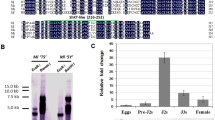
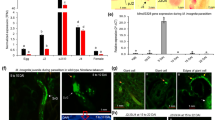
Abstract
Main conclusion
This study revealed novel insights into the function of MSP18 effector during root-knot nematode parasitism in rice roots. MSP18 may modulate host immunity and enhance plant susceptibility to Meloidogyne spp.
Abstract
Rice (Oryza sativa) production is seriously impacted by root-knot nematodes (RKN), including Meloidogyne graminicola, Meloidogyne incognita, and Meloidogyne javanica, in upland and irrigated culture systems. Successful plant infection by RKN is likely achieved by releasing into the host cells some effector proteins to suppress the activation of immune responses. Here, we conducted a series of functional analyses to assess the role of the Meloidogyne-secreted protein (MSP) 18 from M. incognita (Mi-MSP18) during rice infection by RKN. Developmental expression profiles of M. javanica and M. graminicola showed that the MSP18 gene is up-regulated throughout nematode parasitic stages in rice. Reproduction of M. javanica and M. graminicola is enhanced in rice plants overexpressing Mi-MSP18, indicating that the Mi-MSP18 protein facilitates RKN parasitism. Transient expression assays in onion cells suggested that Mi-MSP18 is localized to the cytoplasm of the host cells. In tobacco, Mi-MSP18 suppressed the cell death induced by the INF1 elicitin, suggesting that Mi-MSP18 can interfere with the plant defense pathways. The data obtained in this study highlight Mi-MSP18 as a novel RKN effector able to enhance plant susceptibility and modulate host immunity.






Similar content being viewed by others
Abbreviations
- DAI:
-
Days after inoculation
- HR:
-
Hypersensitive response
- J2/J3/J4:
-
Second/third/fourth stages of Meloidogyne juvenile
- MSP:
-
Meloidogyne-secreted protein
- Mg :
-
Meloidogyne graminicola
- Mi :
-
Meloidogyne incognita
- Mj :
-
Meloidogyne javanica
- PCD:
-
Programmed cell death
- RKN:
-
Root-knot nematodes
- SP:
-
Signal peptide
References
Abad P, Gouzy J, Aury JM, Castagnone-Sereno P, Danchin EG, Deleury E, Perfus-Barbeoch L et al (2008) Genome sequence of the metazoan plant-parasitic nematode Meloidogyne incognita. Nat Biotechnol 26:909–915. https://doi.org/10.1038/nbt.1482
Ali MA, Azeem F, Li H, Bohlmann H (2017) Smart parasitic nematodes use multifaceted strategies to parasitize plants. Front Plant Sci 8:1–21. https://doi.org/10.3389/fpls.2017.01699
Byrd DW Jr, Kirkpatrick T, Barker KR (1983) An improved technique for clearing and staining plant tissues for detection of nematodes. J Nematol 15:142–143
Castagnone-Sereno P, Danchin EGJ (2014) Parasitic success without sex—the nematode experience. J Evol Biol 27:1323–1333. https://doi.org/10.1111/jeb.12337
Castagnone-Sereno P, Danchin EGJ, Perfus-Barbeoch L, Abad P (2013) Diversity and evolution of root-knot nematodes, genus Meloidogyne: new insights from the genomic era. Annu Rev Phytopathol 51:203–220. https://doi.org/10.1146/annurev-phyto-082712-102300
Chen J, Lin B, Huang Q, Hu L, Zhuo K, Liao J (2017) A novel Meloidogyne graminicola effector, MgGPP, is secreted into host cells and undergoes glycosylation in concert with proteolysis to suppress plant defenses and promote parasitism. PLoS Pathog 13:e1006301. https://doi.org/10.1371/journal.ppat.1006301
Cheng X, Xiang Y, Xie H, Xu C-L, Xie T-F, Zhang C, Li Y (2013) Molecular characterization and functions of fatty acid and retinoid binding protein gene (Ab-far-1) in Aphelenchoides besseyi. PLoS One 8:e66011. https://doi.org/10.1371/journal.pone.0066011
