Abstract
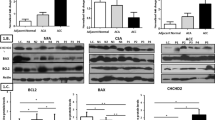
Histamine is involved in the pathogenesis of several tumors; however, there are no data on its possible involvement in human adrenocortical tumorigenesis. The expression of genes and proteins involved in the biosynthesis (histidine decarboxylase, HDC), action (histamine receptors: HRH1–HRH4), and metabolism of histamine is largely unknown both in the normal human adrenal cortex and in adrenocortical tumors. In this study, we examined the expression of histamine-related genes and proteins and histamine content in normal adrenal cortex, benign adrenocortical adenomas, and malignant adrenocortical cancer (ACC). Fifteen normal adrenals and 43 tumors were studied. mRNA expression was examined by real time RT-PCR. Western-blotting and immunohistochemistry were used for the study of proteins. Tissue histamine content was determined by enzyme-linked immunosorbent assay. We found that all proteins involved in histamine biosynthesis and action are present both in the normal adrenal cortex and in the tumors studied. HDC expression and histamine content was highest in the normal tissues and lower in benign tumors, whereas it was significantly less in ACCs. HRH3 expression was significantly higher in ACC samples than in the other groups. Adrenocortical tumorigenesis might, thus, be characterized by reduced histamine biosynthesis; furthermore, different adrenocortical tumor subtypes may show unique histamine receptor expression profiles.




Similar content being viewed by others
Abbreviations
- ACC:
-
Adrenocortical carcinoma
- ACTH:
-
Adrenocorticotropin
- APA:
-
Aldosterone producing adenoma
- cAMP:
-
Cyclic AMP
- CPA:
-
Cortisol-producing adenoma
- DAO:
-
Diamino-oxydase
- DHEA:
-
Dehydroepiandrosterone
- DHEAS:
-
Dehydroepiandrosterone sulfate
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- HDC:
-
Histidine decarboxylase
- HNMT:
-
Histamine-N-methyltransferase
- HRH1:
-
Histamine receptor H1
- HRH2:
-
Histamine receptor H2
- HRH3:
-
Histamine receptor H3
- HRH4:
-
Histamine receptor H4
- IA:
-
Hormonally inactive adenoma
- IFN-γ:
-
Interferon-γ
- IL-10:
-
Interleukin-10
- IL-12:
-
Interleukin-12
- PKA:
-
cAMP-dependent protein kinase
- PRKAR1A:
-
Protein kinase A regulatory-subunit type-I alpha
- QRT-PCR:
-
Quantitative real-time RT-PCR
References
Tomita K, Nakamura E, Okabe S (2005) Histamine regulates growth of malignant melanoma implants via H2R receptors in mice. Inflammopharmacol 13:281–289
Medina V, Croci M, Crescenti E et al (2007) The role of histamine in human mammary carcinogenesis: H3R and H4R receptors as potential therapeutic targets for breast cancer treatment. Cancer Biol Ther 7:28–35
Boer K, Helinger E, Helinger A et al (2008) Decreased expression of histamine H1R and H4R receptors suggests disturbance of local regulation in human colorectal tumors by histamine. Eur J Cell Biol 87:227–236
Tomita K, Izumi K, Okabe S (2003) Roxatidine- and cimetidine-induced angiogenesis inhibition suppresses growth of colon cancer implants in syngeneic mice. J Pharmacol Sci 93:321–330
Morris DL, Adams WJ (1995) Cimetidine and colorectal cancer–old drug, new use? Nat Med 1:1243–1244
Tanimoto A, Matsuki Y, Tomita T et al (2004) Histidine decarboxylase expression in pancreatic endocrine cells and related tumors. Pathol Int 54:408–412
Koch CA, Pacak K, Chrousos GP (2002) The molecular pathogenesis of hereditary and sporadic adrenocortical and adrenomedullary tumors. J Clin Endocrinol Metab 87:5367–5384
