Abstract
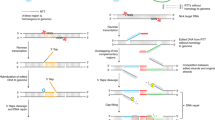
Targeted insertion of large pieces of DNA is an important goal of genetic engineering. However, this goal has been elusive since classical methods for homology-directed repair are inefficient and often not feasible in many systems. Recent advances are described here that enable site-specific genomic insertion of relatively large DNA with much improved efficiency. Using the preferred repair pathway in the cell of nonhomologous end-joining, DNA of up to several kb could be introduced with remarkably good precision by the methods of HITI and ObLiGaRe with an efficiency up to 30–40%. Recent advances utilizing homology-directed repair (methods of PITCh; short homology arms including ssODN; 2H2OP) have significantly increased the efficiency for DNA insertion, often to 40–50% or even more depending on the method and length of DNA. The remaining challenges of integration precision and off-target site insertions are summarized. Overall, current advances provide major steps forward for site-specific insertion of large DNA into genomes from a broad range of cells and organisms.







Similar content being viewed by others
Abbreviations
- AAV:
-
Adeno-associated viral vector
- a-NHEJ:
-
Alternative nonhomologous end-joining
- Cas:
-
CRISPR-associated protein
- c-NHEJ:
-
Canonical nonhomologous end-joining
- CRISPR:
-
Clustered regularly interspaced short palindromic repeat
- crRNA:
-
CRISPR RNA (the guide RNA)
- ctRNP:
-
a complex of pre-assembled crRNA + tracRNA + Cas9 protein
- dCas9:
-
catalytically inactive Cas9
- DSB:
-
Double-strand break
- dsDNA:
-
Double-stranded DNA
- Easi-CRISPR:
-
Efficient additions with ssDNA inserts-CRISPR
- HITI:
-
Homology-independent targeted integration
- HDR:
-
Homology-directed repair
- HMEJ:
-
Homology-mediated end-joining
- HR:
-
Homologous recombination
- Indels:
-
Small insertions and deletions
- MMEJ:
-
Microhomology-mediated end-joining
- NHEJ:
-
Nonhomologous end-joining
- ObLiGaRe:
-
Obligate ligation-gated recombination.
- PAM:
-
Protospacer-adjacent motif
- PITCh:
-
Precise integration into target chromosome
- SDSA:
-
DNA synthesis-dependent strand annealing
- sgRNA:
-
single guide RNA containing a crRNA sequence and tracRNA (for CRISPR-Cas)
- sgRNP:
-
complex of pre-assembled guide RNA and Cas9 protein
- SSA:
-
Single-strand annealing
- ssDNA:
-
Single-stranded DNA
- ssODN:
-
Single-strand oligodeoxynucleotide
- TALEN:
-
Transcription activator-like effector nuclease
- tracrRNA:
-
Transactivating CRISPR RNA (a structural RNA to recruit Cas9)
- 2H2OP:
-
Two-hit by sgRNA and two oligos with a targeting plasmid
- 3H2OP:
-
Three-hit by sgRNA and two oligos with a targeting plasmid
- ZFN:
-
Zinc finger nuclease
References
Aida T, Chiyo K, Usami T, Ishikubo H, Imahashi R, Wada Y, Tanaka KF, Sakuma T, Yamamoto T, Tanaka K (2015) Cloning-free CRISPR/Cas system facilitates functional cassette knock-in in mice. Genome Biol 16:87
Aida T, Nakade S, Sakuma T, Izu Y, Oishi A, Mochida K, Ishikubo H, Usami T, Aizawa H, Yamamoto T, Tanaka K (2016) Gene cassette knock-in in mammalian cells and zygotes by enhances MMEJ. BMC Genomics 17:979
Auer TO, Duroure K, De Cian A, Concordet J-P, Del Bene F (2014) Highly efficient CRISPR/Cas9-mediated knock-in in zebrafish by homology-independent DNA repair. Genome Res 24:142–153
Bachu R, Bergareche I, Chasin LA (2015) CRISPR-Cas targeted plasmid integration into mammalian cells via non-homologous end joining. Biotechnol Bioeng 112:2154–2162