Danchin EGJ, Arguel M-J, Campan-Fournier A, Perfus-Barbeoch L, Magliano M, Rosso M-N, Da Rocha M, Da Silva C, Nottet N, Labadie K, Guy J, Artiguenave F, Abad P (2013) Identification of novel target genes for safer and more specific control of root-knot nematodes from a pan-genome mining. PLoS Pathog 9:e1003745. https://doi.org/10.1371/journal.ppat.1003745
Daykin ME, Hussey RS (1985) Staining and histopathological techniques in nematology. In: Nasser JN, Carter CC (eds) An advanced treatise on Meloidogyne, 2nd edn. North Carolina State University, Raleigh, pp 39–48
Derevnina L, Dagdas YF, De la Concepcion JC, Bialas A, Kellner R, Petre B, Domazakis E, Du J, Wu CH, Lin X, Aguilera-Galvez C, Cruz-Mireles N, Vleeshouwers VG, Kamoun S (2016) Nine things to know about elicitins. New Phytol 212:888–895. https://doi.org/10.1111/nph.14137
Fernandez D, Petitot A-S, Grossi de Sa M, Nguyen VP, de Almeida Engler J, Kyndt T (2015) Recent advances in understanding plant–nematode interactions in monocots. In: Escobar C, Fenoll C (eds) Plant nematode interactions: a view on compatible inter-relationships, 73rd edn. Elsevier, Oxford, pp 189–219
Gasteiger E, Hoogland C, Gattiker A, Duvaud S, Wilkins MR, Appel RD, Bairoch A (2005) Protein identification and analysis tools on the ExPASy server. In: Walker JM (ed) The proteomics protocols handbook, 1st edn. Humana Press, Totowa, pp 571–607
Gilroy EM, Taylor RM, Hein I, Boevink P, Sadanandom A, Birch PRJ (2011) CMPG1-dependent cell death follows perception of diverse pathogen elicitors at the host plasma membrane and is suppressed by Phytophthora infestans RXLR effector AVR3a. New Phytol 190:653–666. https://doi.org/10.1111/j.1469-8137.2011.03643.x
Goverse A, Smant G (2014) The activation and suppression of plant innate immunity by parasitic nematodes. Annu Rev Phytopathol 52:243–265. https://doi.org/10.1146/annurev-phyto-102313-050118
Grossi-de-Sa M (2016) Root-knot nematode effectors: key actors of parasitism. Functional analysis and protein-protein interaction with host plants. Dissertation, University of Montpellier
Hewezi T, Baum TJ (2010) Sequence divergences between cyst nematode effector protein orthologs may contribute to host specificity. Plant Signal Behav 5:187–189
Hewezi T, Howe P, Maier TR, Hussey RS, Mitchum MG, Davis EL, Baum TJ (2008) Cellulose binding protein from the parasitic nematode Heterodera schachtii interacts with Arabidopsis pectin methylesterase: cooperative cell wall modification during parasitism. Plant Cell 20:3080–3093. https://doi.org/10.1105/tpc.108.063065
Hewezi T, Maier TR, Nettleton D, Baum TJ (2012) The Arabidopsis MicroRna396-grf1/grf3 regulatory module acts as a developmental regulator in the reprogramming of root cells during cyst nematode infection. Plant Physiol 159:321–335. https://doi.org/10.1104/pp.112.193649
Hewezi T, Juvale PS, Piya S, Maier TR, Rambani A, Rice JH, Mitchum MG, Davis EL, Hussey RS, Baum TJ (2015) The cyst nematode effector protein 10A07 targets and recruits host posttranslational machinery to mediate its nuclear trafficking and to promote parasitism in Arabidopsis. Plant Cell 27:891–907. https://doi.org/10.1105/tpc.114.135327
Hewitson JP, Grainger JR, Maizels RM (2009) Helminth immunoregulation: the role of parasite secreted proteins in modulating host immunity. Mol Biochem Parasitol 167:1–11. https://doi.org/10.1016/j.molbiopara.2009.04.008
Huang G, Gao B, Maier T, Allen R, Davis EL, Baum TJ, Hussey RS (2003) A profile of putative parasitism genes expressed in the esophageal gland cells of the root-knot nematode Meloidogyne incognita. Mol Plant Microbe Interact 16:376–381. https://doi.org/10.1094/MPMI.2003.16.5.376
