Abstract
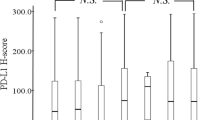
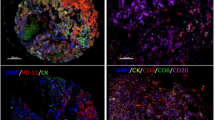
Several cancers, especially non-small cell lung cancer (NSCLC), are able to escape the immunosurveillance of tumor-infiltrating lymphocytes (TILs); among the molecules involved, the indoleamine 2,3-dioxygenase 1 (IDO-1) and the programmed cell death ligand-1 (PD-L1) play a crucial role. These aspects are of great interest in the current immunotherapeutic era, therefore the current study analyses the TILs, IDO-1, and PD-L1 interactions and their correlations with clinicopathological parameters and prognosis in NSCLC. One hundred ninety-three NSCLC surgical specimens, formalin-fixed, and paraffin-embedded were assessed for TILs density, TILs localization, IDO-1 (clone 4.16H1), and PD-L1 (clone E1L3N) immunohistochemical expressions. This data was correlated with clinicopathological parameters, disease free, and overall survivals. IDO-1 and PD-L1 high expressions were related to the solid pattern of adenocarcinomas (respectively p = 0.036 and p = 0.026); high PD-L1 expression was correlated with squamous histotype (p = 0.048). IDO-1 overexpression correlated with former smokers (p = 0.041), higher adenocarcinoma stages (p = 0.039), and with both higher TILs density and PD-L1 expression (respectively p = 0.025 and p = 0.0003). A better prognosis was associated with TILs intratumoral or mixed localizations (p = 0.029). TILs localization affects NSCLC prognosis; the higher expression of IDO-1 and PD-L1 in poorly differentiated and more aggressive lung adenocarcinomas, as well as the correlation between high PD-L1 expression and squamous cell histotype, confirm the more efficient immunoescaping of these NSCLC subgroups.


Similar content being viewed by others
References
Travis WD, Brambilla E, Noguchi M, Nicholson AG, Geisinger KR, Yatabe Y, Beer DG, Powell CA, Riely GJ, van Schil PE, Garg K, Austin JHM, Asamura H, Rusch VW, Hirsch FR, Scagliotti G, Mitsudomi T, Huber RM, Ishikawa Y, Jett J, Sanchez-Cespedes M, Sculier JP, Takahashi T, Tsuboi M, Vansteenkiste J, Wistuba I, Yang PC, Aberle D, Brambilla C, Flieder D, Franklin W, Gazdar A, Gould M, Hasleton P, Henderson D, Johnson B, Johnson D, Kerr K, Kuriyama K, Lee JS, Miller VA, Petersen I, Roggli V, Rosell R, Saijo N, Thunnissen E, Tsao M, Yankelewitz D (2011) International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society International Multidisciplinary Classification of Lung Adenocarcinoma. J Thorac Oncol 6(2):244–285. https://doi.org/10.1097/JTO.0b013e318206a221
Gridelli C, Ardizzoni A, Barberis M, Cappuzzo F, Casaluce F, Danesi R, Troncone G, de Marinis F (2017) Predictive biomarkers of immunotherapy for non-small cell lung cancer: results from an expert panel meeting of the Italian Association of Thoracic Oncology. Transl Lung Cancer Res 6(3):373–386. https://doi.org/10.21037/tlcr.2017.05.09
Thunnissen E, Allen TC, Adam J, Aisner DL, Beasley MB, Borczuk AC, Cagle PT, Capelozzi VL, Cooper W, Hariri LP, Kern I, Lantuejoul S, Miller R, Mino-Kenudson M, Radonic T, Raparia K, Rekhtman N, Roy-Chowdhuri S, Russell P, Schneider F, Sholl LM, Tsao MS, Vivero M, Yatabe Y (2018) Immunohistochemistry of pulmonary biomarkers : a perspective from members of the pulmonary pathology society. Arch Pathol Lab Med 142(3):408–419. https://doi.org/10.5858/arpa.2017-0106-SA
Dunn GV, Old LJ, Schreiber RD (2004) The three Es of cancer immunoediting. Annu Rev Immunol 22:329–360