Barrangou R, Doudna JA (2016) Applications of CRISPR technologies in research and beyond. Nat Biotechnol 34:933–941
Barrangou R, Marraffini LA (2014) CRISPR-Cas systems: prokaryotes upgrade to adaptive immunity. Mol Cell 54:234–244
Bétermier M, Bertrand P, Lopez BS (2014) Is non-homologous end-joining really an inherently error-prone process? PLoS Genet 10:e1004086
Beumer KJ, Trautman JK, Bozas A, Liu JL, Rutter J, Gall JG, Carroll D (2008) Efficient gene targeting in Drosophila by direct embryo injection with zinc-finger nucleases. Proc Natl Acad Sci 105:19821–19826
Beumer KJ, Trautman JK, Mukherjee K, Carroll D (2013) Donor DNA utilization during gene targeting with zinc-finger nucleases. G3: genes/genomes. Genetics 3:657–664
Bevan MW, Chilton MD (1982) T-DNA of the agrobacterium Ti and Ri plasmids. Ann Rev Genet 16:357–384
Bhargava R, Onyango DO, Stark JM (2016) Regulation of single-strand annealing and its role in genome maintenance. Trends Genet 32:566–575
Bibikova M, Carroll D, Segal DJ, Trautman JK, Smith J, Kim YG, Chandrasegaran S (2001) Stimulation of homologous recombination through targeted cleavage by a chimeric nuclease. Mol Cell Biol 21:289–297
Bibikova M, Golic M, Golic KG, Carroll D (2002) Targeted chromosomal cleavage and mutagenesis in Drosophila using zinc-finger nucleases. Genetics 161:1169–1175
Bibikova M, Beumer K, Trautman JK, Carroll D (2003) Enhancing gene targeting using designed zinc finger nucleases. Science 300:764
Bishop KA et al (2016) CRISPR/Cas9-mediated insertion of loxP sites in the mouse Dock7 gene provides an effective alternative to use of targeted embryonic stem cells. G3: genes/genomes. Genetics 6:2051–2061
Boch J, Scholze H, Schornack S, Landgraf A, Hahn S, Kay S, Lahaye T, Nickstadt A, Bonas U (2009) Breaking the code of DNA binding specificity of TAL type III effectors. Science 326:1509–1512
Bozas A, Beumer KJ, Trautman JK, Carroll D (2009) Genetic analysis of zinc-finger nuclease-induced gene targeting in Drosophila. Genetics 182:641–651
Branzei D, Foiani M (2008) Regulation of DNA repair throughout the cell cycle. Nat Rev Mol Cell Biol 9:297–308
Capecchi MR (1989) Altering the genome by homologous recombination. Science 244:1288–1292
Carroll D (2011) Genome engineering with zinc-finger nucleases. Genetics 188:773–782
Carroll D (2014) Genome engineering with targetable nucleases. Annu Rev Biochem 83:409–439
Carroll D, Beumer KJ, Trautman JK (2010) High-efficiency gene targeting in Drosophila with zinc finger nucleases. Methods Mol Biol 649:271–280
Ceccaldi R, Rondinelli B, D’Andrea AD (2016) Repair pathway choices and consequences at the double-strand break. Trends Cell Biol 26:52–64
Chandrasegaran S, Carroll D (2016) Origins of programmable nucleases for genome engineering. J Mol Biol 428:963–989
Chen S, Lee B, Lee AYF, Modzelewski AJ, He L (2016) Highly efficient mouse genome editing by CRISPR ribonucleoprotein electroporation in zygotes. J Biol Chem 291:14457–14467
Christian M, Cermark T, Doyle EL, Schmidt C, Zhang F et al (2010) Targeting DNA double-strand breaks with TAL effector nucleases. Genetics 186:757–761
Chu VT, Weber T, Wefers B, Wurst W, Sander S, Rajewsky K, Kühn R (2015) Increasing the efficiency of homology-directed repair for CRISPR-Cas9-induced precise gene editing in mammalian cells. Nat Biotechnol 33:543–548
Cristea S, Freyvert Y, Santiago Y, Holmes MC, Urnov FD, Gregory PD, Cost GJ (2013) In vivo cleavage of transgene donors promotes nuclease-mediated targeted integration. Biotechnol Bioeng 110:871–880
Danner E, Bashir S, Yumlu S, Wurst W, Wefers B, Kühn R (2017) Control of gene editing by manipulation of DNA repair mechanisms. Mamm Genome 287-8:262–274
