Abstract
Main conclusion
Both G units and S units deposited in the whole lignification process of xylem fiber.
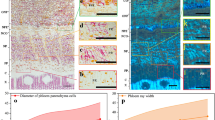
The topochemical variations in newly formed xylem and phloem of Populus × euramericana were investigated by combined microscopic techniques. During xylem formation, earlier cell wall deposition in vessel and afterwards in the neighboring fiber was observed in situ. Raman images in xylem fiber emphasized that cell wall deposition was an ordered process which lignification started in cell corner following carbohydrates deposition. Higher deposition speed of carbohydrates was revealed at the beginning of the cell wall differentiation, and the syringyl (S) units deposition was more pronounced compared with guaiacyl (G) units at the earlier stage of lignification. The comparative analysis of cell wall composition in phloem fiber indicated that phloem formed earlier than xylem and the distribution of lignin monomers varied significantly with phloem fiber location. Furthermore, an interesting phenomenon was found that the outermost phloem fiber near the periderm displayed a multilayered structure with alternating broad and narrow layer, and the broad lamellae showed higher concentration of carbohydrates and S lignin. The cytological information including cell wall composition and lignin structure of xylem and phloem might be helpful to understand the wood growth progresses and facilitate utilization of woody plants.






Similar content being viewed by others

References
Agarwal UP (2006) Raman imaging to investigate ultrastructure and composition of plant cell walls: distribution of lignin and cellulose in black spruce wood (Picea mariana). Planta 224(5):1141–1153. https://doi.org/10.1007/s00425-006-0295-z
Agarwal UP, Atalla RH (1986) In situ Raman microprobe studies of plant cell walls: macromolecular organization and compositional variability in the secondary wall of Picea mariana (Mill.) B.S.P. Planta 169(3):325–332. https://doi.org/10.1007/b00392127
Agarwal UP, Ralph SA (1997) FT-Raman spectroscopy of wood: identifying contributions of lignin and carbohydrate polymers in the spectrum of black spruce (Picea mariana). Appl Spectrosc 51(11):1648–1655. https://doi.org/10.1016/j.agrformet.2013.06.001
Agarwal UP, McSweeny JD, Ralph SA (2011) FT-Raman investigation of milled wood lignins: softwood, hardwood, and chemically modified black spruce lignins. J Wood Chem Technol 31(4):324–344. https://doi.org/10.1080/02773813.2011.562338
Akm G, Hashmi S (1983) Periodicity of cambium and the formation of xylem and phloem in Mimusops elengi L., an evergreen member of tropical India. Flora Morphol Geobot Oekophysiol 197:479–487
Barros J, Serk H, Granlund I et al (2015) The cell biology of lignification in higher plants. Ann Bot 115(7):1053–1074. https://doi.org/10.1093/aob/mcv046
Boerjan W, Ralph J, Baucher M (2003) Lignin biosynthesis. Annu Rev Plant Biol 54(1):519–546. https://doi.org/10.1146/annurev.arplant.54.031902.134938
Christiernin M (2006) Lignin composition in cambial tissues of poplar. Physiol Biochem 44:700–706. https://doi.org/10.1016/j.plaphy.2006.10.012
Čufar K, Cherubini M, Gričar J et al (2011) Xylem and phloem formation in chestnut (Castanea sativa Mill.) during the 2008 growing season. Dendrochronologia 29(3):127–134. https://doi.org/10.1016/j.dendro.2011.01.006
Cui KM, Wei LB, Li JH et al (1995) Periodicity of cambial activity and changes of starch content in Broussonetia papyrifera. Acta Bot Sin 37(1):53–57
Delgado AM, González I, Tarrés Q et al (2016) The key role of lignin in the production of low-cost lignocellulosic nanofibres for papermaking applications. Ind Crops Prod 86:295–300. https://doi.org/10.1016/j.indcrop.2016.04.010
Donaldson LA (2001) Lignification and lignin topochemistry—an ultrastructural view. Phytochemistry 57(6):859–873. https://doi.org/10.1016/s0031-9422(01)00049-8
Donaldson LA, Knox JP (2012) Localization of cell wall polysaccharides in normal and compression wood of radiata pine: relationships with lignification and microfibril orientation. Am Soc Plant Biol 158(2):642–653. https://doi.org/10.1104/pp.111.184036