Anguille S, Smits EL, Bryant C, van Acker H, Goossens H, Lion E, Fromm PD, Hart DN, van Tendeloo V, Berneman ZN (2015) Dendritic cells as pharmacological tools for cancer immunotherapy. Pharmacol Rev 67(4):731–753. https://doi.org/10.1124/pr.114.009456
D'errico G, Machado HL, Sainz B Jr (2017) A current perspective on cancer immune therapy: step-by-step approach to constructing the magic bullet. Clin Transl Med 6(1):3. https://doi.org/10.1186/s40169-016-0130-5
Brambilla E, le Teuff G, Marguet S, Lantuejoul S, Dunant A, Graziano S, Pirker R, Douillard JY, le Chevalier T, Filipits M, Rosell R, Kratzke R, Popper H, Soria JC, Shepherd FA, Seymour L, Tsao MS (2016) Prognostic effect of tumor lymphocytic infiltration in resectable non-small-cell lung cancer. J Clin Oncol 34(11):1223–1230. https://doi.org/10.1200/JCO.2015.63.0970
Ruffini E, Asioli S, Filosso PL, Lyberis P, Bruna MC, Macrì L, Daniele L, Oliaro A (2009) Clinical significance of tumor-infiltrating lymphocytes in lung neoplasm. Ann Thorac Surg 87(2):365–372. https://doi.org/10.1016/j.athoracsur.2008.10.067
Zeng DQ, Yu YF, Ou QY, Li XY, Zhong RZ, Xie CM, Hu QG (2016) Prognostic and predictive value of tumor-infiltrating lymphocytes for clinical therapeutic research in patients with non–small cell lung cancer. Oncotarget 7(12):13765–13781. https://doi.org/10.18632/oncotarget.7282
Levina V, Su Y, Gorelik E (2012) Immunological and nonimmunological effects of indoleamine 2,3-dioxygenase on breast tumor growth and spontaneous metastasis formation. Clin Dev Immunol:173029. https://doi.org/10.1155/2012/173029
Brochez L, Chevolet I, Kruse V (2017) The rationale of indoleamine 2,3-dioxygenase inhibition for cancer therapy. Eur J Cancer 76:167–182. https://doi.org/10.1016/j.ejca.2017.01.011
Schalper KA, Carvajal-Hausdorf D, McLaughlin J, Altan M, Velcheti V, Gaule P, Sanmamed MF, Chen L, Herbst RS, Rimm DL (2017) Differential expression and significance of PD-L1, IDO-1 and B7-H4 in human lung cancer. Clin Cancer Res 23(2):370–378. https://doi.org/10.1158/1078-0432.CCR-16-0150
Munn DH, Mellor AL (2013) Indoleamine 2,3 dioxygenase and metabolic control of immune responses. Trends Immunol 34(3):137–143. https://doi.org/10.1016/j.it.2012.10.001
Munn DH (2012) Blocking IDO activity to enhance anti-tumor immunity. Front Biosci (Elite Ed) 4:734–745
Smith C, Chang MY, Parker K et al (2012) IDO is a nodal pathogenic driver of lung cancer and metastasis development. Cancer Discov 2(8):722–735. https://doi.org/10.1158/2159-8290.CD-12-0014
Theate I, van Baren N, Pilotte L, Moulin P, Larrieu P, Renauld JC, Herve C, Gutierrez-Roelens I, Marbaix E, Sempoux C, van den Eynde BJ (2015) Extensive profiling of the expression of the indoleamine 2,3-dioxygenase 1 protein in normal and tumoral human tissues. Cancer Immunol Res 3(2):161–172. https://doi.org/10.1158/2326-6066.CIR-14-0137
Fallarino F, Grohmann U, Puccetti P (2012) Indoleamine 2,3-dioxygenase: from catalyst to signaling function. Eur J Immunol 42(8):1932–1937. https://doi.org/10.1002/eji.201242572
Albini E, Rosini V, Gargaro M, Mondanelli G, Belladonna ML, Pallotta MT, Volpi C, Fallarino F, Macchiarulo A, Antognelli C, Bianchi R, Vacca C, Puccetti P, Grohmann U, Orabona C (2017) Distinct roles of immunoreceptor tyrosine-based motifs in immunosuppressive indoleamine 2,3 dioxygenase 1. J Cell Mol Med 21(1):165–176. https://doi.org/10.1111/jcmm.12954
Tsao MS, Le Teuff G, Shepherd FA et al (2017) PD-L1 protein expression assessed by immunohistochemistry is neither prognostic nor predictive of benefit from adjuvant chemotherapy in resected non-small cell lung cancer. Ann Oncol 28(4):882–889. https://doi.org/10.1093/annonc/mdx003
