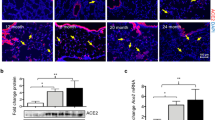
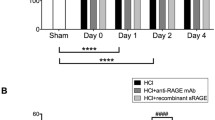
Abstract
The receptor for advanced glycation end-products is mainly expressed in type I alveolar epithelial cells but its importance in lung development and response to neonatal hyperoxia is unclear. Therefore, our study aimed at the analysis of young wildtype and RAGE knockout mice which grew up under normoxic or hyperoxic air conditions for the first 14 days followed by a longer period of normoxic conditions. Lung histology, expression of lung-specific proteins, and respiratory mechanics were analyzed when the mice reached an age of 2 or 4 months. These analyses indicated less but larger and thicker alveoli in RAGE knockout mice, reverse differences in the mRNA and protein amount of pro-surfactant proteins (pro-SP-B, pro-SP-C) and aquaporin-5, and differences in the amount of elastin and CREB, a pro-survival transcription factor, as well as higher lung compliance. Despite this potential disadvantages, RAGE knockout lungs showed less long-term damages mediated by neonatal hyperoxia. In detail, the hyperoxia-mediated reduction in alveoli, enlargement of airspaces, fragmentation of elastic fibers, and increased lung compliance combined with reduced peak airflows was less pronounced in RAGE knockout mice. In conclusion, RAGE supports the alveolarization but makes the lung more susceptible to hyperoxic injury shortly after birth. Blocking RAGE function could still be a helpful tool in reducing hyperoxia-mediated lung pathologies during alveolarization.







Similar content being viewed by others
References
Al-Robaiy S, Weber B, Simm A, Diez C, Rolewska P, Silber RE, Bartling B (2013) The receptor for advanced glycation end-products supports lung tissue biomechanics. Am J Physiol Lung Cell Mol Physiol 305:L491–L500. https://doi.org/10.1152/ajplung.00090.2013
Al-Robaiy S, Kindermann A, Wodischeck S, Simm A, Treede H, Bartling B (2018) Long-term endurance running activity causes pulmonary changes depending on the receptor for advanced glycation end-products. Pflugers Arch-Eur J Physiol 470:1543–1553. https://doi.org/10.1007/s00424-018-2175-3
Barlow CA, Kitiphongspattana K, Siddiqui N, Roe MW, Mossman BT, Lounsbury KM (2008) Protein kinase A-mediated CREB phosphorylation is an oxidant-induced survival pathway in alveolar type II cells. Apoptosis 13:681–692. https://doi.org/10.1007/s10495-008-0203-z
Bartling B, Hofmann H, Weigle B, Silber R, Simm A (2005) Down-regulation of the receptor for advanced glycation end-products (RAGE) supports non-small cell lung carcinoma. Carcinogenesis 26:293–301. https://doi.org/10.1093/carcin/bgh333
Berger J, Bhandari V (2014) Animal models of bronchopulmonary dysplasia. The term mouse models. Am J Physiol Lung Cell Mol Physiol 307:L936–L947. https://doi.org/10.1152/ajplung.00159.2014
Boggaram V (2003) Regulation of lung surfactant protein gene expression. Front Biosci 8:d751–d764
Brandes RP, Weissmann N, Schroder K (2014) Nox family NADPH oxidases: molecular mechanisms of activation. Free Radic Biol Med 76:208–226. https://doi.org/10.1016/j.freeradbiomed.2014.07.046
Carlsson LM, Jonsson J, Edlund T, Marklund SL (1995) Mice lacking extracellular superoxide dismutase are more sensitive to hyperoxia. Proc Natl Acad Sci U S A 92:6264–6268
Englert JM, Kliment CR, Ramsgaard L, Milutinovic PS, Crum L, Tobolewski JM, Oury TD (2011) Paradoxical function for the receptor for advanced glycation end products in mouse models of pulmonary fibrosis. Int J Clin Exp Pathol 4:241–254
Fehrenbach H, Kasper M, Tsherinig T, Shearman M, Schuh D, Mueller M (1998) Receptor for advanced glycation endproducts (RAGE) exhibits highly differential cellular and subcellular localisation in rat and human lung. Cell Mol Biol 44:1147–1157. https://doi.org/10.1155/2010/917108
