Abstract
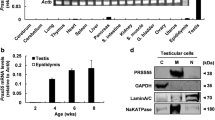
Liprin α3 was reported for the first time using sperm proteomics. Present study reports its localization on sperm and immunochemical characterization. Liprin α3 is identified as a 133 kDa protein in testis and epididymal protein extracts. In testis, immunohistochemical localization was seen in pachytenes, diplotenes, round spermatids whereas it was localized in the epithelial cells and luminal sperm in all the three regions of epididymis. Protein was localized in acrosome of rat sperm, which was further confirmed by sequential treatment of sperm with hypertonic solution. In the spermatogenic cells the protein was found to be located in developing acrosome as evident by its co-localization with Golgi marker. Protein was found to be developmentally regulated. In silico analysis of Liprin α3 revealed presence of the estrogen responsive elements upstream to initiation site and its regulation by estrogen was experimentally validated using a tamoxifen treated rat model. Western blot analysis of epididymosomes showed the presence of Liprin α3, indicating its involvement in trafficking of vesicle. The protein expression was seen in both mouse and human sperm indicating conserved nature and a probable role in acrosome reaction.










Similar content being viewed by others
References
Andrews-Zwilling YS, Kawabe H, Reim K, Varoqueaux F, Brose N (2006) Binding to Rab3A-interacting molecule RIM regulates the presynaptic recruitment of Munc13-1 and ubMunc13-2. J biol chem 281:19720–19731. doi:10.1074/jbc.M601421200
Bello OD, Zanetti MN, Mayorga LS, Michaut Ma (2012) RIM, Munc13, and Rab3A interplay in acrosomal exocytosis. Exp Cell Res 318:478–488. doi:org/10.1016/j.yexcr.2012.01.002
Bérubé B, Lefièvre L, Coutu L, Sullivan R (1996) Regulation of the epididymal synthesis of P26h, a hamster sperm protein. J Androl 17:104–110
D’Souza R, Pathak S, Upadhyay R, Gaonkar R, D’Souza S, Sonawane S, Gill-Sharma M, Balasinor NH (2009) Disruption of tubulobulbar complex by high intratesticular estrogens leading to failed spermiation. Endocrinology 150:1861–1869. doi:10.1210/en.2008-1232
Escalier D, Gallo JM, Schrével J (1997) Immunochemical characterization of a human sperm fibrous sheath protein, its developmental expression pattern, and morphogenetic relationships with actin. J histochem cytochem 45(7):909–922. doi:10.1177/002215549704500701
Girouard J, Frenette G, Sullivan R (2011) Comparative proteome and lipid profiles of bovine epididymosomes collected in the intraluminal compartment of the caput and cauda epididymidis. Int J Androl 34:e475–e486. doi:10.1111/j.1365-2605.2011.01203.x
Hecht NB (1995) The making of a spermatozoon: a molecular perspective. Dev Genet 16:95–103
Iida H, Yoshinaga Y, Tanaka S, Toshimori K, Mori T (1999) Identification of Rab3A GTPase as an acrosome-associated small GTP-binding protein in rat sperm. Dev biol 211:144–155
Katafuchi K, Mori T, Toshimori K, Iida H (2000) Localization of a syntaxin isoform, syntaxin 2, to the acrosomal region of rodent spermatozoa. Mol Reprod Dev 57:375–383. doi:10.1002/1098-2795(200012)57:4<375:AID-MRD10>3.0.CO;2-Z
Khan SA, Jadhav SV, Suryawanshi AR, Bhonde GS, Gajbhiye RK, Khole VV (2011) Evaluation of contraceptive potential of a novel epididymal sperm protein SFP2 in a mouse model. Am J Reprod Immunol 66:185–198. doi:10.1111/j.1600-0897.2011.01030.x
Ko J, Na M, Kim S, Lee J-R, Kim E (2003) Interaction of the ERC family of RIM-binding proteins with the Liprin-α family of multidomain proteins. J biol chem 278:42377–42385. doi:10.1074/jbc.M307561200
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Légaré C, Bérubé B, Boué F, Lefièvre L, Morales CR, El-Alfy M, Sullivan R (1999) Hamster sperm antigen P26h is a phosphatidylinositol-anchored protein. Mol Reprod Dev 52:225–233
Lowry Oh, Nj Rosebrough, Al Farr, Rj Randall (1951) Protein measurement with the Folin phenol reagent. J biol chem 193:265–275
MacGregor JI, Jordan VC (1998) Basic guide to the mechanisms of antiestrogen action. Pharmacol Rev 50:151–196
Maier B (2002) Developmental association of the synaptic activity-regulated protein arc with the mouse acrosomal organelle and the sperm tail. Biol Reprod 68:67–76. doi:10.1095/biolreprod.102.004143