Terzolo M, Bovio S, Pia A et al (2005) Midnight serum cortisol as a marker of increased cardiovascular risk in patients with a clinically inapparent adrenal adenoma. Eur J Endocrinol 153:307–315
Assié G, Antoni G, Tissier F et al (2007) Prognostic parameters of metastatic adrenocortical carcinoma. J Clin Endocrinol Metab 92:148–154
Libé R, Fratticci A, Bertherat J (2007) Adrenocortical cancer: pathophysiology and clinical management. Endcrine-Related Cancer 14:13–28
Patalano A, Brancato V, Mantero F (2009) Adrenocortical cancer treatment. Horm Res 71:99–104
Huang JF, Thurmond RL (2008) The new biology of histamine receptors. Curr Allergy Asthma Rep 8:21–27
Chanda R, Ganguly AK (1995) Diamine-oxidase activity and tissue di- and poly-amine contents of human ovarian, cervical and endometrial carcinoma. Cancer Lett 89:23–28
Pacifici GM, Donatelli P, Giuliani L (1992) Histamine N-methyl transferase: inhibition by drugs. Br J Clin Pharmacol 34:322–327
Kellaway CH, Cowell SJ (1922) On the concentration of the blood and the effects of histamine in adrenal insufficiency. J Physiol 57:82–99
Häppölä O, Soinila S, Päivärinta H et al (1985) Histamine-immunoreactive endocrine cells in the adrenal medulla of the rat. Brain Res 339:393–396
Matsuki Y, Tanimoto A, Hamada T et al (2003) Histidine decarboxylase expression as a new sensitive and specific marker for small cell lung carcinoma. Mod Pathol 16:72–78
Marley PD, Thomson KA, Jachno K et al (1991) Histamine-induced increases in cyclic AMP levels in bovine adrenal medullary cells. Br J Pharmacol 104:839–846
Traiffort E, Vizuete ML, Tardivel-Lacombe J et al (1995) The guinea pig histamine H2 receptor: gene cloning, tissue expression and chromosomal localization of its human counterpart. Biochem Biophys Res Commun 211:570–577
Marley PD (2003) Mechanisms in histamine-mediated secretion from adrenal chromaffin cells. Pharmacol Ther 98:1–34
Horny HP, Ruck P, Kröber S et al (1997) Systemic mast cell disease (mastocytosis). General aspects and histopathological diagnosis. Histol Histopathol 12:1081–1089
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25:402–408
Galli SJ, Tsai M, Wershil BK (1993) The c-kit receptor, stem cell factor and mast cells. What each teaching us about the others. Am J Pathol 142:965–974
Shukla SA, Veerappan R, Whittimore JS et al (2006) Mast cell ultrastructure and saining in tissue. Methods Mol Biol 315:63–76
Fukushima Y, Ohmachi Y, Asano T et al (1999) Localization of the histamine H2 receptor, a target for antiulcer drugs, in gastric parietal cells. Digestion 60:522–527
Tetlow LC, Wooley DE (2005) Histamine, histamine receptors (H1 and H2) and histidine decarboxylase expression by chondrocytes of osteoarthritic cartilage: an immunohistochemical study. Rheumatol Int 26:173–178
Yoshida T, Mio M, Tasaka K et al (1997) Histamine-induced cortisol secretion from bovine adrenocortical cells: co-cultured with bovine adrenomedullary cells. Jpn J Pharmacol 75:115–121
Orsó E, Szalay KS, Tóth IE et al (1995) Effect of histamine on corticosteroid secretion of isolated human and rat adrenocortical cells. Inflamm Res 44:S48–S49
Orsó E, Szalay KS, Fehér T (1997) Interaction of histamine with adrenocorticotropic hormone: a local modulatory role for adrenocortical androgen synthesis? Inflamm Res 46:S57–S58
Zhang DX, Gauthier KM, Campbell WB (2005) Mechanisms of histamine-induced relaxation in bovine small adrenal cortical arteries. Am J Physiol Endocrinol Metab 289:E1058–E1063
