Abstract
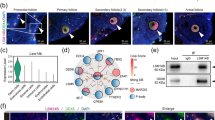
In the somatic cell nucleus, there are several universal domains such as nucleolus, SC35-domains, Cajal bodies (CBs) and histone locus bodies (HLBs). Among them, CBs were described more than 100 years ago; however, we still do not have a final understanding of their nature and biological significance. The giant nucleus of avian and amphibian growing oocytes represents an advantageous model for analysis of functions and biogenesis of various nuclear domains. Nevertheless, in large-sized avian oocytes that contain transcriptionally active lampbrush chromosomes, CB-like organelles have not been identified yet. Here we demonstrate that in the pigeon (Columba livia) oocyte nucleus, characterized by absence of any functional nucleoli, extrachromosomal spherical bodies contain TMG-capped spliceosomal snRNAs, core proteins of Sm snRNPs and the protein coilin typical for CBs, but not splicing factor SC35 nor the histone pre-mRNA 3′-end processing factor symplekin. The results establish that coilin-rich nuclear organelles in pigeon late-stage oocyte are not the equivalents of HLBs but belong to a group of CBs. At the same time, they do not contain the snoRNP/scaRNP protein fibrillarin involved in 2′-O-methylation of snoRNAs and snRNAs. Thus, the nucleus of late-stage pigeon oocytes houses CB-like organelles that have an unusual molecular composition and are implicated in the snRNP biogenesis pathway. These data demonstrate that snRNP-rich non-canonical CBs can form in the absence of nucleolus. We argue that pigeon oocytes represent a new promising model to investigate CB modular organization, functions and formation mechanism.








Similar content being viewed by others
References
Abbott J, Marzluff W, Gall JG (1999) The stem-loop binding protein (SLBP1) is present in coiled bodies of the Xenopus germinal vesicle. Mol Biol Cell 10:487–499
Andrade LEC, Chan EKL, Raska I, Peebles CL, Roos G, Tan EM (1991) Human autoantibody to a novel protein of the nuclear coiled body: immunological characterization and cDNA cloning of p80-coilin. J Exp Med 173:1407–1419
Bauer DW, Gall JG (1997) Coiled bodies without coilin. Mol Biol Cell 8:73–82
Bellini M (2000) Coilin, more than a molecular marker of the cajal (coiled) body. BioEssays 22:861–867
Bogolyubov D, Parfenov V (2008) Structure of the insect oocyte nucleus with special reference to interchromatin granule clusters and cajal bodies. Int Rev Cell Mol Biol 269:59–110
Bogolyubov D, Stepanova I, Parfenov V (2009) Universal nuclear domains of somatic and germ cells: some lessons from oocyte interchromatin granule cluster and Cajal body structure and molecular composition. BioEssays 31:400–409
Callan HG, Gall JG, Murphy C (1991) Histone genes are located at the sphere loci of Xenopus lampbrush chromosomes. Chromosoma 101:245–251
Cioce M, Lamond AI (2005) Cajal bodies: a long history of discovery. Annu Rev Cell Dev Biol 21:105–131
Deryusheva S, Gall JG (2009) Small Cajal body-specific RNAs of Drosophila function in the absence of Cajal bodies. Mol Biol Cell 20:5250–5259
Dundr M, Misteli T (2010) Biogenesis of nuclear bodies. Cold Spring Harb Perspect Biol 2:a000711
Ferreira J, Carmo-Fonseca M (1995) The biogenesis of the coiled body during early mouse development. Development 121:601–612
Frey MR, Matera AG (1995) Coiled bodies contain U7 small nuclear RNA and associated with specific DNA sequences in interphase human cells. Proc Natl Acad Sci USA 92:5915–5919
Gaginskaya ER, Gruzova MN (1969) Peculiarities of the oogenesis of Fringilla coelebs. Tsitologiia 11:1241–1251
Gaginskaya ER, Gruzova MN (1975) Detection of the amplified rDNA in ovarial cells of some insects and birds by hybridization in situ. Tsitologiia 17:1132–1137
Gaginskaya E, Kulikova T, Krasikova A (2009) Avian lampbrush chromosomes: a powerful tool for exploration of genome expression. Cytogenet Genome Res 124:251–267
Galardi S, Fatica A, Bachi A, Scaloni A, Presutti C, Bozzoni I (2002) Purified box C/D snoRNPs are able to reproduce site-specific 2′-O-methylation of target RNA in vitro. Mol Cell Biol 22:6663–6668
