Abstract
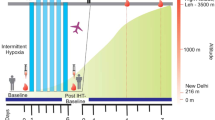
Intermittent hypoxic training (IHT) is a discrete cost-effective method for improving athletic performance and high altitude acclimatization. Unfortunately, IHT protocols widely vary in terms of hypoxia severity, duration, and number of cycles affecting physiological outcomes. In the present study, we evaluated the efficacy of a moderate normobaric IHT protocol (12% FiO2 for 4 h, 4 days) on acclimatization to high altitude (3250 m). Global plasma proteomics studies revealed that IHT elicited acute-phase response proteins like C-reactive protein (CRP), serum amyloid A-1 protein (SAA), and alpha-1-acid glycoprotein 2 (AGP 2) as well as altered levels of several apolipoproteins. On subsequent exposure to high altitude, the IH trained volunteers exhibited significant higher arterial oxygen saturation with concomitant lower incidences of acute mountain sickness (AMS) as compared to controls. Interestingly, IH trained subjects exhibited lower levels of positive acute-phase proteins like C-reactive protein (CRP), serum amyloid A-1 protein (SAA), and fibrinogen (FGA, FGB, and FGG) both after days 4 and 7 of high altitude ascent. High altitude exposure also decreased the levels of HDL, LDL, and associated proteins as well as key enzymes for assembly and maturation of lipoprotein particles like lecithin-cholesterol acyltransferase (LCAT), cholesteryl ester transfer protein (CETP), and phospholipid transfer protein (PLTP). In contrast, IHT curtailed hypoxia-induced alterations of HDL, LDL, Apo-AI, Apo-B, LCAT, CETP, and PLTP. Further validation of results also corroborated attenuation of hypoxia-induced inflammation and dyslipidemia by IHT. These results provide molecular evidences supporting the use of moderate IHT as a potential non-pharmacological strategy for high altitude acclimatization.





Similar content being viewed by others
References
Arya A, Sethy NK, Singh SK, Das M, Bhargava K (2013) Cerium oxide nanoparticles protect rodent lungs from hypobaric hypoxia-induced oxidative stress and inflammation. Int J Nanomedicine 8:4507–4520. https://doi.org/10.2147/IJN.S53032
Bartels K, Grenz A, Eltzschig HK (2013) Hypoxia and inflammation are two sides of the same coin. Proc Natl Acad Sci U S A 110:18351–18352. https://doi.org/10.1073/pnas.1318345110
Basnyat B, Murdoch DR (2003) High-altitude illness. Lancet 361:1967–1974. https://doi.org/10.1016/S0140-6736(03)13591-X
Casas M, Casas H, Pages T, Rama R, Ricart A, Ventura JL, Ibanez J, Rodriguez FA, Viscor G (2000) Intermittent hypobaric hypoxia induces altitude acclimation and improves the lactate threshold. Aviat Space Environ Med 71:125–130
Dale EA, Ben Mabrouk F, Mitchell GS (2014) Unexpected benefits of intermittent hypoxia: enhanced respiratory and nonrespiratory motor function. Physiology (Bethesda) 29:39–48. https://doi.org/10.1152/physiol.00012.2013
Eltzschig HK, Carmeliet P (2011) Hypoxia and inflammation. N Engl J Med 364:656–665. https://doi.org/10.1056/NEJMra0910283
Esteve E, Ricart W, Fernandez-Real JM (2005) Dyslipidemia and inflammation: an evolutionary conserved mechanism. Clin Nutr 24:16–31. https://doi.org/10.1016/j.clnu.2004.08.004
Gabay C, Kushner I (1999) Acute-phase proteins and other systemic responses to inflammation. N Engl J Med 340:448–454. https://doi.org/10.1056/NEJM199902113400607
Gauldie J, Richards C, Harnish D, Lansdorp P, Baumann H (1987) Interferon beta 2/B-cell stimulatory factor type 2 shares identity with monocyte-derived hepatocyte-stimulating factor and regulates the major acute phase protein response in liver cells. Proc Natl Acad Sci U S A 84:7251–7255
