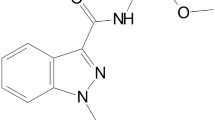
Abstract
This case report centres on the post-mortem distribution of the synthetic cannabinoid MDMB-CHMICA and its metabolites in the case of a 27-year-old man found dead after falling from the 24th floor of a high-rise building. Toxicological analysis of post-mortem samples confirmed, besides consumption of the synthetic cannabinoids MDMB-CHMICA (1.7 ng/mL femoral blood) and EG-018, the abuse of THC (9.3 ng/mL femoral blood), amphetamine (1050 ng/mL femoral blood), MDMA (275 ng/mL femoral blood), and cocaine. Regarding EG-018 and cocaine, only traces were detected in heart blood as well as in the brain (EG-018) and urine (cocaine), respectively, which is why no quantification was conducted in the femoral blood sample. It was concluded from femoral blood analysis that, at the time of death, the man was under the influence of the synthetic cannabinoid MDMB-CHMICA, THC, amphetamine and MDMA. Comprehensive screenings of all post-mortem specimens were conducted to elucidate the post-mortem distribution of MDMB-CHMICA and its metabolites. The MDMB-CHMICA concentrations ranged between 0.01 ng/mL (urine) and 5.5 ng/g (brain). Comparably low concentrations were detected in cardiac and femoral blood (2.1 ng/mL and 1.7 ng/mL, respectively) as well as in the psoas major muscle (1.2 ng/g). Higher concentrations were found in the lung (2.6 ng/g), liver (2.6 ng/g), and kidney (3.8 ng/g). Gastric content yielded a MDMB-CHMICA concentration of 2.4 ng/g (1.1 μg absolute). Screening for MDMB-CHMICA metabolites resulted in the detection of mainly monohydroxylated metabolites in the blood, kidney, and liver specimens. Results indicated that monohydroxylated metabolites of MDMB-CHMICA are appropriate target analytes for detecting MDMB-CHMICA intake.




Similar content being viewed by others
Change history
05 December 2018
The following clarification to the content of Table 3 (MDMB-CHMICA data from previously published cases of acute (non-fatal) and fatal drug intoxications) of the named manuscript needs to be brought to the readership’s attention.
References
Banister SD, Longworth M, Kevin R, Sachdev S, Santiago M, Stuart J, Mack JBC, Glass M, McGregor IS, Connor M, Kassiou M (2016) Pharmacology of Valinate and tert-Leucinate synthetic cannabinoids 5F-AMBICA, 5F-AMB, 5F-ADB, AMB-FUBINACA, MDMB-FUBINACA, MDMB-CHMICA, and their analogues. ACS Chem Neurosci 7:1241–1254. https://doi.org/10.1021/acschemneuro.6b00137
Hill SL, Najafi J, Dunn M, Acheampong P, Kamour A, Grundlingh J, Blain PG, Thomas SHL (2016) Clinical toxicity following analytically confirmed use of the synthetic cannabinoid receptor agonist MDMB-CHMICA. A report from the identification of novel psychoActive substances (IONA) study. Clin Toxicol 54(8):638–643. https://doi.org/10.1080/15563650.2016.1190980
Hermanns-Clausen M, Müller D, Kithinji J, Angerer V, Franz F, Eyer F, Neurath H, Liebetrau G, Auwärter V (2017) Acute side effects after consumption of the new synthetic cannabinoids AB-CHMINACA and MDMBCHMICA. Clin Toxicol 56:404–411. https://doi.org/10.1080/15563650.2017.1393082
EMCDDA (2016) EU early warning system alert - MDMB CHMICA
EMCDDA/EUROPOL (2016) Europol Joint Report - MDMB CHMICA
Haden M, Archer JRH, Dargan PI, Wood DM (2017) MDMB-CHMICA: availability, patterns of use, and toxicity associated with this novel psychoactive substance. Subst Use Misuse 52(2):223–232. https://doi.org/10.1080/10826084.2016.1223692
Castellanos D, Gralnik LM (2016) Synthetic cannabinoids 2015: an update for pediatricians in clinical practice. World J Clin Pediatr 5(1):16–24. https://doi.org/10.5409/wjcp.v5.i1.16
Adamowicz P (2016) Case report: fatal intoxication with synthetic cannabinoid MDMB-CHMICA. Forensic Sci Int 261:e5–e10. https://doi.org/10.1016/j.forsciint.2016.02.024
Seywright A, Torrance HJ, Wylie FM, McKeown DA, Lowe DJ, Stevenson R (2016) Analysis and clinical findings of cases positive for the novel synthetic cannabinoid receptor agonist MDMB-CHMICA. Clin Toxicol 54(8):632–637. https://doi.org/10.1080/15563650.2016.1186805
