Abstract
Objectives
To describe the neurological phenotype of children with prenatal diagnosis of agenesis of corpus callosum (ACC) and interhemispheric cysts associated with malformations of cortical development (MCD).
Methods
We reviewed the neuroimaging, neurologic, EEG, and genetic data of 36 patients (21 males, mean age 7 years) with ACC and interhemispheric cysts. Associations were tested with Chi-squared and Fisher exact tests.
Results
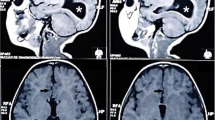
According to the 2001 Barkovich classification, we found 4 type 1c (11.1%), 6 type 2a (16.6%), 18 type 2b (50%, 6/18 girls with Aicardi syndrome), and 9 type 2c cysts (22.2%). EEG showed specific epileptic activity in 27/36 patients (75%). Epilepsy was diagnosed in 16 subjects (16/36, 44.4%), including all Aicardi patients, and was associated with cognitive impairment (p = 0.032). Severe intellectual disability and epilepsy were associated with type 2b cysts, always due to Aicardi patients (p < 0.05). After excluding Aicardi patients, all subjects with type 2b cysts had mild neurological phenotype. Patients with 2a and 2c cysts more frequently had normal cognition (83.3% and 62.5% of cases, respectively). Patients with type 1c cyst mostly had mild/moderate cognitive impairment. Severe neurologic deficits were associated with 1c cysts and 2b cysts with Aicardi syndrome (p < 0.05). Multilobar and/or bilateral MCD were associated with severe neurological and epileptic phenotypes (p < 0.05).
Conclusion
Once excluded Aicardi syndrome, most patients with ACC and interhemispheric cysts have a mild clinical phenotype characterized by borderline/normal cognition and minor neurological signs. Despite the high prevalence of EEG epileptic abnormalities, epilepsy in these cases is infrequent and usually responsive to antiepileptic drugs.

Similar content being viewed by others
References
Paul LK, Brown WS, Adolphs R et al (2007) Agenesis of the corpus callosum: genetic, developmental and functional aspects of connectivity. Nat Rev Neurosci 8:287–299. https://doi.org/10.1038/nrn2107
Edwards TJ, Sherr EH, Barkovich AJ, Richards LJ (2014) Clinical, genetic and imaging findings identify new causes for corpus callosum development syndromes. Brain 137:1579–1613. https://doi.org/10.1093/brain/awt358
Guillem P, Fabre B, Cans C et al (2003) Trends in elective terminations of pregnancy between 1989 and 2000 in a French county (the Isère). Prenat Diagn 23:877–883. https://doi.org/10.1002/pd.711
Hetts SW, Sherr EH, Chao S et al (2006) Anomalies of the corpus callosum: an MR analysis of the phenotypic spectrum of associated malformations. AJR Am J Roentgenol 187:1343–1348. https://doi.org/10.2214/AJR.05.0146
Raybaud C, Girard N (1998) Anatomic MRI study of commissural agenesis and dysplasia of the telencephalon (agenesis of the corpus callosum and related anomalies). Clinical correlations and morphogenetic interpretation. Neurochirurgie 44:38–60
Byrd SE, Radkowski MA, Flannery A, McLone DG (1990) The clinical and radiological evaluation of absence of the corpus callosum. Eur J Radiol 10:65–73
Raybaud C (2010) The corpus callosum, the other great forebrain commissures, and the septum pellucidum: anatomy, development, and malformation. Neuroradiology 52:447–477. https://doi.org/10.1007/s00234-010-0696-3
Romaniello R, Marelli S, Giorda R et al (2017) Clinical characterization, genetics, and long-term follow-up of a large cohort of patients with agenesis of the corpus callosum. J Child Neurol 32:60–71. https://doi.org/10.1177/0883073816664668
Govil-Dalela T, Kumar A, Agarwal R, Chugani HT (2017) Agenesis of the corpus callosum and aicardi syndrome: a neuroimaging and clinical comparison. Pediatr Neurol 68:44–48.e2. https://doi.org/10.1016/j.pediatrneurol.2016.12.002
DAntonio F, Pagani G, Familiari A et al (2016) Outcomes associated with isolated agenesis of the corpus callosum: a meta-analysis. Pediatrics 138:e20160445–e20160445. https://doi.org/10.1542/peds.2016-0445