Fineschi S, De Cunto G, Facchinetti F, Civelli M, Imbimbo BP, Carnini C, Villetti G, Lunghi B, Stochino S, Gibbons DL, Hayday A, Lungarella G, Cavarra E (2013) RAGE contributes to postnatal pulmonary development and adult lung maintenance program in mice. Am J Respir Cell Mol Biol 48:164–171. https://doi.org/10.1165/rcmb.2012-0111OC
Flodby P, Li C, Liu Y, Wang H, Rieger ME, Minoo P, Crandall ED, Ann DK, Borok Z, Zhou B (2017) Cell-specific expression of aquaporin-5 (Aqp5) in alveolar epithelium is directed by GATA6/Sp1 via histone acetylation. Sci Rep 7:3473. https://doi.org/10.1038/s41598-017-03152-7
Folz RJ, Abushamaa AM, Suliman HB (1999) Extracellular superoxide dismutase in the airways of transgenic mice reduces inflammation and attenuates lung toxicity following hyperoxia. J Clin Invest 103:1055–1066. https://doi.org/10.1172/JCI3816
Kallet RH, Matthay MA (2013) Hyperoxic acute lung injury. Respir Care 58:123–141. https://doi.org/10.4187/respcare.01963
Kierdorf K, Fritz G (2013) RAGE regulation and signaling in inflammation and beyond. J Leukoc Biol 94:55–68. https://doi.org/10.1189/jlb.1012519
Lizotte P-P, Hanford L, Enghild J, Nozik-Gayck E, Giles B-L, D Oury T (2007) Developmental expression of the receptor for advanced glycation end-products (RAGE) and its response to hyperoxia in the neonatal rat lung. BMC Dev Biol 7:15. https://doi.org/10.1186/1471-213X-7-15
Nielsen S, King LS, Christensen BM, Agre P (1997) Aquaporins in complex tissues. II. Subcellular distribution in respiratory and glandular tissues of rat. Am J Phys 273:C1549–C1561
Oczypok EA, Perkins TN, Oury TD (2017) All the "RAGE" in lung disease: the receptor for advanced glycation endproducts (RAGE) is a major mediator of pulmonary inflammatory responses. Paediatr Respir Rev 23:40–49. https://doi.org/10.1016/j.prrv.2017.03.012
O'Reilly M, Sozo F, Harding R (2013) Impact of preterm birth and bronchopulmonary dysplasia on the developing lung: long-term consequences for respiratory health. Clin Exp Pharmacol Physiol 40:765–773. https://doi.org/10.1111/1440-1681.12068
Ota C, Ishizawa K, Yamada M, Tando Y, He M, Takahashi T, Yamaya M, Yamamoto Y, Yamamoto H, Kure S, Kubo H (2016) Receptor for advanced glycation end products expressed on alveolar epithelial cells is the main target for hyperoxia-induced lung injury. Respir Investig 54:98–108. https://doi.org/10.1016/j.resinv.2015.08.009
Reynolds PR, Schmitt RE, Kasteler SD, Sturrock A, Sanders K, Bierhaus A, Nawroth PP, Paine R 3rd, Hoidal JR (2010) Receptors for advanced glycation end-products targeting protect against hyperoxia-induced lung injury in mice. Am J Respir Cell Mol Biol 42:545–551. https://doi.org/10.1165/rcmb.2008-0265OC
Reynolds PR, Kasteler SD, Schmitt RE, Hoidal JR (2011) RAGE signals through Ras during tobacco smoke-induced pulmonary inflammation. Am J Respir Cell Mol Biol 2:411–418. https://doi.org/10.1165/rcmb.2010-0231OC
Reynolds PR, Stogsdill JA, Stogsdill MP, Heimann NB (2011) Up-regulation of receptors for advanced glycation end-products by alveolar epithelium influences cytodifferentiation and causes severe lung hypoplasia. Am J Respir Cell Mol Biol 45:1195–1202. https://doi.org/10.1165/rcmb.2011-0170OC
Robinson AB, Johnson KD, Bennion BG, Reynolds PR (2012) RAGE signaling by alveolar macrophages influences tobacco smoke-induced inflammation. Am J Physiol Lung Cell Mol Physiol 302:L1192–L1199. https://doi.org/10.1152/ajplung.00099.2012
Rolewska P, Simm A, Silber RE, Bartling B (2013) Reduced expression level of the cAMP response element-binding protein contributes to lung aging. Am J Respir Cell Mol Biol 50:201–211. https://doi.org/10.1165/rcmb.2013-0057OC
Rudolph D, Tafuri A, Gass P, Hammerling GJ, Arnold B, Schutz G (1998) Impaired fetal T cell development and perinatal lethality in mice lacking the cAMP response element binding protein. Proc Natl Acad Sci U S A 95:4481–4486
