Abstract
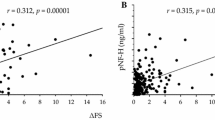
Neurofilament light chain protein (NfL) is currently the most accurate cerebrospinal fluid (CSF) biomarker in amyotrophic lateral sclerosis (ALS) in terms of both diagnostic and prognostic value, but the mechanism underlying its increase is still a matter of debate. Similarly, emerging CSF biomarkers of neurodegeneration and neuroinflammation showed promising results, although further studies are needed to clarify their clinical and pathophysiological roles. In the present study we compared the diagnostic accuracy of CSF NfL, phosphorylated (p)-tau/total (t)-tau ratio, chitinase-3-like protein 1 (YKL-40) and chitotriosidase 1 (CHIT1), in healthy controls (n = 43) and subjects with ALS (n = 80) or ALS mimics (n = 46). In ALS cases, we also investigated the association between biomarker levels and clinical variables, the extent of upper motor neuron (UMN) and lower motor neuron (LMN) degeneration, and denervation activity through electromyography (EMG). ALS patients showed higher levels of CSF NfL, YKL-40, CHIT1, and lower values of p-tau/t-tau ratio compared to both controls and ALS mimics. Among all biomarkers, NfL yielded the highest diagnostic performance (> 90% sensitivity and specificity) and was the best predictor of disease progression rate and survival in ALS. NfL levels showed a significant correlation with the extent of LMN involvement, whereas YKL-40 levels increased together with the number of areas showing both UMN and LMN damage. EMG denervation activity did not correlate with any CSF biomarker change. These findings confirm the highest value of NfL among currently available CSF biomarkers for the diagnostic and prognostic assessment of ALS and contribute to the understanding of the pathophysiological and electrophysiological correlates of biomarker changes.


Similar content being viewed by others
References
Brooks BR, Miller RG, Swash M, Munsat TL, World Federation of Neurology Research Group on Motor Neuron Diseases (2000) El Escorial revisited: revised criteria for the diagnosis of amyotrophic lateral sclerosis. Amyotroph Lateral Scler Other Motor Neuron Disord 1(5):293–299
de Carvalho M, Dengler R, Eisen A, England JD, Kaji R, Kimura J, Mills K, Mitsumoto H, Nodera H, Shefner J, Swash M (2008) Electrodiagnostic criteria for diagnosis of ALS. Clin Neurophysiol 119(3):497–503. https://doi.org/10.1016/j.clinph.2007.09.143
Gille B, De Schaepdryver M, Dedeene L, Goossens J, Claeys KG, Van Den Bosch L, Tournoy J, Van Damme P, Poesen K (2019) Inflammatory markers in cerebrospinal fluid: independent prognostic biomarkers in amyotrophic lateral sclerosis? J Neurol Neurosurg Psychiatry 90(12):1338–1346. https://doi.org/10.1136/jnnp-2018-319586
Steinacker P, Feneberg E, Weishaupt J, Brettschneider J, Tumani H, Andersen PM, von Arnim CA, Böhm S, Kassubek J, Kubisch C, Lulé D, Müller HP, Muche R, Pinkhardt E, Oeckl P, Rosenbohm A, Anderl-Straub S, Volk AE, Weydt P, Ludolph AC, Otto M (2016) Neurofilaments in the diagnosis of motoneuron diseases: a prospective study on 455 patients. J Neurol Neurosurg Psychiatry 87(1):12–20. https://doi.org/10.1136/jnnp-2015-311387
Poesen K, De Schaepdryver M, Stubendorff B, Gille B, Muckova P, Wendler S, Prell T, Ringer TM, Rhode H, Stevens O, Claeys KG, Couwelier G, D'Hondt A, Lamaire N, Tilkin P, Van Reijen D, Gourmaud S, Fedtke N, Heiling B, Rumpel M, Rödiger A, Gunkel A, Witte OW, Paquet C, Vandenberghe R, Grosskreutz J, Van Damme P (2017) Neurofilament markers for ALS correlate with extent of upper and lower motor neuron disease. Neurology 88(24):2302–2309. https://doi.org/10.1212/WNL.0000000000004029