Grocott M, Montgomery H, Vercueil A (2007) High-altitude physiology and pathophysiology: implications and relevance for intensive care medicine. Crit Care 11:203. https://doi.org/10.1186/cc5142
Gupta N, Sahu A, Prabhakar A, Chatterjee T, Tyagi T, Kumari B, Khan N, Nair V, Bajaj N, Sharma M, Ashraf MZ (2017) Activation of NLRP3 inflammasome complex potentiates venous thrombosis in response to hypoxia. Proc Natl Acad Sci U S A 114:4763–4768. https://doi.org/10.1073/pnas.1620458114
Hamlin MJ, Lizamore CA, Hopkins WG (2018) The effect of natural or simulated altitude training on high-intensity intermittent running performance in team-sport athletes: a meta-analysis. Sports Med 48:431–446. https://doi.org/10.1007/s40279-017-0809-9
Hartmann G, Tschop M, Fischer R, Bidlingmaier C, Riepl R, Tschop K, Hautmann H, Endres S, Toepfer M (2000) High altitude increases circulating interleukin-6, interleukin-1 receptor antagonist and C-reactive protein. Cytokine 12:246–252. https://doi.org/10.1006/cyto.1999.0533
Julian CG, Subudhi AW, Wilson MJ, Dimmen AC, Pecha T, Roach RC (2011) Acute mountain sickness, inflammation, and permeability: new insights from a blood biomarker study. J Appl Physiol 111:392–399. https://doi.org/10.1152/japplphysiol.00391.2011
Jung ME, Mallet RT (2018) Intermittent hypoxia training: powerful, non-invasive cerebroprotection against ethanol withdrawal excitotoxicity. Respir Physiol Neurobiol 256:67–78. https://doi.org/10.1016/j.resp.2017.08.007
Karinen HM, Peltonen JE, Kahonen M, Tikkanen HO (2010) Prediction of acute mountain sickness by monitoring arterial oxygen saturation during ascent. High Alt Med Biol 11:325–332. https://doi.org/10.1089/ham.2009.1060
Khovidhunkit W, Kim MS, Memon RA, Shigenaga JK, Moser AH, Feingold KR, Grunfeld C (2004) Effects of infection and inflammation on lipid and lipoprotein metabolism: mechanisms and consequences to the host. J Lipid Res 45:1169–1196. https://doi.org/10.1194/jlr.R300019-JLR200
Kontush A, Chapman MJ (2006) Functionally defective high-density lipoprotein: a new therapeutic target at the crossroads of dyslipidemia, inflammation, and atherosclerosis. Pharmacol Rev 58:342–374. https://doi.org/10.1124/pr.58.3.1
Levine BD, Stray-Gundersen J (1997) Living high-training low: effect of moderate-altitude acclimatization with low-altitude training on performance. J Appl Physiol 83:102–112. https://doi.org/10.1152/jappl.1997.83.1.102
Li J, Savransky V, Nanayakkara A, Smith PL, O'Donnell CP, Polotsky VY (2007) Hyperlipidemia and lipid peroxidation are dependent on the severity of chronic intermittent hypoxia. J Appl Physiol 102:557–563. https://doi.org/10.1152/japplphysiol.01081.2006
Mahat B, Chasse E, Mauger JF, Imbeault P (2016) Effects of acute hypoxia on human adipose tissue lipoprotein lipase activity and lipolysis. J Transl Med 14:212. https://doi.org/10.1186/s12967-016-0965-y
Mallet RT, Manukhina EB, Ruelas SS, Caffrey JL, Downey HF (2018) Cardioprotection by intermittent hypoxia conditioning: evidence, mechanisms, and therapeutic potential. Am J Phys Heart Circ Phys 315:H216–H232. https://doi.org/10.1152/ajpheart.00060.2018
Mandolesi G, Avancini G, Bartesaghi M, Bernardi E, Pomidori L, Cogo A (2014) Long-term monitoring of oxygen saturation at altitude can be useful in predicting the subsequent development of moderate-to-severe acute mountain sickness. Wilderness Environ Med 25:384–391. https://doi.org/10.1016/j.wem.2014.04.015