Sambamurthy N, Leme AS, Oury TD, Shapiro SD (2015) The receptor for advanced glycation end products (RAGE) contributes to the progression of emphysema in mice. PLoS One 10:e0118979. https://doi.org/10.1371/journal.pone.0118979
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675
Shapiro SD, Endicott SK, Province MA, Pierce JA, Campbell EJ (1991) Marked longevity of human lung parenchymal elastic fibers deduced from prevalence of D-aspartate and nuclear weapons-related radiocarbon. J Clin Invest 87:1828–1834. https://doi.org/10.1172/JCI115204
Shirasawa M, Fujiwara N, Hirabayashi S, Ohno H, Iida J, Makita K, Hata Y (2004) Receptor for advanced glycation end-products is a marker of type I lung alveolar cells. Genes Cells 9:165–174
Su X, Looney MR, Gupta N, Matthay MA (2009) Receptor for advanced glycation end-products (RAGE) is an indicator of direct lung injury in models of experimental lung injury. Am J Physiol Lung Cell Mol Physiol 297:L1–L5. https://doi.org/10.1152/ajplung.90546.2008
Sukkar MB, Ullah MA, Gan WJ, Wark PA, Chung KF, Hughes JM, Armour CL, Phipps S (2012) RAGE: a new frontier in chronic airways disease. Br J Pharmacol 167:1161–1176. https://doi.org/10.1111/j.1476-5381.2012.01984.x
Uchida T, Shirasawa M, Ware LB, Kojima K, Hata Y, Makita K, Mednick G, Matthay ZA, Matthay MA (2006) Receptor for advanced glycation end products is a marker of type I cell injury in acute lung injury. Am J Resp Crit Care Med 173:1008–1015. https://doi.org/10.1164/rccm.200509-1477OC
Warburton D, El-Hashash A, Carraro G, Tiozzo C, Sala F, Rogers O, De Langhe S, Kemp PJ, Riccardi D, Torday J, Bellusci S, Shi W, Lubkin SR, Jesudason E (2010) Lung organogenesis. Curr Top Dev Biol 90:73–158. https://doi.org/10.1016/S0070-2153(10)90003-3
Winden DR, Ferguson NT, Bukey BR, Geyer AJ, Wright AJ, Jergensen ZR, Robinson AB, Stogsdill JA, Reynolds PR (2013) Conditional over-expression of RAGE by embryonic alveolar epithelium compromises the respiratory membrane and impairs endothelial cell differentiation. Respir Res 14:108. https://doi.org/10.1186/1465-9921-14-108
Wolf L, Herr C, Niederstrasser J, Beisswenger C, Bals R (2017) Receptor for advanced glycation endproducts (RAGE) maintains pulmonary structure and regulates the response to cigarette smoke. PLoS One 12:e0180092. https://doi.org/10.1371/journal.pone.0180092
Yamakawa N, Uchida T, Matthay MA, Makita K (2011) Proteolytic release of the receptor for advanced glycation end products from in vitro and in situ alveolar epithelial cells. Am J Physiol Lung Cell Mol Physiol 300:L516–L525. https://doi.org/10.1152/ajplung.00118.2010
Yao H, Arunachalam G, Hwang JW, Chung S, Sundar IK, Kinnula VL, Crapo JD, Rahman I (2010) Extracellular superoxide dismutase protects against pulmonary emphysema by attenuating oxidative fragmentation of ECM. Proc Natl Acad Sci U S A 107:15571–15576. https://doi.org/10.1073/pnas.1007625107
Zhang ZQ, Song YL, Chen ZH, Shen Y, Bai CX (2011) Deletion of aquaporin 5 aggravates acute lung injury induced by Pseudomonas aeruginosa. J Trauma 71:1305–1311. https://doi.org/10.1097/TA.0b013e3182128528
Acknowledgements
We thank Holger Lehnich and Dieter Barowsky (Centre of Medical Basic Research; Halle, Germany) for constructing the oxygen chamber, Hans-Jürgen Brömme (Centre of Medical Basic Research; Halle, Germany) for help with the ROS detection, and Anika Küttner for general technical assistance.
Funding
This project was supported by Wilhelm Roux program at the Medical Faculty of the Martin Luther University Halle-Wittenberg, Halle (Saale) (28/14).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kindermann, A., Baier, J., Simm, A. et al. Receptor for advanced glycation end-products modulates lung development and lung sensitivity to hyperoxic injury in newborn mice. Pflugers Arch - Eur J Physiol 471, 983–994 (2019). https://doi.org/10.1007/s00424-019-02267-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-019-02267-2