Bäckberg M, Tworek L, Beck O, Helander A (2017) Analytically confirmed intoxications involving MDMB-CHMICA from the STRIDA project. J Med Toxicol 13:52–60. https://doi.org/10.1007/s13181-016-0584-2
Westin AA, Frost J, Brede WR, Gundersen PO, Einvik S, Aarset H, Slordal L (2015) Sudden cardiac death following use of the synthetic cannabinoid MDMB-CHMICA. J Anal Toxicol 40(1):86–87. https://doi.org/10.1093/jat/bkv110
Angerer V, Franz F, Schwarze B, Moosmann B, Auwärter V (2016) Letter to the editor. Reply to ‘sudden cardiac death following use of the synthetic cannabinoid MDMB-CHMICA’. J Anal Toxicol 40:240–242. https://doi.org/10.1093/jat/bkw004
Kronstrand R, Tyrkko E, Lindstedt D, Roman M (2015) Poster P103: MMB-CHMINACA blood concentrations in recreational users and fatal intoxications. Paper presented at the 53rd the International Association of Forensic Toxicologists (TIAFT) meeting, Firenze, Italy
Franz F, Schwörer N, Angerer V, Moosmann B, Auwarter V (2015) Metabolism and urine analysis of the new synthetic cannabinoid MDMB-CHMICA. Toxichem Krimtech 192(82):1–7
Franz F, Angerer V, Moosmann B, Auwärter V (2017) Phase I metabolism of the highly potent synthetic cannabinoid MDMB-CHMICA and detection in human urine samples. Drug Test Anal 9(5):744–753. https://doi.org/10.1002/dta.2049
Grigoryev A, Kavanagh P, Pechnikov A (2016) Human urinary metabolite pattern of a new synthetic cannabimimetic, methyl 2-(1-(cyclohexylmethyl)-1H-indole-3-carboxamido)-3,3-dimethylbutanoate. Forensic Toxicol 34:316–328. https://doi.org/10.1007/s11419-016-0319-8
Madea B (2015) Rechtsmedizin (in German), vol 3. Springer Verlag
Ruchholtz S, Nast-Kolb D, Waydhas C, Schweiberer L (1996) Das Verletzungsmuster beim Polytrauma (in German). Unfallchirurg 99:633–641
Gaunitz F, Schürenkamp J, Rostamzadeh A, Konkol C, Thevis M, Rothschild MA, Mercer-Chalmers-Bender K (2017) Analysis of taxine B/isotaxine B in a plasma specimen by LC–MS/MS in a case of fatal poisoning: concealed suicide by ingestion of yew (Taxus L.) leaves of a patient with a long-term history of borderline personality disorder. Forensic Toxicol 35(2):421–427. https://doi.org/10.1007/s11419-017-0355-z
Lehmann S, Schulze B, Thomas A, Kamphausen T, Thevis M, Rothschild MA, Mercer-Chalmers-Bender K (2018) Organ distribution of 4-MEC, MDPV, methoxetamine and α-PVP: comparison of QuEChERS and SPE. Forensic Toxicol 36:320–333. https://doi.org/10.1007/s11419-018-0408-y
Lehmann S, Kieliba T, Beike J, Thevis M, Mercer-Chalmers-Bender K (2017) Determination of 74 new psychoactive substances in serum using automated in-line solid-phase extraction-liquid chromatography-tandem mass spectrometry. J Chromatogr B (accepted for publication) 1064:124–138. https://doi.org/10.1016/j.jchromb.2017.09.003
Wintermeyer A, Möller I, Thevis M, Jübner M, Beike J, Rothschild MA, Bender K (2010) In vitro phase I metabolism of the synthetic cannabimimetic JWH-018. Anal Bioanal Chem 398(5):2141–2153. https://doi.org/10.1007/s00216-010-4171-0
Schulz M, Schmoldt A (2003) Therapeutic and toxic blood concentrations of more than 800 drugs and other xenobiotics. Pharmazie 58(7):447–474
Baselt RC (2011) Disposition of toxic drugs and chemicals in man, vol 9. Biomedical Publications
Giroud C, Menetrey A, Augsburger M, Buclin T, Sanchez-Mazas P, Mangin P (2001) THC, 11-OH-THC and THC-COOH plasma or serum to whole blood concentrations distribution ratios in blood samples taken from leaving and dead people. Forensic Sci Int 123:159–164
Liu RH, Liu H-C, Lin D-L (2006) Distribution of methylenedioxymethamphetamine (MDMA) and methylenedioxyamphetamine (MDA) in postmortem and antemortem specimens. J Anal Toxicol 30(8):545–450
Kalant H (2001) The pharmacology and toxicology of “ecstasy” (MDMA) and related drugs. CMAI 165(7):917–928
Krizevski R, Dudai N, Bar E, Lewinsohn E (2007) Developmental patterns of phenylpropylamino alkaloids accumulation in khat (Catha edulis, Forsk.). J Ethnopharmacol 114:432–438. https://doi.org/10.1016/j.jep.2007.08.042
