Abstract
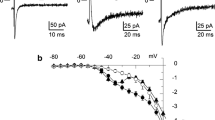
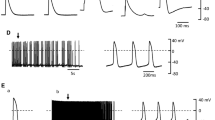
The collar of the pulmonary vein (PV) is the focal point for the initiation of atrial arrhythmias, but the mechanisms underlying how PV cells differ from neighboring left atrial tissue are unclear. We examined the biophysical and molecular properties of INa in cells isolated from the canine pulmonary sleeve and compared the properties to left atrial tissue. PV and left atrial myocytes were isolated and patch clamp techniques were used to record INa. Action potential recordings from either tissue type were made using high-resistance electrodes. mRNA was determined using quantitative RT-PCR and proteins were determined by Western blot. Analysis of the action potential characteristics showed that PV tissue had a lower Vmax compared with left atrial tissue. Fast INa showed that current density was slightly lower in PV cells compared with LA cells (−96 ± 18.7 pA/pF vs. −120 ± 6.7 pA/pF, respectively, p < 0.05). The recovery from inactivation of INa in PV cells was slightly slower but no marked difference in steady-state inactivation was noted. Analysis of late INa during a 225-ms pulse showed that late INa was significantly smaller in PV cells compared to LA cells at all measured time points into the pulse. These results suggest PV cells have lower density of both peak and late INa. Molecular analysis of Nav1.5 and the four beta subunits showed lower levels of Nav1.5 as well as Navβ1 subunits, confirming the biophysical findings. These data show that a lower density of INa may lead to depression of excitability and predispose the PV collar to re-entrant circuits under pathophysiological conditions.







Similar content being viewed by others
Reference
Antzelevitch C (2005) Role of transmural dispersion of repolarization in the genesis of drug-induced torsades de pointes. Heart Rhythm 2:S9–S15
Arora R, Verheule S, Scott L, Navarrete A, Katari V, Wilson E, Vaz D, Olgin JE (2003) Arrhythmogenic substrate of the pulmonary veins assessed by high-resolution optical mapping. Circulation 107:1816–1821
Barajas-Martinez H, Haufe V, Chamberland C, Blais Roy MJ, Fecteau MH, Cordeiro JM, Dumaine R (2009) Larger dispersion of INa in female dog ventricle as a mechanism for gender-specific incidence of cardiac arrhythmias. Cardiovasc Res 81:82–89
Biet M, Barajas-Martinez H, Ton AT, Delabre JF, Morin N, Dumaine R (2012) About half of the late sodium current in cardiac myocytes from dog ventricle is due to non-cardiac-type Na(+) channels. J Mol Cell Cardiol 53:593–598
Burashnikov A, Di Diego JM, Zygmunt AC, Belardinelli L, Antzelevitch C (2007) Atrium-selective sodium channel block as a strategy for suppression of atrial fibrillation: differences in sodium channel inactivation between atria and ventricles and the role of ranolazine. Circulation 116:1449–1457
Calloe K, Goodrow R, Olesen SP, Antzelevitch C, Cordeiro JM (2013) Tissue specific effects of acetylcholine in the canine heart. Am J Physiol Heart Circ Physiol 305:H66–H75
Calloe K, Nof E, Jespersen T, Olesen SP, Di Diego JM, Chlus N, Olesen SP, Antzelevitch C, Cordeiro JM (2011) Comparison of the effects of the transient outward potassium channel activator NS5806 on canine atrial and ventricular cardiomyocytes. J Cardiovasc Electrophysiol 22:1057–1066
Calloe K, Soltysinska E, Jespersen T, Lundby A, Antzelevitch C, Olesen SP, Cordeiro JM (2010) Differential effects of the transient outward K+ current activator NS5806 in the canine left ventricle. J Mol Cell Cardiol 48:191–200
Calvo CJ, Deo M, Zlochiver S, Millet J, Berenfeld O (2014) Attraction of rotors to the pulmonary veins in paroxysmal atrial fibrillation: a modeling study. Biophys J 106:1811–1821
Chen KH, Xu XH, Sun HY, Du XL, Liu H, Yang L, Xiao GS, Wang Y, Jin MW, Li GR (2016) Distinctive property and pharmacology of voltage-gated sodium current in rat atrial vs ventricular myocytes. Heart Rhythm 13:762–770
Cordeiro JM, Mazza M, Goodrow R, Ulahannan N, Antzelevitch C, Di Diego JM (2008) Functionally distinct sodium channels in ventricular epicardial and endocardial cells contribute to a greater sensitivity of the epicardium to electrical depression. Am J Physiol Heart Circ Physiol 295:H154–H162
Datino T, Macle L, Qi XY, Maguy A, Comtois P, Chartier D, Guerra PG, Arenal A, Fernandez-Aviles F, Nattel S (2010) Mechanisms by which adenosine restores conduction in dormant canine pulmonary veins. Circulation 121:963–972
Dumaine R, Cordeiro JM (2007) Comparison of K+ currents in cardiac Purkinje cells isolated from rabbit and dog. J Mol Cell Cardiol 42:378–389
Ehrlich JR, Cha TJ, Zhang L, Chartier D, Melnyk P, Hohnloser SH, Nattel S (2003) Cellular electrophysiology of canine pulmonary vein cardiomyocytes: action potential and ionic current properties. J Physiol 551:801–813
Eldstrom J, Wang Z, Xu H, Pourrier M, Ezrin A, Gibson K, Fedida D (2007) The molecular basis of high-affinity binding of the antiarrhythmic compound vernakalant (RSD1235) to Kv1.5 channels. Mol Pharmacol 72:1522–1534
