Abstract
Main conclusion
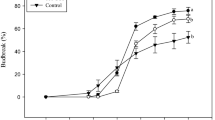
Bud-break assays under forced growth conditions suggest that a drop in ABA content and an increase in sugars are common features in the sprouting of paradormant (PD) and endodormant (ED) grapevine buds. However, increases in cell division and in respiration are unique characteristics of the ED budding.
In tropical and subtropical regions where the variations in day length and temperatures are minor throughout the year, the rupture of grapevine buds can be achieved during the current growing season given rise to a double-cropping system annually. However, it is unknown whether the breaking buds are in the paradormancy (PD) or endodormancy (ED) stage. In this study, we compared the breakage of PD and ED buds under conditions of forced growth. To do this, the expression of genes related to the metabolism of phytohormones and sugars, and of relevant physiological functions such as respiration and cell division was analyzed temporally throughout the incubation period in both types of buds. An early fall in the expression of the ABA biosynthesis gene (VvNCED1) and increases in genes related to sugar metabolism and transports were observed during the incubation period in both types of buds. However, while in the PD buds, the genes related to respiration and the cell cycle did not undergo significant changes in their expression during the incubation period, in the ED buds, the expression of these genes together with those related to auxin and cytokinin biosynthesis experienced a large increase. The results suggest that a drop in ABA content and an increase in sugars are early signals for the onset of bud break in both PD and ED vines, while the increase in respiration and cell division are unique characteristics of the ED buds, which reflect its transition from a resting state to a state of active growth.







Similar content being viewed by others
Abbreviations
- CK:
-
Cytokinin
- ED:
-
Endodormancy
- PD:
-
Paradormancy
References
Afoufa-Bastien D, Medici A, Jeauffre J, Coutos-Thévenot P, Lemoine R, Atanassova R, Laloi M (2010) The Vitis vinifera sugar transporter gene family: phylogenetic overview and macroarray expression profiling. BMC Plant Biol 10:245
Ageorges A, Issaly N, Picaud S, Delrot S, Romieu C (2000) Identification and functional expression in yeast of a grape berry sucrose carrier. Plant Physiol Biochem 38:177–185
Bai XJ, Li Y, Huang JJ, Peng HX, Xie TL (2008) One year two harvest cultural technique for Kyoho grape in southern region of Guanxi. Southwest China J Agric Sci 21:953–955
Bonhomme M, Peuch M, Ameglio T, Rageau R, Guillot A, Decourteix M (2010) Carbohydrate uptake from xylem vessels and its distribution among stem tissues and buds in walnut (Jungla regia L.). Tree Physiol 30:89–102
Boss PK, Buckeridge EJ, Poole A, Thomas MR (2003) New insights into grapevine flowering. Funct Plant Biol 30:593–606
Carmona MJ, Caib J, Martínez-Zapter JM, Thomas MR (2008) A molecular genetic perspective of reproductive development in grapevine. J Exp Bot 59:2579–2596
Chang S, Puryear J, Cairney J (1993) A simple and efficient method for isolating RNA from pine trees. Plant Mol Biol Rep 11:113–116
Chen W, Bai X, Cao M, Cheng G, Cao X, Guo R, Wang Y, He L, Yang X, He F, Duan C, Wang J (2017) Dissecting the variations of ripening progression and flavonoid metabolism in grape berries grown under double cropping system. Front Plant Sci 8:1912
Considine MJ, Considine JA (2016) On the language and physiology of dormancy and quiescence in plants. J Exp Bot 67:3189–3203
Decourteix M, Alves G, Bonhomme M, Peuch M, Baazis KB, Brunel N (2008) Sucrose (JrSUT1) and hexose (JrHT1 and JrHT2) transporter in walnut xylem parenchyma cells: their potential role in early events of growth resumption. Tree Physiol 28:215–224