Deng C, Capecchi MR (1992) Reexamination of gene targeting frequency as a function of the extent of homology between the targeting vector and the target locus. Mol Cell Biol 12:3365–3371
Deng D, Yan C, Pan X, Mahfouz M, Wang J, Zhu JK, Shi Y, Yan N (2012) Structural basis for sequence-specific recognition of DNA by TAL effectors. Science 335:720–723
Doudna JA, Charpentier E (2014) The new frontier of genome engineering with CRISPR-Cas9. Science 346:1077–1086
Doyon Y, Vo TD, Mendel MC, Greenberg SG, Wang J, Xia DF, Miller JC, Urnov FD, Gregory PD, Holmes MC (2011) Enhancing zinc-finger-nuclease activity with improved obligate heterodimer architectures. Nat Methods 8:74–79
Dueva D, Iliakis G (2013) Alternative pathways of non-homologous end joining (NHEJ) in genomic instability and cancer. Transl Cancer Res 2:163–177
Fattah F, Lee EH, Weisensel N, Wang Y, Lichter N, Hendrickson EA (2010) Ku regulates the non-homologous end joining pathway choice of DNA double-strand break repair in human somatic cells. PLoS Genet 6:e1000855
Ferenczi A, Pyott DE, Xipnitou A, Molnar A (2017) Efficient targeted DNA editing and replacement in Chlamydomonas reinhardtii using Cpf1 ribonucleoproteins and single-stranded DNA. Proc Natl Acad Sci 114:13567–13572
Fu Y, Sander JD, Reyon D, Cascio VM, Joung JK (2014) Improving CRISPR-Cas nuclease specificity using truncated guide RNAs. Nat Biotechnol 32:279–284
Gaj T, Gersbach CA, Barbas CF (2013) ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol 31:397–405
Groth AC, Fish M, Nusse R, Calos MP (2004) Construction of transgenic Drosophila by using the site-specific integrase from phage phiC31. Genetics 166:1775–1782
Guilinger JP, Thompson DB, Liu DR (2014) Fusion of catalytically inactive Cas9 to FokI nuclease improves the specificity of genome modification. Nat Biotechnol 32:577–582
Handler AM (2002) Use of the piggyBac transposon for germ-line transformation of insects. Insect Biochem Mol Biol 32:1211–1220
He X, Tan C, Wang F, Wang Y, Zhou R, Cui D, You W, Zhao H, Ren J, Feng B (2016) Knock-in of large reporter genes in human cells via CRISPR/Cas9 induced homology-dependent and independent DNA repair. Nucleic Acids Res 44:e85
Hisano Y, Sakuma T, Nakade S, Ohga R, Ota S, Okamoto H, Yamamoto T, Kawahara A (2015) Precise in-frame integration of exogenous DNA mediated by CRISPR/Cas9 system in zebrafish. Sci Rep 5:8841
Horii T, Morita S, Kimura M, Terawaki N, Shibutani M, Hatada I (2017) Efficient generation of conditional knockout mice via sequential introduction of lox sites. Sci Rep 71:7891
Horn C, Wimmer EA (2000) A versatile vector set for animal transgenesis. Dev Genes Evol 210:630–637
Hsu PD, Lander ES, Zhang F (2014) Development and applications of CRISPR-Cas9 for genome engineering. Cell 157:1262–1278
Iyama T, Wilson DM (2013) DNA repair mechanisms in dividing and nondividing cells. DNA Repair 12:620–636
Jasin M, Haber JE (2016) The democratization of gene editing: insights from site-specific cleavage and double-strand break-repair. DNA Repair 44:6–16
Jiang J, Jing Y, Cost GJ, Chiang JC, Kolpa HJ, Cotton AM, Carone DM, Carone BR, Shivak DA, Guschin DY, Pearl JR, Rebar EJ, Byron M, Gregory PD, Brown CJ, Urnov FD, Hall LL, Lawrence JB (2013) Translating dosage compensation to trisomy 21. Nature 500:296–300
Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E (2012) Programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337:816–821