Feneberg E, Oeckl P, Steinacker P, Verde F, Barro C, Van Damme P, Gray E, Grosskreutz J, Jardel C, Kuhle J, Koerner S, Lamari F, Amador MDM, Mayer B, Morelli C, Muckova P, Petri S, Poesen K, Raaphorst J, Salachas F, Silani V, Stubendorff B, Turner MR, Verbeek MM, Weishaupt JH, Weydt P, Ludolph AC, Otto M (2018) Multicenter evaluation of neurofilaments in early symptom onset amyotrophic lateral sclerosis. Neurology 90(1):e22–e30. https://doi.org/10.1212/WNL.0000000000004761
Illán-Gala I, Alcolea D, Montal V, Dols-Icardo O, Muñoz L, de Luna N, Turón-Sans J, Cortés-Vicente E, Sánchez-Saudinós MB, Subirana A, Sala I, Blesa R, Clarimón J, Fortea J, Rojas-García R, Lleó A (2018) CSF sAPPβ, YKL-40, and NfL along the ALS-FTD spectrum. Neurology 91(17):e1619–e1628. https://doi.org/10.1212/WNL.0000000000006383
Schreiber S, Spotorno N, Schreiber F, Acosta-Cabronero J, Kaufmann J, Machts J, Debska-Vielhaber G, Garz C, Bittner D, Hensiek N, Dengler R, Petri S, Nestor PJ, Vielhaber S (2018) Significance of CSF NfL and tau in ALS. J Neurol 265(11):2633–2645. https://doi.org/10.1007/s00415-018-9043-0
Gille B, De Schaepdryver M, Goossens J, Dedeene L, De Vocht J, Oldoni E, Goris A, Van Den Bosch L, Depreitere B, Claeys KG, Tournoy J, Van Damme P, Poesen K (2019) Serum neurofilament light chain levels as a marker of upper motor neuron degeneration in patients with amyotrophic lateral sclerosis. Neuropathol Appl Neurobiol 45(3):291–304. https://doi.org/10.1111/nan.12511
Menke RA, Gray E, Lu CH, Kuhle J, Talbot K, Malaspina A, Turner MR (2015) CSF neurofilament light chain reflects corticospinal tract degeneration in ALS. Ann Clin Transl Neurol 2(7):748–755. https://doi.org/10.1002/acn3.212
Meeter LHH, Vijverberg EG, Del Campo M, Rozemuller AJM, Kaat LD, de Jong FJ, van der Flier WM, Teunissen CE, van Swieten JC, Pijnenburg YAL (2018) Clinical value of neurofilament and phospho-tau/tau ratio in the frontotemporal dementia spectrum. Neurology 90(14):e1231–e1239. https://doi.org/10.1212/WNL.0000000000005261
Abu-Rumeileh S, Mometto N, Bartoletti-Stella A, Polischi B, Oppi F, Poda R, Stanzani-Maserati M, Cortelli P, Liguori R, Capellari S, Parchi P (2018) Cerebrospinal fluid biomarkers in patients with frontotemporal dementia spectrum: a single-center study. J Alzheimers Dis 66(2):551–563. https://doi.org/10.3233/JAD-180409
Abu-Rumeileh S, Steinacker P, Polischi B, Mammana A, Bartoletti-Stella A, Oeckl P, Baiardi S, Zenesini C, Huss A, Cortelli P, Capellari S, Otto M, Parchi P (2019) CSF biomarkers of neuroinflammation in distinct forms and subtypes of neurodegenerative dementia. Alzheimers Res Ther 12(1):2. https://doi.org/10.1186/s13195-019-0562-4
Grossman M, Elman L, McCluskey L, McMillan CT, Boller A, Powers J, Rascovsky K, Hu W, Shaw L, Irwin DJ, Lee VM, Trojanowski JQ (2014) Phosphorylated tau as a candidate biomarker for amyotrophic lateral sclerosis. JAMA Neurol 71(4):442–448. https://doi.org/10.1001/jamaneurol.2013.6064
Wilke C, Deuschle C, Rattay TW, Maetzler W, Synofzik M (2015) Total tau is increased, but phosphorylated tau not decreased, in cerebrospinal fluid in amyotrophic lateral sclerosis. Neurobiol Aging 36(2):1072–1074. https://doi.org/10.1016/j.neurobiolaging.2014.10.019