Fukushima K, Terashima N (1991) Heterogeneity in formation of lignin. XIV. Formation and structure of lignin in differentiating xylem of Ginkgo biloba. Holzforschung 45:87–94. https://doi.org/10.1515/hfsg.1991.45.2.87
Gričar J, Čufar K (2008) Seasonal dynamics of phloem and xylem formation in silver fir and Norway spruce as affected by drought. Russ J Plant Physiol 55(4):538–543. https://doi.org/10.1134/s102144370804016x
Gričar J, Zupančič M, Čufar K et al (2007) Regular cambial activity and xylem and phloem formation in locally heated and cooled stem portions of Norway spruce. Wood Sci Technol 41(6):463–475. https://doi.org/10.1007/s00226-006-0109-2
Gričar J, Prislan P, Gryc V et al (2014) Plastic and locally adapted phenology in cambial seasonality and production of xylem and phloem cells in Picea abies from temperate environments. Tree Physiol 34(8):869–881. https://doi.org/10.1093/treephys/tpu026
Gričar J, Prislan P, De LM et al (2015) Plasticity in variation of xylem and phloem cell characteristics of Norway spruce under different local conditions. Front Plant Sci 6:730–743. https://doi.org/10.3389/fpls.2015.00730
Ji Z, Ma JF, Zhang ZH et al (2013) Distribution of lignin and cellulose in compression wood tracheids of Pinus yunnanensis determined by fluorescence microscopy and confocal Raman microscopy. Ind Crops Prod 47(3):212–217. https://doi.org/10.1016/j.indcrop.2013.03.006
Jin KX, Cui HS, Liu XE et al (2017) Topochemical correlation between carbohydrates and lignin on Eucommia ulmoides cell wall from tissue to cell level. BioResources 12(1):1064–1076. https://doi.org/10.15376/biores.12.1.1064-1076
Kanbayashi T, Miyafuji H (2015) Raman microscopic analysis of wood after treatment with the ionic liquid, 1-ethyl-3-methylimidazolium chloride. Holzforschung 69(3):273–279. https://doi.org/10.1515/hf-2014-0060
Korhonen JT, Kettunen M, Ras RH et al (2011) Hydrophobic nanocellulose aerogels as floating, sustainable, reusable, and recyclable oil absorbents. ACS Appl Mater Interfaces 3(6):1813–1816. https://doi.org/10.1021/am200475b
Larsen KL, Barsberg S (2010) Theoretical and Raman spectroscopic studies of phenolic lignin model monomers. J Phys Chem B 114(23):8009–8021. https://doi.org/10.1021/jp1028239
Liu XE, Jin KX, Cui HS, Ma JF (2017) The lignin topochemistry of Daemonorops margaritae (Hance) Becc. molecular spectroscopic imagine. Spectrosc Spectr Anal 37(17):3138–3144
Lourenço A, Rencoret J, Chemetova C et al (2016) Lignin composition and structure differs between xylem, phloem and phellem in Quercus suber L. Front Plant Sci 7:1612–1626. https://doi.org/10.3389/fpls.2016.01612
Lybeer B, Koch G (2005) A topochemical and semiquantitative study of the lignification during ageing of bamboo culms (Phyllostachys viridiglaucescens). IAWA J 26(1):99–109. https://doi.org/10.1163/22941932-90001605
Ma JF, Yang GH, Mao JZ et al (2011) Characterization of anatomy, ultrastructure and lignin microdistribution in Forsythia suspensa. Ind Crops Prod 33(2):358–363. https://doi.org/10.1016/j.indcrop.2010.11.009
Marion L, Gričar J, Oven P (2007) Wood formation in urban Norway maple trees studied by the micro-coring method. Dendrochronologia 25(2):97–102. https://doi.org/10.1016/j.dendro.2007.05.001
Moniz P, Lino J, Duarte LC et al (2015) Fractionation of hemicelluloses and lignin from rice straw by combining autohydrolysis and optimized mild organosolv delignification. BioResources 10(2):2626–2641. https://doi.org/10.15376/biores.10.2.2626-2641
Nguyen VP, Cho JS, Lee JH et al (2017) Identification and functional analysis of a promoter sequence for phloem tissue specific gene expression from Populus trichocarpa. J Plant Biol 60(2):129–136. https://doi.org/10.1007/s12374-016-0904-8
Parameswaran N, Liese W (1976) On the fine structure of bamboo. Wood Sci Technol 10(4):231–246. https://doi.org/10.1007/bf00350830
Parameswaran N, Liese W (1977) Structure of septate fibers in bamboo. Holzforschung 31(2):55–57. https://doi.org/10.1515/hfsg.1977.31.2.55
Payvandi S, Daly KR, Jones DL et al (2014) A mathematical model of water and nutrient transport in xylem vessels of a wheat plant. Bull Math Biol 76(3):566–596. https://doi.org/10.1007/s11538-013-9932-4
Pinto PCR, Oliveira C, Costa CA et al (2015) Kraft delignification of energy crops in view of pulp production and lignin valorization. Ind Crops Prod 71:153–162. https://doi.org/10.1016/j.indcrop.2015.03.069