Yang CY, Lin MW, Chang YL, Wu CT, Yang PC (2016) Programmed cell death-ligand 1 expression is associated with favourable immune microenviroment and better overall survival in stage I pulmonary squamous cell carcinoma. Eur J Cancer 57:91–103. https://doi.org/10.1016/j.ejca.2015.12.033
Callea M, Pedica F, Doglioni C (2016) Programmed death 1 (PD-1) and its ligand (PD-L1) as a new frontier in cancer immunotherapy and challenges for pathologist: state of the art. Pathologica 108(2):48–58
Ilie M, Long-Mira E, Bence C, Butori C, Lassalle S, Bouhlel L, Fazzalari L, Zahaf K, Lalvée S, Washetine K, Mouroux J, Vénissac N, Poudenx M, Otto J, Sabourin JC, Marquette CH, Hofman V, Hofman P (2016) Comparative study of the PD-L1 status between surgically resected specimens and matched biopses of NSCLC patients reveal major discordances: a potential issue for anti-PD-L1 therapeutic strategies. Ann Oncol 27(1):147–153. https://doi.org/10.1093/annonc/mdv489
Herbst RS, Baas P, Kim DW, Felip E, Pérez-Gracia JL, Han JY, Molina J, Kim JH, Arvis CD, Ahn MJ, Majem M, Fidler MJ, de Castro G Jr, Garrido M, Lubiniecki GM, Shentu Y, Im E, Dolled-Filhart M, Garon EB (2016) Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet 387(10027):1540–1550. https://doi.org/10.1016/S0140-6736(15)01281-7
Teng MW, Ngiow SF, Ribas A et al (2015) Classifying cancers based on T cell infiltration and PD-L1. Cancer Res 75(11):2139–2145. https://doi.org/10.1158/0008-5472.CAN-15-0255
Ferdinande L, Decaestecker C, Verset L, Mathieu A, Moles Lopez X, Negulescu AM, van Maerken T, Salmon I, Cuvelier CA, Demetter P (2012) Clinicopathological significance of indoleamine 2,3-dioxygenase 1 expression in colorectal cancer. Br J Cancer 106(1):141–147. https://doi.org/10.1038/bjc.2011.513
Soliman H, Rawal B, Fulp J, Lee JH, Lopez A, Bui MM, Khalil F, Antonia S, Yfantis HG, Lee DH, Dorsey TH, Ambs S (2013) Analysis of indoleamine 2-3 dioxygenase (IDO1) expression in breast cancer tissue by immunohistochemistry. Cancer Immunol Immunother 62(5):829–837. https://doi.org/10.1007/s00262-013-1393-y
Jacquemier J, Bertucci F, Finetti P, Esterni B, Charafe-Jauffret E, Thibult ML, Houvenaeghel G, van den Eynde B, Birnbaum D, Olive D, Xerri L (2012) High expression of indolamine 2,3-dioxygenase in the tumor is associated with medullary features and favourable outcome in basal-like breast carcinoma. Int J Cancer 130(1):96–104. https://doi.org/10.1002/ijc.25979
Kim JW, Nam KH, Ahn SH, Park DJ, Kim HH, Kim SH, Chang H, Lee JO, Kim YJ, Lee HS, Kim JH, Bang SM, Lee JS, Lee KW (2016) Prognostic implications of immunosoppressive protein expression in tumors as well as immune cell infiltration within the tumor microenvironment in gastric cancer. Gastric Cancer 19(1):42–52. https://doi.org/10.1007/s10120-014-0440-5
Cooper WA, Tran T, Vilain RE, Madore J, Selinger CI, Kohonen-Corish M, Yip PY, Yu B, O’Toole SA, McCaughan BC, Yearley JH, Horvath LG, Kao S, Boyer M, Scolyer RA (2015) PD-L1 expression is a favorable prognostic factor in early stage non-small cell carcinoma. Lung Cancer 89(2):181–188. https://doi.org/10.1016/j.lungcan.2015.05.007
Sun JM, Zhou W, Choi YL, Choi SJ, Kim SE, Wang Z, Dolled-Filhart M, Emancipator K, Wu D, Weiner R, Frisman D, Kim HK, Choi YS, Shim YM, Kim J (2016) Prognostic significance of PD-L1 in patients with non–small cell lung cancer: a large cohort study of surgically resected cases. J Thorac Oncol 11(7):1003–1011. https://doi.org/10.1016/j.jtho.2016.04.007