Krishnaswamy G, Ajitawi O, Chi DS (2006) The human mast cell: an overview. Methods Mol Biol 315:13–34
Hinson JP, Vinson GP, Kapas S et al (1991) The relationship between adrenal vascular events and steroid secretion: the role of mast cells and endothelin. J Steroid Biochem Mol Biol 40:381–389
Lefebvre H, Contesse V, Delarue C et al (1992) Serotonin-induced stimulation of cortisol secretion from human adrenocortical tissue is mediated through activation of a serotonin4 receptor subtype. Neuroscience 47:999–1007
Aiba M, Iri H, Suzuki H et al (1985) Numerous mast cells in an 11-deoxycorticosterone-producing adrenocortical tumor. Arch Pathol Lab Med 109:357–360
Lefebvre H, Cartier D, Duparc C et al (2002) Characterization of serotonin4 receptors in adrenocortical aldosterone-producing adenomas: in vivo and in vitro studies. J Clin Endocrinol Metab 87:1211–1216
Zhang PJ, Genega EM, Tomaszewsky JE et al (2003) The role of calretinin, inhibin, melan-A, BCL-2, and c-kit in differentiating adrenal cortical and medullary tumors: an immunohistochemical study. Mod Pathol 16:591–597
Falus A, Hegyesi H, Lázár-Molnár E et al (2001) Paracrine and autocrine interactions in melanoma: histamine is a relevant player in local regulation. Trends Immunol 22:648–652
Pós Z, Wiener Z, Pócza P et al (2008) Histamine suppresses fibulin-5 and insulin-like growth factor-II receptor expression in melanoma. Cancer Res 68:1997–2005
Jelinek I, László V, Buzás E et al (2007) Increased antigen presentation and T(h)1 polarization in genetically histamine-free mice. Int Immunol 19:51–58
Tomita K, Okabe S (2005) Exogenous histamine stimulates colorectal cancer implant growth via immunosuppression in mice. J Pharmacol Sci 97:116–123
Igaz P, Wiener Z, Szabó P et al (2006) Functional genomics approaches for the study of sporadic adrenal tumor pathogenesis. Clinical implications. J Steriod Biochem Mol Biol 101:87–96
de Esch IJ, Thurmond RL, Jongejan A et al (2005) The histamine H4 receptor as a new therapeutic target for inflammation. Trends Pharmacol Sci 26:462–469
Akdis CA, Simons FE (2006) Histamine receptors are hot in immunopharmacology. Eur J Pharmachol 533:69–76
Soon PS, McDonald KL, Robinson BG et al (2008) Molecular markers and the pathogenesis of adrenocortical cancer. Oncologist 13:548–561
Groussin L, Horvath A, Jullian E et al (2006) A PRKAR1A mutation associated with primary pigmented nodular adrenocortical disease in 12 kindreds. J Clin Endocrinol Metab 91:1943–1949
Acknowledgements
Hungarian Ministry of Health 089/2006, Semmelweis University Research Grant, Hungarian Scientific Research Fund (OTKA) PD72306, Swiss National Fund (FNRS) 3200BO-105657/1) and Fondation pour la Recherche en Endocrinologie, Diabétologie et Métabolisme. Attila Patócs MD PhD is a recipient of a János Bolyai Research Grant. The authors would also like to thank Dr. Gábor L. Kovács (National Health Center, Budapest, Hungary) for supplying some tissue samples.
Conflict of interest
The authors have no conflict of interest to report.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Szabó, P.M., Wiener, Z., Tömböl, Z. et al. Differences in the expression of histamine-related genes and proteins in normal human adrenal cortex and adrenocortical tumors. Virchows Arch 455, 133–142 (2009). https://doi.org/10.1007/s00428-009-0807-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-009-0807-x