Dennis FG (2003) Problems in standardizing methods for evaluating the chilling requirements for the breaking of dormancy in buds of woody plants. HortScience 38:347–350
Favero AC, Amorim D, Mota R, Soares AM, Souza CR, Regina M (2011) Double-pruning of Syrah grapevine, a management strategy to harvest wine during the winter in the Brazilian Southeast. Vitis 50:151–158
Fillion L, Ageorges A, Picaud S, Coutos-Thevenot P, Lemoine R, Romieu C, Delrot S (1999) Cloning and expression of a hexose transporter gene expressed during the ripening of grape berry. Plant Physiol 120:1083–1094
Grant TNL, Gargrave J, Dami IE (2013) Morphological, physiological, and biochemical changes in Vitis genotypes in response to photoperiod regimes. Am J Enol Vitic 64:466–475
Gu S, Jacobs S, McCarthy BS, Gohil HL (2012) Forcing vine regrowth and shifting fruit ripening in warm region to enhance fruit quality in Cabernet Sauvignon grapevine (Vitis vinifera L.). J Hortic Sci Biotechnol 87:287–292
Hayes MA, Davies C, Dry IB (2007) Isolation, functional characterization and expression analysis of grapevine (Vitis vinera L.) hexose transporters: differential roles in sink and source tissues. J Exp Bot 58:1985–1997
He D, Mathiason K, Fennell A (2012) Auxin and cytokinin related gene expression during active shoot growth and latent bud dormancy paradormancy in Vitis riparia grapevine. J Plant Physiol 169:643–648
Koussa T, Broquedis M, Bouard J (1994) Changes of abscisic acid level during the development of grape latent buds, particularly in the phase of dormancy break. Vitis 33:63–67
Kühn N, Ormeño-Nuñez J, Jaque-Zamora G, Pérez FJ (2009) Photoperiod modifies the diurnal expression profile of VvPHYA and VvPHYB transcripts in field-grown grapevine leaves. J Plant Physiol 166:1172–1180
Lang GA, Early JD, Martin GC, Darnell RL (1987) Endo-para and ecodormancy: physiological terminology and classification for dormancy research. Hort Sci 22:381–387
Lavee S, May R (1997) Dormancy of grapevine buds-facts and speculation. Aust J Grape Wine Res 3:31–46
Li-Mallet A, Rabot A, Geny L (2016) Factors controlling inflorescence primordia formation of grapevine: their role in latent bud fruitfulness? A review. Botany 94:147–163
Lin CH, Lin JH, Chang LR, Lin HS (1985) The regulation of the Golden Muscat grape production season in Taiwan. Am J Enol Vitic 36:114–117
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real time quantitative PCR and the ΔΔCT method. Methods 25:402–408
Manning K, Davies C, Bowen HC, White PJ (2001) Functional characterization of two ripening-related sucrose transporters from grape berries. Ann Bot Lond 87:125–129
Marquat C, Vandamme M, Gendraud M, Gendraud G (1999) Dormancy in vegetative buds of peach: relation between carbohydrate absorption potentials and carbohydrate concentration in the bud during dormancy and its release. Sci Hort 79:151162
Maurel K, Leite GB, Bonhomme M, Guillot A, Rageau R, Petel G (2004) Trophic control of bud break in peach. Tree Physiol 24:579–588
Noriega X, Pérez FJ (2017a) ABA biosynthesis genes are down-regulated while auxin and cytokinin biosynthesis gene are up-regulated during the release of grapevine buds from endodormancy. J Plant Growth Regul 36:814–823
Noriega X, Pérez FJ (2017b) Cell cycle genes are activated earlier than respiratory genes during release of grapevine buds from endodormancy. Plant Signal Behav 12(10):e1321189
Noriega X, Burgos B, Pérez FJ (2007) Short-day photoperiod triggers and low temperature increase expression of peroxidase RNA transcripts and basic peroxidase isoenzyme activity in grape-buds. Phytochemistry 68:1376–1383
Or E, Belausov E, Popilevsky I, Tal YB (2000) Changes in endogenous ABA level in relation to the dormancy cycle in grapevines grown in a hot climate. J Hortic Sci Biotechnol 75:190–194