Majó G, Lorenzo MJ, Blasi J, Aguado F (1999) Exocytotic protein components in rat pituitary gland after long-term estrogen administration. J Endocrinol 161:323–331. doi:10.1677/joe.0.1610323
Moreno RD, Palomino J, Schatten G (2006) Assembly of spermatid acrosome depends on microtubule organization during mammalian spermiogenesis. Dev Biol 293:218–227. doi:10.1016/j.ydbio.2006.02.001
Pathak S, D’Souza R, Ankolkar M, Gaonkar R, Balasinor NH (2010) Potential role of estrogen in regulation of the insulin-like growth factor2-H19 locus in the rat testis. Mol Cell Endocrinol 314:110–117. doi:10.1016/j.mce.2009.08.005
Ramalho-Santos J, Moreno RD, Sutovsky P, Chana W, Hewitson L, Wessel GM, Simerly CR, Schatten G (2000) SNAREs in mammalian sperm: possible implications for fertilization. Dev biol 223:54–69. doi:org/10.1006/dbio.2000.9745
Rezaul K, Thumar JK, Lundgren DH, Eng JK, Claffey KP, Wilson L, Han DK (2010) Differential protein expression profiles in estrogen receptor-positive and -negative breast cancer tissues using label-free quantitative proteomics. Genes cancer 1:251–271. doi:10.1177/1947601910365896
Schoch S, Castillo PE, Jo T, Mukherjee K, Geppert M, Wang Y, Schmitz F, Malenka RC, Südhof TC (2002) RIM1α forms a protein scaffold for regulating neurotransmitter release at the active zone. Nature 415:321–326. doi:10.1038/415321a
Schuel H, Burkman LJ (2005) A tale of two cells: endocannabinoid-signaling regulates functions of neurons and sperm. Biol Reprod 73:1078–1086. doi:org/10.1095/biolreprod.105.049106
Spangler SA, Hoogenraad CC (2007) Liprin-α proteins: scaffold molecules for synapse maturation. Biochem Soc Trans 35:1278–1282. doi:10.1042/BST0351278
Suryawanshi AR, Khan SA, Gajbhiye RK, Gurav MY, Khole VV (2011) Differential proteomics leads to identification of domain-specific epididymal sperm proteins. J Androl 32:240–259. doi:10.2164/jandrol.110.010967
Syntin P, Cornwall Ga (1999) Immunolocalization of CRES (cystatin-related epididymal spermatogenic) protein in the acrosomes of mouse spermatozoa. Biol Reprod 60:1542–1552. doi:10.1095/biolreprod60.6.1542
Tomes CN, Michaut M, De Blas G, Visconti P, Matti U, Mayorga LS (2002) SNARE complex assembly is required for human sperm acrosome reaction. Dev biol 243:326–338. doi:org/10.1006/dbio.2002.0567
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA 76:4350–4354
Wakle MS, Joshi SA, Khole VV (2005) Monoclonal antibody from vasectomized mouse identifies a conserved testis-specific antigen TSA70. J androl 26(6):761–771. doi:10.2164/jandrol.05045
Wang Y, Okamoto M, Schmitz F, Hofmann K, Südhof TC (1997) Rim is a putative Rab3 effector in regulating synaptic-vesicle fusion. Nature 388:593–598
WHO laboratory manual for the examination and processing of human semen (2010), 5th edn. ISBN 978 92 4 154778 9
Wright PK, May FE, Darby S, Saif R, Lennard TW, Westley BR (2009) Estrogen regulates vesicle trafficking gene expression in EFF-3, EFM-19 and MCF-7 breast cancer cells. Int J Clin Exp Pathol 2:463–475
Zhen M, Jin Y (1999) The Liprin protein SYD-2 regulates the differentiation of presynaptic termini in C. elegans. Nature 401:371–375
Zhou Y, Zheng M, Shi Q, Zhang L, Zhen W, Chen W, Zhang Y (2008) An epididymis-specific secretory protein hongres1 critically regulates sperm capacitation and male fertility. PLoS ONE 3:12. doi:10.1371/journal.pone.0004106
Zitranski N, Borth H, Ackermann F, Meyer D, Viewig L, Breit A, Gudermann T, Boekhoff I (2010) The “acrosomal synapse”: subcellular organization by lipid rafts and scaffolding proteins exhibits high similarities in neurons and mammalian spermatozoa. Comm Integr biol 3:513–521. doi:10.4161/cib.3.6.13137
Acknowledgments
The authors thank Ms. Shobha and Ms. Reshma at the confocal department of National Institute for Research in Reproductive Health for their assistance in image acquisition and processing. The authors also wish to thank Dr. Rahul Gajabhiye, clinician for providing semen samples from normal individuals. Financial support for implementation of this project and award of junior research fellowship to one of us (CJ) from the Department of Science and Technology, New Delhi is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Joshi, C.S., Suryawanshi, A.R., Khan, S.A. et al. Liprin α3: a putative estrogen regulated acrosomal protein. Histochem Cell Biol 139, 535–548 (2013). https://doi.org/10.1007/s00418-012-1044-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-012-1044-y