McGarry T, Biniecka M, Veale DJ, Fearon U (2018) Hypoxia, oxidative stress and inflammation. Free Radic Biol Med 125:15–24. https://doi.org/10.1016/j.freeradbiomed.2018.03.042
Navarrete-Opazo A, Mitchell GS (2014) Therapeutic potential of intermittent hypoxia: a matter of dose. Am J Physiol Regul Integr Comp Physiol 307:R1181–R1197. https://doi.org/10.1152/ajpregu.00208.2014
Nozik-Grayck E, Woods C, Taylor JM, Benninger RK, Johnson RD, Villegas LR, Stenmark KR, Harrison DG, Majka SM, Irwin D, Farrow KN (2014) Selective depletion of vascular EC-SOD augments chronic hypoxic pulmonary hypertension. Am J Physiol Lung Cell Mol Physiol 307:L868–L876. https://doi.org/10.1152/ajplung.00096.2014
O'Brien KA, Atkinson RA, Richardson L, Koulman A, Murray AJ, Harridge SDR, Martin DS, Levett DZH, Mitchell K, Mythen MG, Montgomery HE, Grocott MPW, Griffin JL, Edwards LM (2019) Metabolomic and lipidomic plasma profile changes in human participants ascending to Everest Base camp. Sci Rep 9:2297. https://doi.org/10.1038/s41598-019-38832-z
Oliver SJ, Sanders SJ, Williams CJ, Smith ZA, Lloyd-Davies E, Roberts R, Arthur C, Hardy L, Macdonald JH (2012) Physiological and psychological illness symptoms at high altitude and their relationship with acute mountain sickness: a prospective cohort study. J Travel Med 19:210–219. https://doi.org/10.1111/j.1708-8305.2012.00609.x
Padhy G, Sethy NK, Ganju L, Bhargava K (2013) Abundance of plasma antioxidant proteins confers tolerance to acute hypobaric hypoxia exposure. High Alt Med Biol 14:289–297. https://doi.org/10.1089/ham.2012.1095
Ricart A, Casas H, Casas M, Pages T, Palacios L, Rama R, Rodriguez FA, Viscor G, Ventura JL (2000) Acclimatization near home? Early respiratory changes after short-term intermittent exposure to simulated altitude. Wilderness Environ Med 11:84–88
Richalet JP, Bittel J, Herry JP, Savourey G, Le Trong JL, Auvert JF, Janin C (1992) Use of a hypobaric chamber for pre-acclimatization before climbing Mount Everest. Int J Sports Med 13(Suppl 1):S216–S220. https://doi.org/10.1055/s-2007-1024644
Rodriguez FA, Casas H, Casas M, Pages T, Rama R, Ricart A, Ventura JL, Ibanez J, Viscor G (1999) Intermittent hypobaric hypoxia stimulates erythropoiesis and improves aerobic capacity. Med Sci Sports Exerc 31:264–268
Rodriguez FA, Ventura JL, Casas M, Casas H, Pages T, Rama R, Ricart A, Palacios L, Viscor G (2000) Erythropoietin acute reaction and haematological adaptations to short, intermittent hypobaric hypoxia. Eur J Appl Physiol 82:170–177. https://doi.org/10.1007/s004210050669
Scholz CC, Cavadas MA, Tambuwala MM, Hams E, Rodriguez J, von Kriegsheim A, Cotter P, Bruning U, Fallon PG, Cheong A, Cummins EP, Taylor CT (2013) Regulation of IL-1beta-induced NF-kappaB by hydroxylases links key hypoxic and inflammatory signaling pathways. Proc Natl Acad Sci U S A 110:18490–18495. https://doi.org/10.1073/pnas.1309718110
Semenza GL (2012) Hypoxia-inducible factors in physiology and medicine. Cell 148:399–408. https://doi.org/10.1016/j.cell.2012.01.021
Semenza GL, Wang GL (1992) A nuclear factor induced by hypoxia via de novo protein synthesis binds to the human erythropoietin gene enhancer at a site required for transcriptional activation. Mol Cell Biol 12:5447–5454
Serebrovska TV, Portnychenko AG, Drevytska TI, Portnichenko VI, Xi L, Egorov E, Gavalko AV, Naskalova S, Chizhova V, Shatylo VB (2017) Intermittent hypoxia training in prediabetes patients: beneficial effects on glucose homeostasis, hypoxia tolerance and gene expression. Exp Biol Med 242:1542–1552. https://doi.org/10.1177/1535370217723578