Huang G, Dong R, Allen R, Davis EL, Baum TJ, Hussey RS (2006) A root-knot nematode secretory peptide functions as a ligand for a plant transcription factor. Mol Plant Microbe Interact 19:463–470. https://doi.org/10.1094/MPMI-19-0463
Iberkleid I, Vieira P, de Almeida Engler J, Firester K, Spiegel Y, Horowitz SB (2013) Fatty acid-and retinol-binding protein, Mj-FAR-1 induces tomato host susceptibility to root-knot nematodes. PLoS One 8:e64586. https://doi.org/10.1371/journal.pone.0064586
Jaouannet M, Magliano M, Arguel M-J, Gourgues M, Evangelisti E, Abad P, Rosso M-N (2012) The root-knot nematode calreticulin Mi-CRT is a key effector in plant defense suppression. Mol Plant Microbe Interact 26:97–105. https://doi.org/10.1094/MPMI-05-12-0130-R
Käll L, Krogh A, Sonnhammer EL (2004) A combined transmembrane topology and signal peptide prediction method. J Mol Biol 338:1027–1036. https://doi.org/10.1016/j.jmb.2004.03.016
Kamoun S, van West P, Vleeshouwers VG, de Groot KE, Govers F (1998) Resistance of Nicotiana benthamiana to Phytophthora infestans is mediated by the recognition of the elicitor protein INF1. Plant Cell 10:1413–1426. https://doi.org/10.1105/tpc.10.9.1413
Kanzaki H, Yoshida K, Saitoh H, Tamiru M, Terauchi R (2014) Protoplast cell death assay to study Magnaporthe oryzae AVR gene function in rice. In: Bird P, Jones J, Bos J (eds) Plant–pathogen interactions. Methods in molecular biology (methods and protocols), 2nd edn. Humana Press, Totowa, pp 269–275
Karssen G, Wesemael W, Moens M (2013) Root-knot nematodes. In: Perry RN, Moens M (eds) Plant nematology, 2nd edn. CAB International, Wallingford, pp 73–108
Kyndt T, Fernandez D, Gheysen G (2014) Plant-parasitic nematode infections in rice: molecular and cellular insights. Annu Rev Phytopathol 52:135–153. https://doi.org/10.1146/annurev-phyto-102313-050111
Li X-Q, Wei J-Z, Tan A, Aroian RV (2007) Resistance to root-knot nematode in tomato roots expressing a nematicidal Bacillus thuringiensis crystal protein. Plant Biotechnol J 5:455–464. https://doi.org/10.1111/j.1467-7652.2007.00257.x
Lin B, Zhuo K, Chen S, Hu L, Sun L, Wang X, Zhang L-H, Liao J (2015) A novel nematode effector suppresses plant immunity by activating host reactive oxygen species-scavenging system. New Phytol 209:1159–1173. https://doi.org/10.1111/nph.13701
Lunt DH, Kumar S, Koutsovoulos G, Blaxter ML (2014) The complex hybrid origins of the root knot nematodes revealed through comparative genomics. PeerJ 2:e356. https://doi.org/10.7717/peerj.356
Mattos VS, Mulet K, Cares JE, Gomes CB, Fernandez D, Grossi-de-Sá MF, Carneiro RMDG, Castagnone-Sereno P (2019) Development of diagnostic SCAR markers for Meloidogyne graminicola, M. oryzae, and M. salasi associated with irrigated rice fields in Americas. Plant Dis 103:83–88. https://doi.org/10.1094/PDIS-12-17-2015-RE
Nguyen P, Bellafiore S, Petitot A-S, Haidar R, Bak A, Abed A, Gantet P, Mezzalira I, de Almeida-Engler J, Fernandez D (2014) Meloidogyne incognita—rice (Oryza sativa) interaction: a new model system to study plant–root-knot nematode interactions in monocotyledons. Rice 7:23. https://doi.org/10.1186/s12284-014-0023-4
Nimchuk Z, Marois E, Kjemtrup S, Leister RT, Katagiri F, Dangl JL (2000) Eukaryotic fatty acylation drives plasma membrane targeting and enhances function of several type III effector proteins from Pseudomonas syringae. Cell 101:353–363. https://doi.org/10.1016/S0092-8674(00)80846-6
Niu J, Liu P, Liu Q, Chen C, Guo Q, Yin J, Yang G, Jian H (2016) Msp40 effector of root-knot nematode manipulates plant immunity to facilitate parasitism. Sci Rep 6:19443. https://doi.org/10.1038/srep19443