Grue-Sorensen G, Spenser ID (1994) Biosynthetic route to the Ephedra alkaloids. J Am Chem Soc 116:6195–6200
Mathys K, Brenneisen R (1992) Determination of (S)-(-)-cathinone and its metabolites (R,S’)-(-)-norephedrine and (R,R)-(-)-norpseudoephedrine in urine by high-performance liquid chromatography with photodiode-array detection. J Chromatogr 593:78–85
Toennes SW, Kauert GF (2002) Excretion and detection of cathinone, cathine, and phenylpropanolamine in urine after Kath chewing. Clin Chem 48(10):1715–1719
Toennes SW, Harder S, Schramm M, Niess C, Kauert GF (2003) Pharmacokinetics of cathinone, cathine and norephedrine after the chewing of khat leaves. J Clin Pharmacol 56:125–130
Taylor KB (1973) Dopamine-β-hydroxylase: stereochemical course of the reaction. J Biol Chem 249(2):454–458
Maurer HH, Bickeboeller-Friedrich J, Kraemer T, Peters FT (2000) Toxicokinetics and analytical toxicology of amphetamine-derived designer drugs (‘ecstasy’). Toxicol Lett 112-113:133–142
Feio-Azevedo R, Costa VM, Ferreira LM, Branco PS, Pereira FC, Bastos ML, Carvalho F, Capela JP (2017) Toxicity of the amphetamine metabolites 4-hydroxyamphetamine and 4-hydroxynorephedrine in human dopaminergic differentiated SH-SY5Y cells. Toxicol Lett 269:65–76. https://doi.org/10.1016/j.toxlet.2017.01.012
Huestis MA, Henningfield JE, Cone EJ (1992) Blood cannabinoids. I. Absorption of THC and formation of 11-OH-THC and THCCOOH during and after smoking marijuana. J Anal Toxicol 16:276–282
Skopp G, Richter B, Pötsch L (2003) Cannabinoidbefunde im serum 24-48 Stunden nach Rauchkonsum (in German) [serum cannabinoid levels 24 to 48 hours after cannabis smoking]. Arch Kriminol 212(3–4):83–95
Fabritius M, Augsburger M, Chtioui H, Favrat B, Giroud C (2014) Fitness to drive and cannabis: validation of two blood THCCOOH thresholds to distinguish occasional users from heavy smokers. Forensic Sci Int 242:1–8. https://doi.org/10.1016/j.forsciint.2014.05.014
Fabritius M, Favrat B, Chtioui H, Battistella G, Annoni J-M, Appenzeller M, Dao K, Fornari E, Lauer E, Mall J-F, Maeder P, Mangin P, Stauba C, Girouda C (2014) THCCOOH concentrations in whole blood: are they useful in discriminating occasional from heavy smokers? Drug Test Anal 6:155–163
Pélissier-Alicot A-L, Gaulier J-M, Champsaur P, Marquet P (2003) Mechanisms underlying postmortem redistribution of drugs: a review. J Anal Toxicol 27:533–544
Skopp G (2004) Preanalytic aspects in postmortem toxicology. Forensic Sci Int 142:75–100. https://doi.org/10.1016/j.forsciint.2004.02.012
Acknowledgments
The authors wish to thank the laboratory staff for performing the routine systematic toxicological analysis. In addition, the authors thank Tobias Kieliba and Sibylle Banaschak (Institute of Legal Medicine, Medical Faculty, University of Cologne) for technical support. Special thanks are directed to June Mercer-Chalmers-Bender for editorial support and English language editing.
Funding
This project was funded by the German Federal Ministry for Economic Affairs and Energy and ZIM (Central Innovations Programs for Medium-sized enterprises) (KF2429613MD3) and the German ‘Bund gegen Alkohol und Drogen im Straßenverkehr e.V.’ (a non-profit association against alcohol and drugs on the road).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Compliance with the law
All experiments comply with the current laws of the Federal Republic of Germany. The public prosecutor’s office and the legal representative of the deceased gave permission for this case to be published.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Gaunitz, F., Lehmann, S., Thomas, A. et al. Post-mortem distribution of the synthetic cannabinoid MDMB-CHMICA and its metabolites in a case of combined drug intoxication. Int J Legal Med 132, 1645–1657 (2018). https://doi.org/10.1007/s00414-018-1911-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-018-1911-8