Siervo M, Riley HL, Fernandez BO, Leckstrom CA, Martin DS, Mitchell K, Levett DZ, Montgomery HE, Mythen MG, Grocott MP, Feelisch M, Caudwell Xtreme Everest Research G (2014) Effects of prolonged exposure to hypobaric hypoxia on oxidative stress, inflammation and gluco-insular regulation: the not-so-sweet price for good regulation. PLoS One 9:e94915. https://doi.org/10.1371/journal.pone.0094915
Silva Afonso M, Marcondes Machado R, Ferrari Lavrador MS, Carlos Rocha Quintao E, Moore KJ, Lottenberg AM (2018) Molecular pathways underlying cholesterol homeostasis. Nutrients 10. doi:https://doi.org/10.3390/nu10060760
Soria R, Egger M, Scherrer U, Bender N, Rimoldi SF (2016) Pulmonary artery pressure and arterial oxygen saturation in people living at high or low altitude: systematic review and meta-analysis. J Appl Physiol 121:1151–1159. https://doi.org/10.1152/japplphysiol.00394.2016
Tan H, Lu H, Chen Q, Tong X, Jiang W, Yan H (2018) The effects of intermittent whole-body hypoxic preconditioning on patients with carotid artery stenosis. World Neurosurg 113:e471–e479. https://doi.org/10.1016/j.wneu.2018.02.059
Taralov ZZ, Terziyski KV, Dimov PK, Marinov BI, Kostianev SS (2018) Assessment of the impact of 10-day intermittent hypoxia on the autonomic control measured by heart rate variability. Physiol Int 105:386–396. https://doi.org/10.1556/2060.105.2018.4.31
Taylor CT (2008) Interdependent roles for hypoxia inducible factor and nuclear factor-kappaB in hypoxic inflammation. J Physiol 586:4055–4059. https://doi.org/10.1113/jphysiol.2008.157669
Viscor G, Torrella JR, Corral L, Ricart A, Javierre C, Pages T, Ventura JL (2018) Physiological and biological responses to short-term intermittent hypobaric hypoxia exposure: from sports and mountain medicine to new biomedical applications. Front Physiol 9:814. https://doi.org/10.3389/fphys.2018.00814
Wang C, Jiang H, Duan J, Chen J, Wang Q, Liu X, Wang C (2018) Exploration of acute phase proteins and inflammatory cytokines in early stage diagnosis of Acute Mountain sickness. High Alt Med Biol 19:170–177. https://doi.org/10.1089/ham.2017.0126
West JB (2017) Physiological effects of chronic hypoxia. N Engl J Med 376:1965–1971. https://doi.org/10.1056/NEJMra1612008
Young T, Peppard PE, Gottlieb DJ (2002) Epidemiology of obstructive sleep apnea: a population health perspective. Am J Respir Crit Care Med 165:1217–1239
Acknowledgements
The authors acknowledge all the study participants and The Indian Army for facilitating this study. The technical help provided by Dr. Dishari Ghosh, Harish Kumar, Sanjiva Kumar, and Deepak Das is highly appreciated. AG is recipient of Senior Research Fellow from DST-INSPIRE, India, and Pooja is a recipient of Senior Research Fellowship from UGC, India.
Funding
The study was supported by research grants (Project DIP-251) from DRDO
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Experiments were conducted in accordance with the Indian Council for Medical Research (ICMR) guidelines (National ethical guidelines for biomedical and health research involving human participants) and were approved by the Defence Institute of Physiology and Allied Sciences Ethics Committee.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 4995 kb)
Rights and permissions
About this article
Cite this article
Gangwar, A., Pooja, Sharma, M. et al. Intermittent normobaric hypoxia facilitates high altitude acclimatization by curtailing hypoxia-induced inflammation and dyslipidemia. Pflugers Arch - Eur J Physiol 471, 949–959 (2019). https://doi.org/10.1007/s00424-019-02273-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-019-02273-4