Gall JG (2000) Cajal bodies: the first 100 years. Annu Rev Cell Dev Biol 16:273–300
Gall JG, Stephenson EC, Erba HP, Diaz MO, Barsacchi-Pilone G (1981) Histone genes are located at the sphere loci of newt lampbrush chromosomes. Chromosoma 84:159–171
Gall JG, Bellini M, Wu Z, Murphy C (1999) Assembly of the nuclear transcription and processing machinery: Cajal bodies (coiled bodies) and transcriptosomes. Mol Biol Cell 10:4385–4402
Gall JG, Wu Z, Murphy C, Gao H (2004) Structure in the amphibian germinal vesicle. Exp Cell Res 296:28–34
Gilder A, Hebert M (2011) Relationship of the Cajal Body to the Nucleolus. Nucleolus Protein Rev 15:361–380
Greenfield ML (1966) The oocyte of the domestic chicken shortly after hatching, studied by electron microscopy. J Embryol Exp Morphol 15:297–316
Hofmann I, Schnolzer M, Kaufmann I, Franke WW (2002) Symplekin, a constitutive protein of karyo- and cytoplasmic particles involved in mRNA biogenesis in Xenopus laevis oocytes. Mol Biol Cell 13:1665–1676
Kaiser TE, Intine RV, Dundr M (2008) De novo formation of a subnuclear body. Science 322:1713–1717
Khutinaeva MA, Kropotova EV, Gaginskaya ER (1989) Morpho-functional organization peculiarities of the lampbrush chromosomes of oocytes in the pigeon Columba livia. Tsitologiia 10:1185–1192
Kiss AM, Jady BE, Darzacq X, Verheggen C, Bertrand E, Kiss T (2002) A Cajal body-specifc pseudouridylation guide RNA is composed of two box H/ACA snoRNA-like domains. Nucleic Acids Res 30:4643–4649
Kolev NG, Steitz JA (2005) Symplekin and multiple other polyadenylation factors participate in 3′-end maturation of histone mRNAs. Genes Dev 19:2583–2592
Krasikova AV (2007) Centromere regions of chromosomes and associated structures in nuclei of avian growing oocytes. Dissertation, Saint-Petersburg State University
Krasikova A, Kulikova T, Saifitdinova A, Derjusheva S, Gaginskaya E (2004) Centromeric protein bodies on avian lampbrush chromosomes contain a protein detectable with an antibody against DNA topoisomerase II. Chromosoma 113:316–323
Krasikova A, Barbero JL, Gaginskaya E (2005) Cohesion proteins are present in centromere protein bodies associated with avian lampbrush chromosomes. Chromosom Res 13:675–685
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lafarga M, Casafont I, Bengoechea R, Tapia O, Berciano MT (2009) Cajal’s contribution to the knowledge of the neuronal cell nucleus. Chromosoma 118:437–443
Lemm I, Girard C, Kuhn AN, Watkins NJ, Schneider M, Bordonné R, Lührmann R (2006) Ongoing U snRNP biogenesis is required for the integrity of Cajal bodies. Mol Biol Cell 17:3221–3231
Lerner EA, Lerner MR, Janeway CA, Steitz J (1981) Monoclonal antibodies to nucleic acid-containing cellular constituents: probes for molecular biology and autoimmune disease. Proc Natl Acad Sci USA 78:2737–2741
Liu Q, Dreyfuss G (1996) A novel nuclear structure containing the survival of motor neurons protein. EMBO J 15:3555–3565
Liu J-L, Buszczak M, Gall JG (2006) Nuclear bodies in the Drosophila germinal vesicle. Chromosom Res 14:465–475
Liu J-L, Wu Z, Nizami Z, Deryusheva S, Rajendra TK, Beumer KJ, Gao H, Matera AG, Carroll D, Gall JG (2009) Coilin is essential for Cajal body organization in Drosophila melanogaster. Mol Biol Cell 20:1661–1670
Marz M, Mosig A, Stadler BM, Stadler PF (2007) U7 snRNAs: a computational survey. Genomics Proteomics Bioinforma 5:187–195
Maslova AV, Krasikova AV (2011) Spatial arrangement of macro-, midi-, and microchromosomes in transcriptionally active nuclei of growing oocytes in birds of order galliformes. Cell Tissue Biol 5:281–293
Matera AG (2006) Drosophila Cajal bodies: accessories not included. J Cell Biol 172:791–793
Matera AG, Shpargel KB (2006) Pumping RNA: nuclear bodybuilding along the RNP pipeline. Curr Opin Cell Biol 18:317–324
Millevoi S, Vagner S (2010) Molecular mechanisms of eukaryotic pre-mRNA 3′ end processing regulation. Nucleic Acids Res 38:2757–2774
Mintz PJ, Patterson SD, Neuwald AF, Spahr CS, Spector DL (1999) Purification and biochemical characterization of interchromatin granule clusters. EMBO J 18:4308–4320
Morgan GT (2002) Lampbrush chromosomes and associated bodies: new insights into principles of nuclear structure and function. Chromosome Res 10:177–200