Folliot-Le Doussal L, Chadie A, Brasseur-Daudruy M et al (2018) Neurodevelopmental outcome in prenatally diagnosed isolated agenesis of the corpus callosum. Early Hum Dev 116:9–16. https://doi.org/10.1016/j.earlhumdev.2017.10.004
Al-Hashim AH, Blaser S, Raybaud C, MacGregor D (2016) Corpus callosum abnormalities: neuroradiological and clinical correlations. Dev Med Child Neurol 58:475–484. https://doi.org/10.1111/dmcn.12978
Yeh H-R, Park H-K, Kim H-J et al (2018) Neurodevelopmental outcomes in children with prenatally diagnosed corpus callosal abnormalities. Brain Dev 40:634–641. https://doi.org/10.1016/j.braindev.2018.04.012
Chadie A, Radi S, Trestard L et al (2008) Neurodevelopmental outcome in prenatally diagnosed isolated agenesis of the corpus callosum. Acta Paediatr 97:420–424. https://doi.org/10.1111/j.1651-2227.2008.00688.x
Barkovich AJ, Simon EM, Walsh CA (2001) Callosal agenesis with cyst: a better understanding and new classification. Neurology 56:220–227
Desikan RS, Barkovich AJ (2016) Malformations of cortical development. Ann Neurol 80:797–810. https://doi.org/10.1002/ana.24793
Aicardi J, Chevrie JJ, Rousselie F (1969) Spasma-in-flexion syndrome, callosal agenesis, chorioretinal abnormalities. Arch Fr Pediatr 26:1103–1120
Aicardi J (2005) Aicardi syndrome. Brain Dev 27:164–171. https://doi.org/10.1016/j.braindev.2003.11.011
Pavone P, Barone R, Baieli S et al (2005) Callosal anomalies with interhemispheric cyst: expanding the phenotype. Acta Paediatr 94:1066–1072. https://doi.org/10.1080/08035250510027372
Griebel ML, Williams JP, Russell SS et al (1995) Clinical and developmental findings in children with giant interhemispheric cysts and dysgenesis of the corpus callosum. Pediatr Neurol 13:119–124
Lena G, van Calenberg F, Genitori L, Choux M (1995) Supratentorial interhemispheric cysts associated with callosal agenesis: surgical treatment and outcome in 16 children. Childs Nerv Syst 11:568–573
Haverkamp F, Heep A, Woelfle J (2002) Psychomotor development in children with early diagnosed giant interhemispheric cysts. Dev Med Child Neurol 44:556–560
Oh KY, Kennedy AM, Selden NR et al (2012) Asymmetric ventriculomegaly, interhemispheric cyst, and dysgenesis of the corpus callosum (AVID): an imaging triad. J Ultrasound Med 31:1811–1820
Moeschler JB, Shevell M, Committee on Genetics (2014) Comprehensive evaluation of the child with intellectual disability or global developmental delays. Pediatrics 134:e903–e918. https://doi.org/10.1542/peds.2014-1839
Riou EM, Ghosh S, Francoeur E, Shevell MI (2009) Global developmental delay and its relationship to cognitive skills. Dev Med Child Neurol 51:600–606. https://doi.org/10.1111/j.1469-8749.2008.03197.x
Kwan P, Arzimanoglou A, Berg AT et al (2010) Definition of drug resistant epilepsy: consensus proposal by the ad hoc Task Force of the ILAE commission on therapeutic strategies. Epilepsia 51:1069–1077. https://doi.org/10.1111/j.1528-1167.2009.02397.x
Caldarelli M, Di Rocco C (1996) Surgical options in the treatment of interhemispheric arachnoid cysts. Surg Neurol 46:212–221
Pilu G, Falco P, Perolo A et al (1997) Differential diagnosis and outcome of fetal intracranial hypoechoic lesions: report of 21 cases. Ultrasound Obstet Gynecol 9:229–236. https://doi.org/10.1046/j.1469-0705.1997.09040229.x
Bannister C, Russell S, Rimmer S, Mowle D (1999) Fetal arachnoid cysts: their site, progress, prognosis and differential diagnosis. Eur J Pediatr Surg 9:27–28. https://doi.org/10.1055/s-2008-1072308
Tange Y, Aoki A, Mori K et al (2000) Interhemispheric glioependymal cyst associated with agenesis of the corpus callosum–case report. Neurol Med Chir (Tokyo) 40:536–542
Blaicher W, Prayer D, Kuhle S et al (2001) Combined prenatal ultrasound and magnetic resonance imaging in two fetuses with suspected arachnoid cysts. Ultrasound Obstet Gynecol 18:166–168. https://doi.org/10.1046/j.1469-0705.2001.00503.x
Cinalli G, Peretta P, Spennato P et al (2006) Neuroendoscopic management of interhemispheric cysts in children. J Neurosurg Pediatr 105:194–202. https://doi.org/10.3171/ped.2006.105.3.194