Fedida D, Eldstrom J, Hesketh JC, Lamorgese M, Castel L, Steele DF, Van Wagoner DR (2003) Kv1.5 is an important component of repolarizing K+ current in canine atrial myocytes. Circ Res 93:744–751
Ferrier GR, Moffat MP, Lukas A (1985) Possible mechanisms of ventricular arrhythmias elicited by ischemia followed by reperfusion. Studies on isolated canine ventricular tissues. Circ Res 56:184–194
Gilmour RF Jr, Evans JJ, Zipes DP (1984) Purkinje-muscle coupling and endocardial response to hyperkalemia, hypoxia, and acidosis. Am J Phys 247:H303–H311
Gilmour RF Jr, Zipes DP (1980) Different electrophysiological responses of canine endocardium and epicardium to combined hyperkalemia, hypoxia, and acidosis. Circ Res 46:814–825
Haissaguerre M, Jais P, Shah DC, Garrigue S, Takahashi A, Lavergne T, Hocini M, Peng JT, Roudaut R, Clementy J (2000) Electrophysiological end point for catheter ablation of atrial fibrillation initiated from multiple pulmonary venous foci. Circulation 101:1409–1417
Haufe V, Cordeiro JM, Zimmer T, Wu YS, Schiccitano S, Benndorf K, Dumaine R (2005) Contribution of neuronal sodium channels to the cardiac fast sodium current I Na is greater in dog heart Purkinje fibers than in ventricles. Cardiovasc Res 65:117–127
Hutchison L, Rankin A, Drummond R, Rowan E The properties of sodium channels in rat left atrial and pulmonary vein cardiomyocytes. Proceedings of The Physiological Society 31, C07. 1–1-2014. Ref Type: Abstract
Isom LL (2001) Sodium channel b subunits: anything but auxiliary. Neuroscientist 7:42–54
Isom LL, De Jongh KS, Catterall WA (1994) Auxiliary subunits of voltage-gated ion channels. Neuron 12:1183–1194
Jais P, Hocini M, Macle L, Choi KJ, Deisenhofer I, Weerasooriya R, Shah DC, Garrigue S, Raybaud F, Scavee C, Le Metayer P, Clementy J, Haissaguerre M (2002) Distinctive electrophysiological properties of pulmonary veins in patients with atrial fibrillation. Circulation 106:2479–2485
Kaab S, Nuss HB, Chiamvimonvat N, O'Rourke B, Pak PH, Kass DA, Marban E, Tomaselli GF (1996) Ionic mechanism of action potential prolongation in ventricular myocytes from dogs with pacing-induced heart failure. Circ Res 78:262–273
Li GR, Lau CP, Shrier A (2002) Heterogeneity of sodium current in atrial vs epicardial ventricular myocytes of adult Guinea pig hearts. J Mol Cell Cardiol 34:1185–1194
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-DDCT Method. Methods 25:402–408
Maier SK, Westenbroek RE, Schenkman KA, Feigl EO, Scheuer T, Catterall WA (2002) An unexpected role for brain-type sodium channels in coupling of cell surface depolarization to contraction in the heart. Proc Natl Acad Sci U S A 99:4073–4078
Mubagwa K, Carmeliet E (1983) Effects of acetylcholine on electrophysiological properties of rabbit cardiac Purkinje fibers. Circ Res 53:740–751
Murphy L, Renodin DM, Antzelevitch C, Di Diego JM, Cordeiro JM (2011) Extracellular proton depression of peak and late sodium current in the canine left ventricle. Am J Physiol Heart Circ Physiol 301:H936–H944
Verheule S, Wilson EE, Arora R, Engle SK, Scott LR, Olgin JE (2002) Tissue structure and connexin expression of canine pulmonary veins. Cardiovasc Res 55:727–738
Yatani A, Brown AM, Akaike N (1984) Effect of extracellular pH on sodium current in isolated, single rat ventricular cells. J Membr Biol 78:163–168
Zhang Y, Hartmann HA, Satin J (1999) Glycosylation influences voltage-dependent gating of cardiac and skeletal muscle sodium channels. J Membr Biol 171:195–207
Zygmunt AC, Eddlestone GT, Thomas GP, Nesterenko VV, Antzelevitch C (2001) Larger late sodium conductance in M cells contributes to electrical heterogeneity in canine ventricle. Am J Phys 281:H689–H697
Zygmunt AC, Nesterenko VV, Rajamani S, Hu D, Barajas-Martinez H, Belardinelli L, Antzelevitch C (2011) Mechanisms of atrial-selective block of sodium channel by ranolazine I. Experimental analysis of the use-dependent block. Am J Physiol Heart Circ Physiol 301:H1606–H1614
Acknowledgements
We are grateful to Judy Hefferon for excellent technical assistance in the isolation of myocytes.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This investigation conforms to the Guide for Care and Use of Laboratory Animals published by the National Institutes of Health (The Eighth Edition of the Guide for the Care and Use of Laboratory Animals (NRC 2011)).
Funding Sources
This study was supported by the Free and Accepted Masons of New York, Florida, Massachusetts, Connecticut, Maryland, Wisconsin, Washington, and Rhode Island (to JMC).
Conflicts of Interests
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Barajas-Martinez, H., Goodrow, R.J., Hu, D. et al. Biophysical and molecular comparison of sodium current in cells isolated from canine atria and pulmonary vein. Pflugers Arch - Eur J Physiol 469, 703–712 (2017). https://doi.org/10.1007/s00424-017-1956-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-017-1956-4