Steinacker P, Verde F, Fang L, Feneberg E, Oeckl P, Roeber S, Anderl-Straub S, Danek A, Diehl-Schmid J, Fassbender K, Fliessbach K, Foerstl H, Giese A, Jahn H, Kassubek J, Kornhuber J, Landwehrmeyer GB, Lauer M, Pinkhardt EH, Prudlo J, Rosenbohm A, Schneider A, Schroeter ML, Tumani H, von Arnim CAF, Weishaupt J, Weydt P, Ludolph AC, Yilmazer Hanke D, Otto M, FTLDC study group (2018) Chitotriosidase (CHIT1) is increased in microglia and macrophages in spinal cord of amyotrophic lateral sclerosis and cerebrospinal fluid levels correlate with disease severity and progression. J Neurol Neurosurg Psychiatry 89(3):239–247. https://doi.org/10.1136/jnnp-2017-317138
Oeckl P, Weydt P, Steinacker P, Anderl-Straub S, Nordin F, Volk AE, Diehl-Schmid J, Andersen PM, Kornhuber J, Danek A, Fassbender K, Fliessbach K, Jahn H, German Consortium for Frontotemporal Lobar Degeneration, Lauer M, Müller K, Knehr A, Prudlo J, Schneider A, Thal DR, Yilmazer-Hanke D, Weishaupt JH, Ludolph AC, Otto M (2019) Different neuroinflammatory profile in amyotrophic lateral sclerosis and frontotemporal dementia is linked to the clinical phase. J Neurol Neurosurg Psychiatry 90(1):4–10. https://doi.org/10.1136/jnnp-2018-318868
Oeckl P, Weydt P, Thal DR, Weishaupt JH, Ludolph AC, Otto M (2019) Proteomics in cerebrospinal fluid and spinal cord suggests UCHL1, MAP2 and GPNMB as biomarkers and underpins importance of transcriptional pathways in amyotrophic lateral sclerosis. Acta Neuropathol. https://doi.org/10.1007/s00401-019-02093-x
Thompson AG, Gray E, Bampton A, Raciborska D, Talbot K, Turner MR (2019) CSF chitinase proteins in amyotrophic lateral sclerosis. J Neurol Neurosurg Psychiatry 90(11):1215–1220. https://doi.org/10.1136/jnnp-2019-320442
Swinnen B, Robberecht W (2014) The phenotypic variability of amyotrophic lateral sclerosis. Nat Rev Neurol 10(11):661–670. https://doi.org/10.1038/nrneurol.2014.184
Krarup C (2011) Lower motor neuron involvement examined by quantitative electromyography in amyotrophic lateral sclerosis. Clin Neurophysiol 122(2):414–422. https://doi.org/10.1016/j.clinph.2010.06.027
Sato Y, Nakatani E, Watanabe Y, Fukushima M, Nakashima K, Kannagi M, Kanatani Y, Mizushima H (2015) Prediction of prognosis of ALS: Importance of active denervation findings of the cervical-upper limb area and trunk area. Intractable Rare Dis Res 4(4):181–189. https://doi.org/10.5582/irdr.2015.01043
Abu-Rumeileh S, Giannini G, Polischi B, Albini-Riccioli L, Milletti D, Oppi F, Stanzani-Maserati M, Capellari S, Mantovani P, Palandri G, Cortelli P, Cevoli S, Parchi P (2019) Revisiting the cerebrospinal fluid biomarker profile in idiopathic normal pressure hydrocephalus: the Bologna Pro-Hydro Study. J Alzheimers Dis 68(2):723–733. https://doi.org/10.3233/JAD-181012
Westeneng HJ, Debray TPA, Visser AE, van Eijk RPA, Rooney JPK, Calvo A, Martin S, McDermott CJ, Thompson AG, Pinto S, Kobeleva X, Rosenbohm A, Stubendorff B, Sommer H, Middelkoop BM, Dekker AM, van Vugt JJFA, van Rheenen W, Vajda A, Heverin M, Kazoka M, Hollinger H, Gromicho M, Körner S, Ringer TM, Rödiger A, Gunkel A, Shaw CE, Bredenoord AL, van Es MA, Corcia P, Couratier P, Weber M, Grosskreutz J, Ludolph AC, Petri S, de Carvalho M, Van Damme P, Talbot K, Turner MR, Shaw PJ, Al-Chalabi A, Chiò A, Hardiman O, Moons KGM, Veldink JH, van den Berg LH (2018) Prognosis for patients with amyotrophic lateral sclerosis: development and validation of a personalised prediction model. Lancet Neurol 17(5):423–433. https://doi.org/10.1016/S1474-4422(18)30089-9
Beers DR, Appel SH (2019) Immune dysregulation in amyotrophic lateral sclerosis: mechanisms and emerging therapies. Lancet Neurol 18(2):211–220. https://doi.org/10.1016/S1474-4422(18)30394-6