Kim Y-G, Cha J, Chandrasegaran S (1996) Hybrid restriction enzymes: zinc finger fusions to FokI cleavage domain. Proc Natl Acad Sci 93:1156–1160
Kim D, Kim S, Kim S, Park J, Kim JS (2016) Genome-wide target specificities of CRISPR-Cas9 nucleases revealed by multiplex Digenome-sEq. Genome Res 26:406–415
Kleinstiver BP, Pattanayak V, Prew MS, Tsai SQ, Nguyen NT, Zheng Z, Joung JK (2016) High-fidelity CRISPR-Cas9 nucleases with no detectable genome-wide off-target effects. Nature 529:490–495
Lanza DG, Gaspero A, Lorenzo I, Liao L, Zheng P et al (2017) Employing single-stranded DNA donors for the high throughput production of conditional knockout alleles in mice. BioRxiv. https://doi.org/10.1101/195651
Li L, Wu LP, Chandrasegaran S (1992) Functional domains in FokI restriction endonuclease. Proc Natl Acad Sci 89:4275–4279
Liang X, Potter J, Kumar S, Ravinder N, Chesnut JD (2017) Enhanced CRISPR/Cas9-mediated precise genome editing by improved design and delivery of gRNA, Cas9 nuclease, and donor DNA. J Biotechnol 241:136–146
Lin S, Staahl BT, Alla RK, Doudna JA (2014) Enhanced homology-directed human genome engineering by controlled timing of CRISPR/Cas9 delivery. eLIFE 3:e04766
Lin C, Li H, Hao M, Xiong D, Luo Y, Huang C, Yuan Q, Zhang J, Xia N (2016) Increasing the efficiency of CRISPR/ Cas9-mediated precise genome editing of HSV-1 virus in human cells. Sci Rep 6:34531
Liu X, Homma A, Sayadi J, Yang S, Ohashi J, Takumi T (2016) Sequence features associated with cleavage efficiency of CRISPR/Cas9 system. Sci Rep 6:19675
Lobo N, Li X, Fraser MJ (1999) Transposition of the piggyBac element in embryos of Drosophila melanogaster, Aedes aegypti and Tricholposia ni. Mol Gen Genet 261:803–810
Ma X, Chen C, Veevers J, Zhou X, Ross RS, Feng W, Chen J (2017) CRISPR/Cas9-mediated gene manipulation to create single-amino-acid-substituted and floxed mice with a cloning-free method. Sci Rep 7:42244
Mak AN, Bradley P, Cernadas RA, Bogdanov AJ, Stoddard BL (2012) The crystal structure of TAL effector PthXo1 bound to its DNA target. Science 335:716–719
Mali P, Aach J, Stranges PB, Esvelt KM, Moosburner M, Kosuri S, Yang L, Church GM (2013) CAS9 transcriptional activators for target specificity screening and paired nickases for cooperative genome engineering. Nat Biotechnol 31:833–838
Mansour SL, Thomas KR, Capecchi MR (1988) Disruption of the proto-oncogene int-2 in mouse embryo-derived stem cells: a general strategy for targeting mutations to non-selectable genes. Nature 336:348–352
Maresca M, Lin V, Guo N, Yang Y (2013) Obligate ligation-gated recombination (ObLiGaRe): custom-designed nuclease-mediated targeted integration through nonhomologous end joining. Genome Res 23:539–546
Maruyama T, Dougan SK, Truttmann MC, Bilate AM, Ingram JR, Ploegh HL (2015) Increasing the efficiency of precise genome editing with CRISPR-Cas9 by inhibition of nonhomologous end joining. Nat Biotechnol 33:538–542
Miano JM, Zhu QM, Lowenstein CJ (2016) A CRISPR path to engineering new genetic mouse models for cardiovascular research. Arterioscler Thromb Vasc Biol 36:1058–1975
Miller JC, Holmes MC, Wang J, Guschin DY, Lee Y-L, Rupniewski I, Beausejour CM, Waite AJ, Wang NS, Kim KA, Gregory PD, Pabo CO, Rebar EJ (2007) An improved zinc-finger nuclease architecture for highly specific genome cleavage. Nat Biotechnol 25:778–785
Miller JC, Tan S, Qiao G, Barlow KA, Wang J, Xia DF, Meng X, Paschon DE, Leung E, Hinkley SJ, Dulay GP, Hua KL, Ankoudinova I, Cost GJ, Urnov FD, Zhang HS, Holmes MC, Zhang L, Gregory PD, Rebar EJ (2011) A TALE nuclease architecture for efficient genome editing. Nat Biotechnol 29:143–148