Opperman CH, Bird DM, Williamson VM, Rokhsar DS, Burke M, Cohn J, Cromer J, Diener S, Gajan J, Graham S, Houfek TD, Liu Q, Mitros T, Schaff J, Schaffer R, Scholl E, Sosinski BR, Thomas VP, Windham E (2008) Sequence and genetic map of Meloidogyne hapla: a compact nematode genome for plant parasitism. Proc Natl Acad Sci USA 105:14802–14807. https://doi.org/10.1073/pnas.0805946105
Petersen TN, Brunak S, von Heijne G, Nielsen H (2011) SignalP 4.0: discriminating signal peptides from transmembrane regions. Nat Methods 8:785–786. https://doi.org/10.1038/nmeth.1701
Petitot A-S, Dereeper A, Agbessi M, Da Silva C, Guy J, Ardisson M, Fernandez D (2016) Dual RNA-seq reveals Meloidogyne graminicola transcriptome and candidate effectors during the interaction with rice plants. Mol Plant Pathol 17:860–874. https://doi.org/10.1111/mpp.12334
Quevillon E, Silventoinen V, Pillai S, Harte N, Mulder N, Apweiler R, Lopez R (2005) InterProScan: protein domains identifier. Nucleic Acids Res 33:116–120. https://doi.org/10.1093/nar/gki442
Randig O, Bongiovanni M, Carneiro RM, Castagnone-Sereno P (2002) Genetic diversity of root-knot nematodes from Brazil and development of SCAR markers specific for the coffee-damaging species. Genome 45:862–870. https://doi.org/10.1139/g02-054
Reversat G, Boyer J, Sannier C, Pando-Bahuon A (1999) Use a mixture of sand and water-absorbent synthetic polymer as substrate for the xenic culturing of plant-parasitic nematodes in the laboratory. Nematology 1:209–212
Sallaud C, Meynard D, van Boxtel J, Gay C, Bès M, Brizard JP, Larmande P, Ortega D, Raynal M, Portefaix M, Ouwerkerk PB, Rueb S, Delseny M, Guiderdoni E (2003) Highly efficient production and characterization of T-DNA plants for rice (Oryza sativa L.) functional genomics. Theor Appl Genet 106:1396–1408. https://doi.org/10.1007/s00122-002-1184-x
Sasser JN, Krishnappa K (1980) The development of nematology on a world basis. J Nematol 12:1583–1587
Schultz J, Milpetz F, Bork P, Ponting CP (1998) SMART, a simple modular architecture research tool: identification of signaling domains. Proc Natl Acad Sci USA 95:5857–5864
Shivakumara TN, Papolu PK, Dutta TK, Kamaraju D, Chaudhary S, Rao U (2016) RNAi-induced silencing of an effector confers transcriptional oscillation in another group of effectors in the root-knot nematode, Meloidogyne incognita. Nematology 18:857–870. https://doi.org/10.1163/15685411-00003003
Shivakumara TN, Chaudhary S, Kamaraju D, Dutta TK, Papolu PK, Banakar P, Sreevathsa R, Singh B, Manjaiah KM, Rao U (2017) Host-induced silencing of two pharyngeal gland genes conferred transcriptional alteration of cell wall-modifying enzymes of Meloidogyne incognita vis-à-vis perturbed nematode infectivity in eggplant. Front Plant Sci 8:473. https://doi.org/10.3389/fpls.2017.00473
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing. http://www.R-project.org. Accessed 17 Sept 2016
Wubben MJ 2nd, Su H, Rodermel SR, Baum TJ (2001) Susceptibility to the sugar beet cyst nematode is modulated by ethylene signal transduction in Arabidopsis thaliana. Mol Plant Microbe Interact 14:1206–1212. https://doi.org/10.1094/MPMI.2001.14.10.1206
Wylie T, Martin JC, Dante M, Mitreva MD, Clifton SW, Chinwalla A, Waterson RH, Wilson RK, McCarter JP (2004) Nematode.net: a tool for navigating sequences from parasitic and free-living nematodes. Nucleic Acids Res 32:D423–D426. https://doi.org/10.1093/nar/gkh010
