Abstract
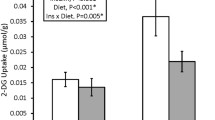
Nitric oxide (NO) is involved in skeletal muscle glucose uptake during exercise and also in the increase in insulin sensitivity after exercise. Given that neuronal nitric oxide synthase (NOS) isoform mu (nNOSμ) is a major isoform of NOS in skeletal muscle, we examined if the increase in skeletal muscle insulin-stimulated glucose uptake 3.5 h following ex vivo contraction of extensor digitorum longus (EDL) is reduced in muscles from nNOSμ+/− and nNOSμ−/− mice compared with nNOSμ+/+ mice. 3.5 h post-contraction/basal, muscles were exposed to saline or insulin (120μU/ml) with or without the presence of the NOS inhibitor NG-monomethyl-L-arginine (L-NMMA) during the last 30 min and glucose uptake was determined by radioactive tracers. Skeletal muscle insulin-stimulated glucose uptake from nNOSμ+/+, nNOSμ+/−, and nNOSμ−/− mice increased approximately twofold 3.5 h following ex vivo contraction when compared to rest. L-NMMA significantly attenuated this increase in muscle insulin-stimulated glucose uptake by around 50%, irrespective of genotype. Low levels of NOS activity were detected in muscles from nNOSμ−/− mice. In conclusion, NO mediates increases in mouse skeletal muscle insulin response following ex vivo contraction independently of nNOSμ.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Balon TW, Nadler JL (1997) Evidence that nitric oxide increases glucose transport in skeletal muscle. J Appl Physiol 82:359–363
Bradley SJ, Kingwell BA, McConell GK (1999) Nitric oxide synthase inhibition reduces leg glucose uptake but not blood flow during dynamic exercise in humans. Diabetes 48:1815–1821
Brenman JE, Chao DS, Gee SH, McGee AW, Craven SE, Santillano DR, Wu Z, Huang F, Xia H, Peters MF, Froehner SC, Bredt DS (1996) Interaction of nitric oxide synthase with the postsynaptic density protein PSD-95 and alpha1-syntrophin mediated by PDZ domains. Cell 84:757–767
Cartee GD (2014) Let’s get real about the regulation of TBC1D1 and TBC1D4 phosphorylation in skeletal muscle. J Physiol 592:253–254. https://doi.org/10.1113/jphysiol.2013.269092
Cartee GD, Young DA, Sleeper MD, Zierath J, Wallberg-Henriksson H, Holloszy JO (1989) Prolonged increase in insulin-stimulated glucose transport in muscle after exercise. Am J Phys 256:E494–E499
Castorena CM, Arias EB, Sharma N, Cartee GD (2014) Postexercise improvement in insulin-stimulated glucose uptake occurs concomitant with greater AS160 phosphorylation in muscle from normal and insulin-resistant rats. Diabetes 63:2297–2308. https://doi.org/10.2337/db13-1686
Chen S, Wasserman DH, MacKintosh C, Sakamoto K (2011) Mice with AS160/TBC1D4-Thr649Ala knockin mutation are glucose intolerant with reduced insulin sensitivity and altered GLUT4 trafficking. Cell Metab 13:68–79. https://doi.org/10.1016/j.cmet.2010.12.005
Funai K, Schweitzer GG, Castorena CM, Kanzaki M, Cartee GD (2010) In vivo exercise followed by in vitro contraction additively elevates subsequent insulin-stimulated glucose transport by rat skeletal muscle. Am J Physiol Endocrinol Metab 298:E999–E1010. https://doi.org/10.1152/ajpendo.00758.2009
Gao J, Gulve EA, Holloszy JO (1994) Contraction-induced increase in muscle insulin sensitivity: requirement for a serum factor. Am J Phys 266:E186–E192
Hakim TS, Sugimori K, Camporesi EM, Anderson G (1996) Half-life of nitric oxide in aqueous solutions with and without haemoglobin. Physiol Meas 17:267–277
