2016.08.015
Prinz AA, Bucher D, Marder E (2004) Similar network activity from disparate circuit parameters. Nat Neurosci 7(12):1345–1352. https://doi.org/10.1038/nn1352
Ratajczak P, Kus K, Murawiecka P, Slodzinska I, Giermaziak W, Nowakowska E (2015) Biochemical and cognitive impairments observed in animal models of schizophrenia induced by prenatal stress paradigm or methylazoxymethanol acetate administration. Acta Neurobiol Exp (Wars) 75(3):314–325
Risher WC, Ustunkaya T, Singh Alvarado J, Eroglu C (2014) Rapid Golgi analysis method for efficient and unbiased classification of dendritic spines. PLoS One 9(9):e107591. https://doi.org/10.1371/journal.pone.0107591
Sanderson TM, Cotel MC, O’Neill MJ, Tricklebank MD, Collingridge GL, Sher E (2012) Alterations in hippocampal excitability, synaptic transmission and synaptic plasticity in a neurodevelopmental model of schizophrenia. Neuropharmacology 62(3):1349–1358. https://doi.org/10.1016/j.neuropharm.2011.08.005
Schulz DJ, Goaillard JM, Marder EE (2007) Quantitative expression profiling of identified neurons reveals cell-specific constraints on highly variable levels of gene expression. Proc Natl Acad Sci U S A 104(32):13187–13191. https://doi.org/10.1073/pnas.0705827104
Soriano E, Del Rio JA, Martinez A, Super H (1994) Organization of the embryonic and early postnatal murine hippocampus. I. Immunocytochemical characterization of neuronal populations in the subplate and marginal zone. J Comp Neurol 342(4):571–595. https://doi.org/10.1002/cne.903420406
Talbot ZN, Sparks FT, Dvorak D, Curran BM, Alarcon JM, Fenton AA (2018) Normal CA1 place fields but discoordinated network discharge in a Fmr1-null mouse model of fragile X syndrome. Neuron 97(3):684–697 e684. https://doi.org/10.1016/j.neuron.2017.12.043
Vorstman JA, Breetvelt EJ, Thode KI, Chow EW, Bassett AS (2013) Expression of autism spectrum and schizophrenia in patients with a 22q11.2 deletion. Schizophr Res 143(1):55–59. https://doi.org/10.1016/j.schres.2012.10.010
Weinberger DR (1987) Implications of normal brain development for the pathogenesis of schizophrenia. Arch Gen Psychiatry 44(7):660–669
Weinberger DR (1996) On the plausibility of “the neurodevelopmental hypothesis” of schizophrenia. Neuropsychopharmacology 14(3 Suppl):1S–11S. https://doi.org/10.1016/0893-133X(95)00199-N
Wu K, Leung LS (2003) Increased dendritic excitability in hippocampal ca1 in vivo in the kainic acid model of temporal lobe epilepsy: a study using current source density analysis. Neuroscience 116(2):599–616
Acknowledgements
This work was supported by NIH-R01MH084038 and NIH-R25NS080686-06. The authors would like to thank Drs. Hsin-Yi Kao and Edith Lesburgueres for help generating control and GD17-MAM rats. We would also like to thank Yi-Wen Chen and Dr. Chiye Aoki for assistance with the Golgi studies. Preliminary reports of these data have been present at the Annual meetings for Society for Neuroscience (2016) and the Canadian Association for Neuroscience (2016).
Author information
Authors and Affiliations
Contributions
KCO, AVP, and MIP collected data, KCO and ERJL performed analyses, and KCO and AAF designed research and wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts to report.
Animal welfare
All methods complied with Public Health and Service Policy on Humane Care and Use of Laboratory Animals and were approved by the New York University Animal Welfare Committee.
Rights and permissions
About this article
Cite this article
O’Reilly, K.C., Levy, E.R.J., Patino, A.V. et al. Sub-circuit alterations in dorsal hippocampus structure and function after global neurodevelopmental insult. Brain Struct Funct 223, 3543–3556 (2018). https://doi.org/10.1007/s00429-018-1704-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-018-1704-3