Xie J, Li S, Mo C, Wang G, Xiao X, Xiao Y (2016) A novel Meloidogyne incognita effector Misp12 suppresses plant defense response at latter stages of nematode parasitism. Front Plant Sci 7:964. https://doi.org/10.3389/fpls.2016.00964
Xue B, Hamamouch N, Li C, Huang G, Hussey RS, Baum TJ, Davis EL (2013) The 8D05 parasitism gene of Meloidogyne incognita is required for successful infection of host roots. Phytopathology 103:175–181. https://doi.org/10.1094/PHYTO-07-12-0173-R
Yang Y, Li R, Qi M (2000) In vivo analysis of plant promoters and transcription factors by agroinfiltration of tobacco leaves. Plant J 22:543–551. https://doi.org/10.1046/j.1365-313x.2000.00760.x
Zhang L, Davies LJ, Elling AA (2015) A Meloidogyne incognita effector is imported into the nucleus and exhibits transcriptional activation activity in planta. Mol Plant Pathol 16:48–60. https://doi.org/10.1111/mpp.12160
Zhuo K, Chen J, Lin B, Wang J, Sun F, Hu L, Liao JA (2016) A novel Meloidogyne enterolobii effector MeTCTP promotes parasitism by suppressing programmed cell death in host plants. Mol Plant Pathol 18:45–54. https://doi.org/10.1111/mpp.12374
Zhuo K, Naalden D, Nowak S, Xuan Huy N, Bauters L, Gheysen G (2018) A Meloidogyne graminicola C-type lectin, Mg01965, is secreted into the host apoplast to suppress plant defence and promote parasitism. Mol Plant Pathol 20:346–355. https://doi.org/10.1111/mpp.12759
Acknowledgements
We are grateful to Delphine Mieulet and Emmanuel Guiderdoni (CIRAD, Montpellier France) from the Rice Functional Genomics Platform (http://www.refuge-platform.org/) for assistance in rice transformation, and to Geert Smant and José Lozano-Torres (Wageningen University, Netherlands) for providing with INF1 constructs.
Funding
MGS received a Ph.D. grant, DXA received a junior post doc grant, and DF was supported by a visiting scientist grant from the Science without Boarders program (Project no. 400328/2012-7) from the Brazilian National Council for Scientific and Technological Development (CNPq, Brasil). MAB, IM and MELS received mobility grants from the Coordination for the Improvement of Higher Education Personnel (CAPES, Brazil). Part of this research was funded by Agropolis Fondation (Montpellier, France) under the reference ID “Identification of nematode (Meloidogyne spp.) effectors of pathogenicity in rice (O. sativa)” AA1002–003 and under the program “Investissement d’avenir” ANR-10-LABX-001-01, Labex Agro.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest
Data availability
Sequence data from this article can be found in the GenBank under accession numbers MK628545 and MK628546.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
425_2019_3205_MOESM2_ESM.pdf
Online Resource S2 Sources and platforms for genomic and transcriptomic analyses used for MSP18 BLAST searches (PDF 204 kb)
Online Resource S3 Meloidogyne javanica developmental stages at 21 days after infection (PDF 343 kb)
425_2019_3205_MOESM6_ESM.pdf
Online Resource S6 Root systems morphological and architectural comparison of independent transgenic rice lines (MB3-) and the wild-type Nipponbare (PDF 353 kb)
425_2019_3205_MOESM7_ESM.pdf
Online Resource S7 Meloidogyne graminicola MSP18 protein without the signal peptide (Mg-MSP18-SP) was not able to suppress PCD induced by INF1 elicitin from Phytophthora infestans in tobacco (PDF 275 kb)
Rights and permissions
About this article
Cite this article
Grossi-de-Sa, M., Petitot, AS., Xavier, D.A. et al. Rice susceptibility to root-knot nematodes is enhanced by the Meloidogyne incognita MSP18 effector gene. Planta 250, 1215–1227 (2019). https://doi.org/10.1007/s00425-019-03205-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-019-03205-3