Murphy A-M, Brenner C, Ann Lynch S (2006) Agenesis of the corpus callosum with interhemispheric cyst, hepatic haemangioma and trisomy 21. Clin Dysmorphol 15:149–151. https://doi.org/10.1097/01.mcd.0000198933.16953.db
Rizk E, Awad AJ, Tubbs RS et al (2013) Dorsal third ventricular cysts revisited. Child’s Nerv Syst 29:2271–2274. https://doi.org/10.1007/s00381-012-2009-0
Dall’Asta A, van Oostrum N, Basheer S et al (2018) Etiology and prognosis of severe ventriculomegaly diagnosed at late gestation. Ultraschall der Medizin Eur J Ultrasound. https://doi.org/10.1055/a-0627-7173
Solt LC, Deck JH, Baim RS, TerBrugge K (1980) Interhemispheric cyst of neuroepithelial origin in association with partial agenesis of the corpus callosum. Case report and review of the literature. J Neurosurg 52:399–403. https://doi.org/10.3171/jns.1980.52.3.0399
Morimoto T, Kaneko M, Nishikawa R et al (1986) Ependymal cyst–case report. No Shinkei Geka 14:351–356
Ulu MO, Kafadar AM, Dashti R et al (2010) Treatment of symptomatic interhemispheric arachnoid cysts by cystoperitoneal shunting. J Clin Neurosci 17:700–705. https://doi.org/10.1016/j.jocn.2009.09.029
Fuchs F, Moutard ML, Blin G et al (2008) Prenatal and postnatal follow-up of a fetal interhemispheric arachnoid cyst with partial corpus callosum agenesis, asymmetric ventriculomegaly and localized polymicrogyria. Fetal Diagn Ther 24:385–388. https://doi.org/10.1159/000165511
Uematsu Y, Kubo K, Nishibayashi T et al (2000) Interhemispheric neuroepithelial cyst associated with agenesis of the corpus callosum. Pediatr Neurosurg 33:31–36. https://doi.org/10.1159/000028972
Inagaki H, Kurosaki M, Hori T et al (1992) [Interhemispheric choroidal epithelial cyst associated with partial agenesis of the corpus callosum: case report and review of the literature]. No Shinkei Geka 20:1301–1306
Korsic M, Jugović D, Porcnik A (2013) Endoscopic treatment of in utero diagnosed multiloculated interhemispheric cyst in a newborn: case report. Acta Clin Croat 52:119–124
Mankotia DS, Sardana H, Sinha S et al (2016) Pediatric interhemispheric arachnoid cyst: an institutional experience. J Pediatr Neurosci 11:29–34. https://doi.org/10.4103/1817-1745.181258
Fruhman G, Eble TN, Gambhir N et al (2012) Ophthalmologic findings in Aicardi syndrome. J AAPOS 16:238–241. https://doi.org/10.1016/j.jaapos.2012.01.008
Laclef C, Anselme I, Besse L et al (2015) The role of primary cilia in corpus callosum formation is mediated by production of the Gli3 repressor. Hum Mol Genet 24:4997–5014. https://doi.org/10.1093/hmg/ddv221
Putoux A, Baas D, Paschaki M et al (2018) Altered GLI3 and FGF8 signaling underlies Acrocallosal syndrome phenotypes in Kif7 depleted mice. Hum Mol Genet. https://doi.org/10.1093/hmg/ddy392
Wieland I, Jakubiczka S, Muschke P et al (2004) Mutations of the Ephrin-B1 Gene Cause Craniofrontonasal Syndrome. Am J Hum Genet 74:1209–1215. https://doi.org/10.1086/421532
Utsunomiya H, Yamashita S, Takano K et al (2006) Midline cystic malformations of the brain: imaging diagnosis and classification based on embryologic analysis. Radiat Med 24:471–481. https://doi.org/10.1007/s11604-006-0049-7
Acknowledgements
The authors thank Carola Martinetti, Radiology Unit, University of Genoa, Genoa, Italy. They are also thank the patients and family members, and the ASSACCI (Associazione Anomalie Corpo Calloso Italia) Association.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical standards
This was a double-center retrospective observational study performed at the Istituto Giannina Gaslini Children’s Hospital (Genoa, Italy) and McGill University (Montreal, Canada). The Ethical Committees of both institutions waived written parental consent due to the retrospective nature of the study. This study has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Uccella, S., Accogli, A., Tortora, D. et al. Dissecting the neurological phenotype in children with callosal agenesis, interhemispheric cysts and malformations of cortical development. J Neurol 266, 1167–1181 (2019). https://doi.org/10.1007/s00415-019-09247-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-019-09247-7