Parada F, Noriega X, Dantas D, Bressan-Smith R, Pérez FJ (2016) Differences in respiration between dormant and non-dormant buds suggest the involvement of ABA in the development of endodormancy in grapevines. J Plant Physiol 201:71–78
Pérez FJ, Vergara R, Or E (2009) On the mechanism of dormancy reléase in grapevine buds a comparative study between hydrogen cyanamide and sodium azide. Plant Growth Regul 59:145–152
Rabot A, Henry C, Ben Baaziz K, Mortreau E, Azri W, Lothier J, Hamama L, Boummaza R, Leduc N, Pelleschi-Travier S, Le Gourrierec J, Sakr S (2012) Insight into the role of sugars in bud burst under light in the rose. Plant Cell Physiol 53:1068–1082
Rinne LHP, Paul KL, Vahala J, Ruonala R, Kangasjärvi J, van der Schoot C (2015) Long and short photoperiod buds in hybrid aspen share structural development and expression patterns of marker genes. J Exp Bot 66:6745–6760
Rohde A, Bhalerao RP (2007) A molecular timetable for apical bud formation and dormancy induction in poplar. Plant Cell 19:2370–2390
Rolland F, Baena-Gonzalez E, Sheen J (2006) Sugar sensing and signaling in plants: conserved and novel mechanisms. Annu Rev Plant Biol 57:675–709
Rozen S, Skaletsky H (2000) Primer3 on the www for general users and for biologist programmers. Methods Mol Biol 132:365–386
Rubio S, Donoso A, Pérez FJ (2014) The dormancy-breaking stimuli “chilling, hypoxia and cyanamide exposure” up-regulate the expression of amylase genes in grapevine buds. J Plant Physiol 171:373–381
Sakami T, Casimiro MC, Ju X, Quong AA, Katiyar S, Liu M, Jiao X, Li A, Zhang X, Lu Y (2006) Cyclin D1 determines mitochondrial function in vivo. Mol Cell Biol 26:5449–5469
Schaller EG, Street IH, Kieber JJ (2014) Cytokinin and the cell cycle. Curr Opin Plant Biol 21:7–15
Silva A, Noronha H, Dai Z, Delrot S, Gerós H (2017) Low source-sink ratio reduces reserve starch in grapevine woody canes and modulates sugar transport and metabolism at transcriptional and enzyme activity levels. Planta 246:525–535
Srinivasan C, Mullins G (1978) Control of flowering in the grapevine: formation of inflorescence in vitro by isolated tendrils. Plant Physiol 61:127–130
Vergara R, Pérez FJ (2010) Similarities between natural and chemically induced bud-endodormancy release in grapevine Vitis vinifera L. Sci Hortic 125:648–653
Vergara R, Noriega X, Aravena K, Prieto H, Pérez FJ (2017) ABA represses the expression of cell cycle genes and may modulate the development of endodormancy in grapevine buds. Front Plant Sci 8:812
Vignault C, Vachaud M, Cakir B, Glissant D, Dédaldéchamp F, Büttner M, Atanassova R, Fleurat-Lessard P, Lemoine R, Delrot S (2005) VvHT1 encodes a monosaccharide transporter expressed in the conducting complex of the grape berry floem. J Exp Bot 56:1409–1418
Wang Z, Fang M, Candas D, Zhang T, Qin L, Elridge A, Waschman-Hogiu S, Ahmed KM, Chromy BA, Nantajit D (2014) CyclinB1/cdka coordinates mitochondrial respiration for cell-cycle G2M/progression. Dev Cell 29:217–232
Zhang YL, Meng Q, Zhu HL, Guo Y, Gao HY, Luo YB, Jiang L (2008) Functional characterization of a LAHC sucrose transporter isolated from grape berries in yeast. Plant Growth Regul 54:71–79
Zheng CH, Halaly T, Acheampong AK, Takebayashi Y, Jikumaru Y, Kamiya Y, Or E (2015) Abscisic acid (ABA) regulates grape bud dormancy, and dormancy release stimuli may act through modification of ABA metabolism. J Exp Bot 66:1527–1542
Acknowledgements
The financial support of Fondecyt 1140138 and Vid-enlaze 2017 is gratefully acknowledged.
Author contribution statement
FJP designed research, analysed data and wrote the ms. XN performed the experiments and analysis the data.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pérez, F.J., Noriega, X. Sprouting of paradormant and endodormant grapevine buds under conditions of forced growth: similarities and differences. Planta 248, 837–847 (2018). https://doi.org/10.1007/s00425-018-2941-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-018-2941-7