Sorensen SF, Zhou W, Dolled-Filhart M, Georgsen JB, Wang Z, Emancipator K, Wu D, Busch-Sørensen M, Meldgaard P, Hager H (2016) PD-L1 expression and survival among patients with advanced non-small cell lung cancer treated with chemotherapy. Transl Oncol 9(1):64–69. https://doi.org/10.1016/10.1016/j.tranon.2016.01.003
Chen Y, Mu C, Huang J (2012) Clinical significance of programmed death-1 ligand-1 expression in patients with non-small cell lung cancer: a 5-year-follow-up study. Tumori 98(6):751–755. https://doi.org/10.1700/1217.13499
Kim MY, Koh J, Kim S, Go H, Jeon YK, Chung DH (2015) Clinicopathological analysis of PD-L1 and PD-L2 expression in pulmonary squamous cell carcinoma: comparison with tumor-infiltrating T cells and the status of oncogenic drivers. Lung Cancer 88(1):24–33. https://doi.org/10.1016/j.lungcan.2015.01.016
Wang A, Wang HY, Liu Y, Zhao MC, Zhang HJ, Lu ZY, Fang YC, Chen XF, Liu GT (2015) The prognostic value of PD-L1 expression for non-small cell lung cancer patients: a meta-analysis. Eur J Surg Oncol 41(4):450–456. https://doi.org/10.1016/j.ejso.2015.01.020
McLaughlin JF, Schalper K, Carvajal-Hausdforf DE et al (2014) Domain-specific PD-L1 protein measurement in non-small cell lung cancer (NSCLC). J Clin Oncol 32(15):8064. https://doi.org/10.1200/jco.2014.32.15_suppl.8064
Naito Y, Saito K, Shiiba K, Ohuchi A, Saigenji K, Nagura H, Ohtani H (1998) CD8+ T cells infiltrated within cancer cell nests as a prognostic factor in human colorectal cancer. Cancer Res 58(16):3491–3494
Salgado R, Denkert C, Demaria S, Sirtaine N, Klauschen F, Pruneri G, Wienert S, van den Eynden G, Baehner FL, Penault-Llorca F, Perez EA, Thompson EA, Symmans WF, Richardson AL, Brock J, Criscitiello C, Bailey H, Ignatiadis M, Floris G, Sparano J, Kos Z, Nielsen T, Rimm DL, Allison KH, Reis-Filho JS, Loibl S, Sotiriou C, Viale G, Badve S, Adams S, Willard-Gallo K, Loi S, International TILs Working Group 2014 (2015) The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: recommendations by an international TILs working group 2014. Ann Oncol 26(2):259–271. https://doi.org/10.1093/annonc/mdu450
Astigiano S, Morandi B, Costa R, Mastracci L, D'Agostino A, Ratto GB, Melioli G, Frumento G (2005) Eosinophil granulocytes account for indoleamine 2,3-dioxygenase–mediated immune escape in human non-small cell lung cancer. Neoplasia 7(4):390–396. https://doi.org/10.1593/neo.04658
Parra ER, Villalobos P, Zhang J, Behrens C, Mino B, Swisher S, Sepesi B, Weissferdt A, Kalhor N, Heymach JV, Moran C, Zhang J, Lee J, Rodriguez-Canales J, Gibbons D, Wistuba II (2018) Immunohistochemical and image analysis-based study demonstrate that several immune checkpoints are co-expressed in non-small cell lung carcinoma tumors. J Thorac Oncol 13(6):779–791. https://doi.org/10.1016/j.jtho.2018.03.002
Volaric A, Gentzler R, Hall R, Mehaffey JH, Stelow EB, Bullock TN, Martin LW, Mills AM (2018) Indoleamine-2,3-dioxygenase in non-small cell lung cancer: a targetable mechanism of immune resistance frequently coexpressed with PD-L1. Am J Surg Pathol 42(9):1216–1223. https://doi.org/10.1097/PAS.0000000000001099
Brunnström H, Johansson A, Westbom-Fremer S, Backman M, Djureinovic D, Patthey A, Isaksson-Mettävainio M, Gulyas M, Micke P (2017) PD-L1 immunohistochemistry in clinical diagnostics of lung cancer: inter-pathologist variability is higher than assay variability. Mod Pathol 30(10):1411–1421. https://doi.org/10.1038/modpathol.2017.59
Osoegawa A, Hiraishi H, Hashimoto T, Takumi Y, Abe M, Takeuchi H, Miyawaki M, Okamoto T, Sugio K (2018) The positive relationship between γH2AX and PD-L1 expression in lung squamous cell carcinoma. In Vivo 32(1):171–177. https://doi.org/10.21873/invivo.11221