Hamada T, Arias EB, Cartee GD (2006) Increased submaximal insulin-stimulated glucose uptake in mouse skeletal muscle after treadmill exercise. J Appl Physiol 101:1368–1376. https://doi.org/10.1152/japplphysiol.00416.2006
Hirschfield W, Moody MR, O'Brien WE, Gregg AR, Bryan RM Jr, Reid MB (2000) Nitric oxide release and contractile properties of skeletal muscles from mice deficient in type III NOS. Am J Physiol Regul Integr Comp Physiol 278:R95–R100
Hong YH, Frugier T, Zhang X, Murphy RM, Lynch GS, Betik AC, Rattigan S, McConell GK (2015) Glucose uptake during contraction in isolated skeletal muscles from neuronal nitric oxide synthase mu knockout mice. J Appl Physiol (1985) 118:1113–1121. https://doi.org/10.1152/japplphysiol.00056.2015
Hurt KJ, Sezen SF, Champion HC, Crone JK, Palese MA, Huang PL, Sawa A, Luo X, Musicki B, Snyder SH, Burnett AL (2006) Alternatively spliced neuronal nitric oxide synthase mediates penile erection. Proc Natl Acad Sci U S A 103:3440–3443. https://doi.org/10.1073/pnas.0511326103
Jorgensen NO, Wojtaszewski JFP, Kjobsted R (2018) Serum is not necessary for prior pharmacological activation of AMPK to increase insulin sensitivity of mouse skeletal muscle. Int J Mol Sci 19. https://doi.org/10.3390/ijms19041201
Kapur S, Bedard S, Marcotte B, Cote CH, Marette A (1997) Expression of nitric oxide synthase in skeletal muscle: a novel role for nitric oxide as a modulator of insulin action. Diabetes 46:1691–1700
Kingwell B, Formosa M, Muhlmann M, Bradley S, McConell G (2002) Nitric oxide synthase inhibition reduces glucose uptake during exercise in individuals with type 2 diabetes more than in control subjects. Diabetes 51(8):2572–2580
Kjobsted R, Munk-Hansen N, Birk JB, Foretz M, Viollet B, Bjornholm M, Zierath JR, Treebak JT, Wojtaszewski JF (2017) Enhanced muscle insulin sensitivity after contraction/exercise is mediated by AMPK. Diabetes 66:598–612. https://doi.org/10.2337/db16-0530
Kjobsted R, Treebak JT, Fentz J, Lantier L, Viollet B, Birk JB, Schjerling P, Bjornholm M, Zierath JR, Wojtaszewski JF (2015) Prior AICAR stimulation increases insulin sensitivity in mouse skeletal muscle in an AMPK-dependent manner. Diabetes 64:2042–2055. https://doi.org/10.2337/db14-1402
Lau KS, Grange RW, Isotani E, Sarelius IH, Kamm KE, Huang PL, Stull JT (2000) nNOS and eNOS modulate cGMP formation and vascular response in contracting fast-twitch skeletal muscle. Physiol Genomics 2:21–27
Lee-Young RS, Ayala JE, Hunley CF, James FD, Bracy DP, Kang L, Wasserman DH (2010) Endothelial nitric oxide synthase is central to skeletal muscle metabolic regulation and enzymatic signaling during exercise in vivo. Am J Physiol Regul Integr Comp Physiol 298:R1399–R1408. https://doi.org/10.1152/ajpregu.00004.2010
Liu X, Miller MJ, Joshi MS, Sadowska-Krowicka H, Clark DA, Lancaster JR Jr (1998) Diffusion-limited reaction of free nitric oxide with erythrocytes. J Biol Chem 273:18709–18713
McConell GK, Bradley SJ, Stephens TJ, Canny BJ, Kingwell BA, Lee-Young RS (2007) Skeletal muscle nNOS mu protein content is increased by exercise training in humans. Am J Physiol Regul Integr Comp Physiol 293:R821–R828
Merry TL, Lynch GS, McConell GK (2010) Downstream mechanisms of nitric oxide-mediated skeletal muscle glucose uptake during contraction. Am J Physiol Regul Integr Comp Physiol 299:R1656–R1665. https://doi.org/10.1152/ajpregu.00433.2010
Merry TL, Steinberg GR, Lynch GS, McConell GK (2010) Skeletal muscle glucose uptake during contraction is regulated by nitric oxide and ROS independently of AMPK. Am J Physiol Endocrinol Metab 298:E577–E585. https://doi.org/10.1152/ajpendo.00239.2009