Miura H, Gurumurthy CB, Sato T, Sato M, Ohtsuka M (2015) CRISPR/Cas9-based generation of knockdown mice by intronic insertion of artificial microRNA using longer single-stranded DNA. Sci Rep 5:12799
Miura H, Quadros RM, Gurumurthy CB, Ohtsuka M (2018) Easi-CRISPR for creating knock-in and conditional knockout mouse models using long ssDNA donors. Nat Protoc 13:195–215
Mladenov E, Magin S, Soni A, Iliakis G (2016) DNA double-strand-break repair in higher eukaryotes and its role in genomic instability and cancer, cell cycle and proliferation-dependent regulation. Semin Cancer Biol 37-38:51–64
Moehle EA, Rock JM, Lee YL, Jouvenot Y, DeKelver RC et al (2007) Targeted gene addition into a specified location in the human genome using designed zinc finger nucleases. Proc Natl Acad Sci 104:3055–3060
Morton J, Davis MW, Jorgensen EM, Carroll D (2006) Induction and repair of zinc finger nuclease-targeted double-strand breaks in Caenorhabditis elegans somatic cells. Proc Natl Acad Sci 103:16370–16375
Mussolino C, Morbitzer R, Lütge F, Dannemann N, Lahaye T, Cathomen T (2011) A novel TALE nuclease scaffold enables high genome editing activity in combination with low toxicity. Nucleic Acids Res 39:9283–9293
Nakade S, Tsubota T, Sakane Y, Kume S, Sakamot N et al (2014) Microhomology-mediated end-joining-dependent integration of donor DNA in cells and animals using TALENs and CRISPR/Cas9. Nature Comm 5:5560
O'Brochta DA, Atkinson PW (1996) Transposable elements and gene transformation in non-drosophilid insects. Insect Biochem Mol Biol 26:739–753
Orlando SJ, Santiago Y, DeKelver RC, Freyvert Y, Boydston EA et al (2010) Zinc-finger nuclease-driven targeted integration into mammalian genomes using donors with limited chromosomal homology. Nucleic Acids Res 38:e152
Orr-Weaver TL, Szostak JW, Rothstein RJ (1981) Yeast transformation: a model system for the study of recombination. Proc Natl Acad Sci 78:6354–6358
Orthwein A, Noordermeer SM, Wilson MD, Landry S, Enchev RI et al (2015) Mechanism for the suppression of homologous recombination in G1 cells. Nature 528:422–426
Paix A, Wang Y, Smith HE, Lee CY, Calidas D et al (2014) Scalable and versatile genome editing using linear DNAs with microhomology to Cas9 sites in Caenorhabditis elegans. Genetics 198:1347–1356
Paix A, Schmidt H, Seydoux G (2016) Cas9-assisted recombineering in C. elegans: genome editing using in vivo assembly of linear DNAs. Nucleic Acids Res 44:e128
Paix A, Folkmann A, Goldman DH, Kulaga H, Grzelak MJ, Rasoloson D, Paidemarry S, Green R, Reed RR, Seydoux G (2017a) Precision genome editing using synthesis-dependent repair of Cas9-induced DNA breaks. Proc Natl Acad Sci 114:E10745–E10754
Paix A, Folkmann A, Seydoux G (2017b) Precision genome editing using CRISPR-Cas9 and linear repair templates in C. elegans. Methods 121-122:86–93
Pan Y, Xiao L, Li ASS, Zhang X, Sirois P, Zhang J, Li K (2013) Biological and biomedical applications of engineered nucleases. Mol Biotechnol 55:54–62
Peng Y, Clark KJ, Campbell JM, Panetta M, Guo Y et al (2014) Making designer mutants in model organisms. Development 142:4042–4054
Porteus MH, Baltimore D (2003) Chimeric nucleases stimulate gene targeting in human cells. Science 300:763
Qi LS, Larson MH, Gilbert LA, Doudna JA, Weissman JS, Arkin AP, Lim WA (2013) Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression. Cell 152:1173–1183