Driver BR, Miller RA, Miller T, Deavers M, Gorman B, Mody D, Ge Y, Barrios R, Bernicker E, Kim M, Cagle PT (2017) Programmed death Ligand-1 (PD-L1) expression in either tumor cells or tumor-infiltrating immune cells correlates with solid and high-grade lung adenocarcinomas. Arch Pathol Lab Med 141(11):1529–1532. https://doi.org/10.5858/arpa.2017-0028-OA
Xie Q, Wang L, Zhu B, Wang Y, Gu J, Chen Z (2008) The expression and significance of indoleamine -2,3-dioxygenase in non small cell lung cancer cell. Zhongguo Fei Ai Za Zhi 11(1):115–119. https://doi.org/10.3779/j.issn.1009-3419.2008.01.025
Tumeh PC, Harview CL, Yearley JH, Shintaku IP, Taylor EJM, Robert L, Chmielowski B, Spasic M, Henry G, Ciobanu V, West AN, Carmona M, Kivork C, Seja E, Cherry G, Gutierrez AJ, Grogan TR, Mateus C, Tomasic G, Glaspy JA, Emerson RO, Robins H, Pierce RH, Elashoff DA, Robert C, Ribas A (2014) PD-1 blockade induces responses by inhibiting adaptive immune resistance. Nature 515(7528):568–571. https://doi.org/10.1038/nature13954
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pagès C, Tosolini M, Camus M, Berger A, Wind P, Zinzindohoué F, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Pagès F (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313(5795):1960–1964. https://doi.org/10.1126/science.1129139
Hiraoka K, Miyamoto M, Cho Y, Suzuoki M, Oshikiri T, Nakakubo Y, Itoh T, Ohbuchi T, Kondo S, Katoh H (2006) Concurrent infiltration by CD8+ T cells and CD4+ T cells is a favourable prognostic factor in non small cell lung carcinoma. Br J Cancer 94(2):275–280. https://doi.org/10.1038/sj.bjc.6602934
Lin G, Fan X, Zhu W, Huang C, Zhuang W, Xu H, Lin X, Hu D, Huang Y, Jiang K, Miao Q, Li C (2017) Prognostic significance of PD-L1 expression and tumor infiltrating lymphocyte in surgically resectable non-small cell lung cancer. Oncotarget 8(48):83986–83994. https://doi.org/10.18632/oncotarget.20233
Gaule P, Smithy JW, Toki M, Rehman J, Patell-Socha F, Cougot D, Collin P, Morrill P, Neumeister V, Rimm DL (2016) A quantitative comparison of antibodies to programmed cell death 1 ligand 1. JAMA Oncol 3:256. https://doi.org/10.1001/jamaoncol.2016.3015
Rimm DL, Han G, Taube JM, Yi ES, Bridge JA, Flieder DB, Homer R, West WW, Wu H, Roden AC, Fujimoto J, Yu H, Anders R, Kowalewski A, Rivard C, Rehman J, Batenchuk C, Burns V, Hirsch FR, Wistuba II (2017) A prospective, multi-institutional assessment of four assays for PD-L1 expression in NSCLC by immunohistochemistry. JAMA Oncol 3(8):1051–1058. https://doi.org/10.1001/jamaoncol.2017.0013
McLaughlin J, Han G, Schalper KA, Carvajal-Hausdorf D, Pelekanou V, Rehman J, Velcheti V, Herbst R, LoRusso P, Rimm DL (2016) Quantitative assessment of the heterogeneity of PD-L1 expression in non-small-cell lung cancer. JAMA Oncol 2(1):46–54. https://doi.org/10.1001/jamaoncol.2015.3638
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This manuscript is not under consideration elsewhere. There are no financial disclosures for all authors or funding sources for the manuscript. The work has been prepared according to ethical adherence regarding the informed consent of the involved human participants (Number of Local Ethic Committee Decision: N. 2216/13).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mandarano, M., Bellezza, G., Belladonna, M.L. et al. Assessment of TILs, IDO-1, and PD-L1 in resected non-small cell lung cancer: an immunohistochemical study with clinicopathological and prognostic implications. Virchows Arch 474, 159–168 (2019). https://doi.org/10.1007/s00428-018-2483-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-018-2483-1