Mikines KJ, Sonne B, Farrell PA, Tronier B, Galbo H (1988) Effect of physical exercise on sensitivity and responsiveness to insulin in humans. Am J Phys 254:E248–E259
Oki K, Arias EB, Kanzaki M, Cartee GD (2018) Prior treatment with the AMPK activator AICAR induces subsequently enhanced glucose uptake in isolated skeletal muscles from 24-month-old rats. Appl Physiol Nutr Metab 43:795–805. https://doi.org/10.1139/apnm-2017-0858
Pehmoller C, Brandt N, Birk JB, Hoeg LD, Sjoberg KA, Goodyear LJ, Kiens B, Richter EA, Wojtaszewski JF (2012) Exercise alleviates lipid-induced insulin resistance in human skeletal muscle-signaling interaction at the level of TBC1 domain family member 4. Diabetes 61:2743–2752. https://doi.org/10.2337/db11-1572
Percival JM, Anderson KN, Huang P, Adams ME, Froehner SC (2010) Golgi and sarcolemmal neuronal NOS differentially regulate contraction-induced fatigue and vasoconstriction in exercising mouse skeletal muscle. J Clin Invest 120:816–826. https://doi.org/10.1172/JCI40736
Richter EA, Garetto LP, Goodman MN, Ruderman NB (1982) Muscle glucose metabolism following exercise in the rat: increased sensitivity to insulin. J Clin Invest 69:785–793
Richter EA, Mikines KJ, Galbo H, Kiens B (1989) Effect of exercise on insulin action in human skeletal muscle. J Appl Physiol (1985) 66:876–885
Roberts CK, Barnard RJ, Scheck SH, Balon TW (1997) Exercise-stimulated glucose transport in skeletal muscle is nitric oxide dependent. Am J Phys 273:E220–E225
Ross RM, Wadley GD, Clark MG, Rattigan S, McConell GK (2007) Local nitric oxide synthase inhibition reduces skeletal muscle glucose uptake but not capillary blood flow during in situ muscle contraction in rats. Diabetes 56:2885–2892. https://doi.org/10.2337/db07-0745
Sjoberg KA, Frosig C, Kjobsted R, Sylow L, Kleinert M, Betik AC, Shaw CS, Kiens B, Wojtaszewski JFP, Rattigan S, Richter EA, McConell GK (2017) Exercise increases human skeletal muscle insulin sensitivity via coordinated increases in microvascular perfusion and molecular signaling. Diabetes 66:1501–1510. https://doi.org/10.2337/db16-1327
Wojtaszewski JF, Hansen BF, Gade, Kiens B, Markuns JF, Goodyear LJ, Richter EA (2000) Insulin signaling and insulin sensitivity after exercise in human skeletal muscle. Diabetes 49:325–331
Wojtaszewski JF, Nielsen P, Hansen BF, Richter EA, Kiens B (2000) Isoform-specific and exercise intensity-dependent activation of 5'-AMP-activated protein kinase in human skeletal muscle. J Physiol 528:221–226
Wojtaszewski JF, Richter EA (2006) Effects of acute exercise and training on insulin action and sensitivity: focus on molecular mechanisms in muscle. Essays Biochem 42:31–46. https://doi.org/10.1042/bse0420031
Zhang X, Hiam D, Hong YH, Zulli A, Hayes A, Rattigan S, McConell GK (2017) Nitric oxide is required for the insulin sensitizing effects of contraction in mouse skeletal muscle. J Physiol 595:7427–7439. https://doi.org/10.1113/JP275133
Funding
This study was funded by the National Health and Medical Research Council (NHMRC, Project grant number 1012181 to GKM) and the Diabetes Australia Research Trust (DART, Project grant number Y16G-ZHAM to XZ) in Australia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, X., Lin, X. & McConell, G.K. Normal increases in insulin-stimulated glucose uptake after ex vivo contraction in neuronal nitric oxide synthase mu (nNOSμ) knockout mice. Pflugers Arch - Eur J Physiol 471, 961–969 (2019). https://doi.org/10.1007/s00424-019-02268-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-019-02268-1