Prats MB, Hauser MT, Heredia A et al (2016) Waterproofing in Arabidopsis: following phenolics and lipids in situ by confocal Raman microscopy. Front Chem 4:10–22. https://doi.org/10.3389/fchem.2016.00010
Prislan P, Koch G, Čufar K et al (2009) Topochemical investigations of cell walls in developing xylem of beech (Fagus sylvatica L.). Holzforschung 63(4):482–490. https://doi.org/10.1515/hf.2009.079
Prislan P, Gričar J, Luis MD et al (2013) Phenological variation in xylem and phloem formation in Fagus sylvatica, from two contrasting sites. Agric For Meteorol 180(8):142–151. https://doi.org/10.1016/j.agrformet.2013.06.001
Rencoret J, Gutiérrez A, Nieto L et al (2011) Lignin composition and structure in young versus adult Eucalyptus globulus plants. Plant Physiol 155:667–682. https://doi.org/10.1104/pp.110.167254
Richter S, Müssig G, Gierlinger N (2011) Functional plant cell wall design revealed by the Raman imaging approach. Planta 233(4):763–772. https://doi.org/10.1007/s00425-010-1338-z
Rossi S, Morin H, Deslauriers A (2012) Causes and correlations in cambium phenology: towards an integrated framework of xylogenesis. J Exp Bot 63(5):2117–2126. https://doi.org/10.1093/jxb/err423
Rowe N, Speck T (2005) Plant growth forms: an ecological and evolutionary perspective. N Phytol 166(1):61. https://doi.org/10.1111/j.1469-8137.2004.01309.x
Saariaho AM, Jääskeläinen AS, Nuopponen M et al (2003) Ultra violet resonance Raman spectroscopy in lignin analysis: determination of characteristic vibrations of p-hydroxyphenyl, guaiacyl, and syringyl lignin structures. Appl Spectrosc 57(1):58–66. https://doi.org/10.1366/000370203321165214
Sun L, Varanasi P, Yang F et al (2012) Rapid determination of syringyl: guaiacyl ratios using FT-Raman spectroscopy. Biotechnol Bioeng 109(3):647–656. https://doi.org/10.1002/bit.24348
Terashima N, Fukushima K (1988) Heterogeneity in formation of lignin—XI: an autoradiographic study of the heterogeneous formation and structure of pine lignin. Wood Sci Technol 22(3):259–270. https://doi.org/10.1007/bf00386021
Voxeur A, Wang Y, Sibout R (2015) Lignification: different mechanisms for a versatile polymer. Curr Opin Plant Biol 23:83–90. https://doi.org/10.1016/j.pbi.2014.11.006
Wiley JH, Atalla RH (1987) Band assignments in the Raman spectra of celluloses. Carbohydr Res 160(87):113–129. https://doi.org/10.1016/0008-6215(87)80306-3
Wu H, Xu H, Li H et al (2016) Seasonal development of cambial activity in relation to xylem formation in Chinese fir. J Plant Physiol 195:23–30. https://doi.org/10.1016/j.jplph.2015.12.013
Xi EH, Zhao GJ (2013) Seasonal development of cambium and secondary xylem in Populus × euramericana ck. ‘74/76’ during the 2010 growing season. Wood Res 58(3):425–438
Xu JK, Sun YC, Sun RC (2015) Synergistic effects of ionic liquid plus alkaline pretreatments on eucalyptus: lignin structure and cellulose hydrolysis. Process Biochem 50(6):955–965. https://doi.org/10.1016/j.procbio.2015.03.014
Yin YF, Jiang XM, Wei LB (2002) Periodicity of cambium activity and changes of peroxidase isozymes in Populus tomentosa Carr. Sci Silvae Sin 38(1):103–110
Zeng YN, Zhao S, Yang S, Ding SY (2014) Lignin plays a negative role in the biochemical process for producing lignocellulosic biofuels. Curr Opin Biotechnol 27(6):38–45. https://doi.org/10.1016/j.copbio.2013.09.008
Zhang J, Nieminen K, Serra JA et al (2014) The formation of wood and its control. Curr Opin Plant Biol 17(1):56–63. https://doi.org/10.1016/j.pbi.2013.11.003
Zhao X, Zhang L, Liu D et al (2012) Biomass recalcitrance. Part I: the chemical compositions and physical structures affecting the enzymatic hydrolysis of lignocellulose. Biofuels Bioprod Biorefin 6(4):465–482. https://doi.org/10.1002/bbb.1331
Acknowledgements
We would like to thank the National Key R&D Program of China (2017YFD0600804) and the National Natural Science Foundation of China (Nos. 31500497, 31500472) for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jin, K., Liu, X., Wang, K. et al. Imaging the dynamic deposition of cell wall polymer in xylem and phloem in Populus × euramericana. Planta 248, 849–858 (2018). https://doi.org/10.1007/s00425-018-2931-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-018-2931-9