Nizami ZF, Deryusheva S, Gall JG (2010a) The Cajal body and histone locus body. Cold Spring Harb Perspect Biol. doi:10.1101/cshperspect.a000653
Nizami ZF, Deryusheva S, Gall JG (2010b) Cajal Bodies and Histone Locus Bodies in Drosophila and Xenopus. Cold Spring Harb Symp Quant Biol 75:313–320
Ochs RL, Stein TW Jr, Andrade LEC, Gallo D, Chan EKL, Tan EM, Brasch K (1995) Formation of nuclear bodies in hepatocytes of estrogen-treated roosters. Mol Biol Cell 6:345–356
Pollard KM, Lee DK, Casiano CA, Bluthner M, Johnston MM, Tan EM (1997) The autoimmunity-inducing xenobiotic mercury interacts with the autoantigen fibrillarin and modifies its molecular and antigenic properties. J Immunol 158:3521–3528
Pontes O, Pikaard CS (2008) siRNA and miRNA processing: new functions for Cajal bodies. Curr Opin Genet Dev 18:197–203
Reynolds ES (1963) The use of lead citrate at high pH as an electron-opaque stain in electron microscopy. J Cell Biol 17:208–212
Saifitdinova A, Derjusheva S, Krasikova A, Gaginskaya E (2003) Lampbrush chromosomes of the chaffinch (Fringilla coelebs L.). Chromosom Res 11:99–113
Schmidt-Zachmann M, Hügle B, Scheer U, Franke WW (1984) Identification and localization of a novel nucleolar protein of high molecular weight by a monoclonal antibody. Exp Cell Res 153:327–346
Schmidt-Zachmann MS, Hügle-Dörr B, Franke WW (1987) A constitutive nucleolar protein identified as a member of the nucleoplasmin family. EMBO J 6:1881–1890
Shaw DJ, Eggleton P, Young PJ (2008) Joining the dots: production, processing and targeting of U snRNP to nuclear bodies. Biochim Biophys Acta 1783:2137–2144
Solovei I, Gaginskaya E, Hutchison N, Macgregor H (1993) Avian sex chromosomes in the lampbrush form: the ZW lampbrush bivalents from six species of bird. Chromosom Res 1:153–166
Solovei IV, Joffe BI, Gaginskaya ER, Macgregor HC (1996) Transcription on lampbrush chromosomes of a centromerically localized highly repeated DNA in pigeon (Columba) relates to sequence arrangement. Chromosom Res 4:588–603
Sullivan KD, Steiniger M, Marzluff WF (2009) A core complex of CPSF73, CPSF100, and Symplekin may form two different cleavage factors for processing of poly(A) and histone mRNAs. Mol Cell 34:322–332
Tucker TE, Berciano MT, Jacobs EY, LePage DF, Shpargel KB, Rossire JJ, Chan EK, Lafarga M, Conlon RA, Matera AG (2001) Residual Cajal bodies in coilin knockout mice fail to recruit Sm snRNPs and SMN, the spinal muscular atrophy gene product. J Cell Biol 154:293–307
Wu CH, Gall JG (1993) U7 small nuclear RNA in C snurposomes of the Xenopus germinal vesicle. Proc Natl Acad Sci 90:6257–6259
Wu Z, Murphy C, Gall JG (1994) Human p80-coilin is targeted to sphere organelles in the amphibian germinal vesicle. Mol Biol Cell 5:1119–1127
Xie J, Zhang M, Zhou T, Hua X, Tang L, Wu W (2007) Sno/scaRNAbase: a curated database for small nucleolar and Cajal body-specific RNAs. Nucleic Acids Res 35:183–187
Acknowledgments
We are grateful to J.G. Gall (Carnegie Institution for Science, Department of Embryology, Baltimore, USA) for mAb against symplekin (BD Transduction Laboratories). The authors thank anonymous reviewers for helpful comments and suggestions. This investigation was supported by research Grant of the President of Russian Federation (project # 3299.2010.4) and Federal Grant-in-Aid Program "Human Capital for Science and Education in Innovative Russia" (Governmental Contracts ## P1367 and 14.740.11.1189). The authors acknowledge Saint-Petersburg State University for a research Grant No. 1.40.703.2011 and the Core Facility ‘CHROMAS’ (Saint-Petersburg State University) for technical support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Video a in Online Resource 2 (MOV 2340 kb)
Video c in Online Resource 2 (MOV 1147 kb)
Rights and permissions
About this article
Cite this article
Khodyuchenko, T., Gaginskaya, E. & Krasikova, A. Non-canonical Cajal bodies form in the nucleus of late stage avian oocytes lacking functional nucleolus. Histochem Cell Biol 138, 57–73 (2012). https://doi.org/10.1007/s00418-012-0938-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-012-0938-z