Thompson AG, Gray E, Thézénas ML, Charles PD, Evetts S, Hu MT, Talbot K, Fischer R, Kessler BM, Turner MR (2018) Cerebrospinal fluid macrophage biomarkers in amyotrophic lateral sclerosis. Ann Neurol 83(2):258–268. https://doi.org/10.1002/ana.25143
Olsson B, Alberg L, Cullen NC, Michael E, Wahlgren L, Kroksmark AK, Rostasy K, Blennow K, Zetterberg H, Tulinius M (2019) NFL is a marker of treatment response in children with SMA treated with nusinersen. J Neurol 266(9):2129–2136. https://doi.org/10.1007/s00415-019-09389-8
Wilke C, Rattay TW, Hengel H, Zimmermann M, Brockmann K, Schöls L, Kuhle J, Schüle R, Synofzik M (2018) Serum neurofilament light chain is increased in hereditary spastic paraplegias. Ann Clin Transl Neurol 5(7):876–882. https://doi.org/10.1002/acn3.583
Wurster CD, Steinacker P, Günther R, Koch JC, Lingor P, Uzelac Z, Witzel S, Wollinsky K, Winter B, Osmanovic A, Schreiber-Katz O, Al Shweiki R, Ludolph AC, Petri S, Hermann A, Otto M, MND-Net (2019) Neurofilament light chain in serum of adolescent and adult SMA patients under treatment with nusinersen. J Neurol. https://doi.org/10.1007/s00415-019-09547-y
Jacobsen AB, Kristensen RS, Witt A, Kristensen AG, Duez L, Beniczky S, Fuglsang-Frederiksen A, Tankisi H (2018) The utility of motor unit number estimation methods versus quantitative motor unit potential analysis in diagnosis of ALS. Clin Neurophysiol 129(3):646–653. https://doi.org/10.1016/j.clinph.2018.01.002
Gaiani A, Martinelli I, Bello L, Querin G, Puthenparampil M, Ruggero S, Toffanin E, Cagnin A, Briani C, Pegoraro E, Sorarù G (2017) Diagnostic and prognostic biomarkers in amyotrophic lateral sclerosis: neurofilament light chain levels in definite subtypes of disease. JAMA Neurol 74(5):525–532. https://doi.org/10.1001/jamaneurol.2016.5398
Acknowledgements
This article is in memory of Valeriano Fantin, a strong man called away by this terrible disease. This work was supported by the Italian Ministry of Health (“Ricerca Corrente”). BoReALS group: I. Bartolomei, R. Plasmati, F. Pastorelli, C. C. Quarta, V. Reale, V. Mariano, D. Milletti, R. Nasca, F. Rizzi, A. Cherici, D. Rusolo, L. Valeriani, F. Anzolin, E. Fantoni, A. Fiorito, L. Andrini, P. Avoni, V. A. Donadio, S. Capellari, S. de Pasqua, G. Rizzo, V. Vacchiano, E. Fileccia, L. Morandi, F. Marliani, L. Albini-Riccioli, P. Parchi, M. P. Foschini, A. Pession, S. Battaglia, R. Poda, D. Valenti, S. Asioli, L. Vignatelli, F. Oppi, M. Stanzani-Maserati, A. Bartoletti-Stella, C. Colombo, S. Maselli, M.P. Giannoccaro, M. Moresco, F. Salvi, R. Liguori.
Author information
Authors and Affiliations
Consortia
Contributions
Conceptualization: SAR, VV, RL and PP; Methodology acquisition and analysis of data: all authors; Writing-original draft preparation: SAR, VV, RL and PP; Writing-review and editing: PP based on the critical revision of all authors; Supervision: PP.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical standard
The study was conducted according to the revised Declaration of Helsinki and Good Clinical Practice guidelines and approved by the “Area Vasta Emilia Centro” ethics committee. Informed consent was given by study participants or the next of kin.
Additional information
The members of the BoReALS group are listed in the Acknowledgements.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Abu-Rumeileh, S., Vacchiano, V., Zenesini, C. et al. Diagnostic-prognostic value and electrophysiological correlates of CSF biomarkers of neurodegeneration and neuroinflammation in amyotrophic lateral sclerosis. J Neurol 267, 1699–1708 (2020). https://doi.org/10.1007/s00415-020-09761-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-020-09761-z