Quadros RM, Miura H, Harms DW, Akatsuka H, Sato T, Aida T, Redder R, Richardson GP, Inagaki Y, Sakai D, Buckley SM, Seshacharyulu P, Batra SK, Behlke MA, Zeiner SA, Jacobi AM, Izu Y, Thoreson WB, Urness LD, Mansour SL, Ohtsuka M, Gurumurthy CB (2017) Easi-CRISPR: a robust method for one-step generation of mice carrying conditional and insertion alleles using long ssDNA donors and CRISPR ribonucleoproteins. Genome Biol 18:92
Ramalingam S, Kandavelou K, Rajenderan R, Chandrasegaran S (2011) Creating designed zinc finger nucleases with minimal cytotoxicity. J Mol Biol 405:630–641
Ran FA, Hsu PD, Lin CY, Gootenberg JS, Konermann S, Trevino AE, Scott DA, Inoue A, Matoba S, Zhang Y, Zhang F (2013) Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity. Cell 154:1380–1389 Erratum in Cell 155: 479–480
Reyon D, Tsai SQ, Khayter C, Foden JA, Sander JD, Joung JK (2012) FLASH assembly of TALENs for high-throughput genome editing. Nat Biotechnol 30:460–465
Richardson CD, Ray GJ, DeWitt MA, Curie GL, Corn JE (2016) Enhancing homology-directed genome editing by catalytically active and inactive CRISPR-Cas9 using asymmetric donor DNA. Nat Biotechnol 34:339–344
Rodgers K, McVey M (2016) Error-prone repair of DNA double-strand breaks. J Cell Physiol 231:15–24
Rouet P, Smih F, Jasin M (1994) Expression of a site-specific endonuclease stimulates homologous recombination in mammalian cells. Proc Natl Acad Sci 91:6064–6068
Rubin GM, Spradling AC (1982) Genetic transformation of Drosophila with transposable element vectors. Science 218:348–533
Saito S, Maeda R, Adachi N (2017) Dual loss of human POLQ and LIG4 abolishes random integration. Nat Commun 8:16112
Sakuma T, Shota Nakade S, Sakane Y, Suzuki K-IT, Yamamoto T (2016) MMEJ-assisted gene knock-in using TALENs and CRISPR-Cas9 with the PITCh systems. Nat Protoc 11:118–133
Salsman J, Dellaire G (2017) Precision genome editing in the CRISPR era. Biochem Cell Biol 95:187–201
Sander JD, Joung JK (2014) CRISPR-Cas systems for editing, regulating and targeting genomes. Nat Biotechnol 32:347–355
Scherer S, Davis RW (1979) Replacement of chromosome segments with altered DNA sequences constructed in vitro. Proc Natl Acad Sci 76:4951–4955
Sedivy JM, Sharp PA (1989) Positive genetic selection for gene disruption in mammalian cells by homologous recombination. Proc Natl Acad Sci 86:227–231
Segal DJ, Meckler JF (2013) Genome engineering at the dawn of the golden age. Annu Rev Genomics Hum Genet 14:135–158
Sfeir A, Symington LS (2015) Microhomology-mediated end joining: a back-up survival mechanism or dedicated pathway? Trends Biochem Sci 40:701–714
Singh P, Schimenti JC, Bolcun-Filas E (2015) A mouse geneticist's practical guide to CRISPR applications. Genetics 199:1–15
Slaymaker IM, Gao L, Zetsche B, Scott DA, Yan WX, Zhang F (2016) Rationally engineered Cas9 nucleases with improved specificity. Science 351:84–88
Smih F, Rouet P, Romanienko PJ, Jasin M (1995) Double-strand breaks at the target locus stimulate gene targeting in embryonic stem cells. Nucleic Acids Res 23:5012–5019
Smithies O, Gregg RG, Boggs SS, Koralewski MA, Kucherlapati RS (1985) Insertion of DNA sequences into the human chromosomal beta-globin locus by homologous recombination. Nature 317:230–234
Söllü C, Pars K, Cornu TI, Thibodeau-Beganny S, Maeder ML et al (2010) Autonomous zinc-finger nuclease pairs for targeted chromosomal deletion. Nucleic Acids Res 38:8269–8276
Spradling AC, Rubin GM (1982) Transposition of cloned P elements into Drosophila germ line chromosomes. Science 218:341–347
Stella S, Montoya G (2016) The genome editing revolution: a CRISPR-Cas TALE off-target story. Bioessays 38:S4–S13
Suzuki K, Tsunekawa Y, Hernandez-Benitez R, Wu J, Zhu J, Kim EJ, Hatanaka F, Yamamoto M, Araoka T, Li Z, Kurita M, Hishida T, Li M, Aizawa E, Guo S, Chen S, Goebl A, Soligalla RD, Qu J, Jiang T, Fu X, Jafari M, Esteban CR, Berggren WT, Lajara J, Nuñez-Delicado E, Guillen P, Campistol JM, Matsuzaki F, Liu GH, Magistretti P, Zhang K, Callaway EM, Zhang K, Belmonte JCI (2016) In vivo genome editing via CRISPR/Cas9 mediated homology-independent targeted integration. Nature 540:144–149
Szczepek M, Brondani V, Buchel J, Serrano L, Segal DJ et al (2007) Structure-based redesign of the dimerization interface reduces the toxicity of zinc-finger nucleases. Nat Biotechnol 25:786–793
Thomas KR, Folger KR, Capecchi MR (1986) High frequency targeting of genes to specific sites in the mammalian genome. Cell 44:419–428
Tsai SQ, Joung JK (2016) Defining and improving the genome-wide specificities of CRISPR-Cas9 nucleases. Nat Rev Genet 17:300–312
Tsai SQ, Wyvekens N, Khayter C, Foden JA, Thapar V, Reyon D, Goodwin MJ, Aryee MJ, Joung JK (2014) Dimeric CRISPR RNA-guided FokI nucleases for highly specific genome editing. Nat Biotechnol 32:569–576
Tsai SQ, Zheng Z, Nguyen NT, Liebers M, Topkar VV, Thapar V, Wyvekens N, Khayter C, Iafrate AJ, le LP, Aryee MJ, Joung JK (2015) GUIDE-seq enables genome-wide profiling of off-target cleavage by CRISPR-Cas nucleases. Nat Biotechnol 33:187–197
Urnov FD, Miller JC, Lee Y-L, Beausejour CM, Rock JM, Augustus S, Jamieson AC, Porteus MH, Gregory PD, Holmes MC (2005) Highly efficient endogenous human gene correction using designed zinc-finger nucleases. Nature 435:646–651
Urnov FD, Rebar EJ, Holmes MC, Zhang HS, Gregory PD (2010) Genome editing with engineered zinc finger nucleases. Nat Rev Genet 11:636–646
Venken KJ, He Y, Hoskins RA, Bellen HJ (2006) P[acman]: a BAC transgenic platform for targeted insertion of large DNA fragments in D. melanogaster. Science 314:1747–1751
Venken KJ, Carlson JW, Schulze KL, Pan H, He Y, Spokony R, Wan KH, Koriabine M, de Jong PJ, White KP, Bellen HJ, Hoskins RA (2009) Versatile P[acman] BAC libraries for transgenesis studies in Drosophila melanogaster. Nat Methods 6:431–434
Wang L, Shao Y, Guan Y, Li L, Wu L, Chen F, Liu M, Chen H, Ma Y, Ma X, Liu M, Li D (2015) Large genomic fragment deletion and functional gene cassette knock-in via Cas9 protein mediated genome editing in once-cell rodent embryos. Sci Rep 5:17517
Weinthal DM, Taylor RA, Tzfira T (2013) Nonhomologous end joining-mediated gene replacement in plant cells. Plant Physiol 162:390–400
Wesolowska N, Rong YS (2013) Long-range targeted manipulation of the Drosophila genome by site-specific integration and recombinational resolution. Genetics 193:411–419
Xu H, Xiao T, Chen CH, Li W, Meyer CA, Wu Q, Wu D, Cong L, Zhang F, Liu JS, Brown M, Liu XS (2015) Sequence determinants of improved sgRNA design. Genome Res 25:1147–1157
Yamamoto Y, Bliss J, Gerbi SA (2015) Whole organism genome editing: targeted large DNA insertion via ObLiGaRe nonhomologous end-joining in vivo capture. G3: Genes/Genomes/Genetics 5:1843–1847
Yang H, Wang H, Shivalila CS, Cheng AW, Shi L, Jaenisch R (2013) One-step generation of mice carrying reporter and conditional alleles by CRISPR/Cas-mediated genome engineering. Cell 154:1370–1379
Yao X, Wang X, Hu X, Liu Z, Liu J, Zhou H et al (2017) Homology-mediated end joining-based targeted integration using CRISPR/Cas9. Cell Res 276:801–814
Yao X, Wang X, Liu J, Shi L, Huang P, Yang H (2018) CRISPR/Cas9-mediated targeted integration in vivo using a homology-mediated end joining-based strategy. J Vis Exp (133). https://doi.org/10.3791/56844
Yoshimi K, Kunihiro Y, Kaneko T, Nagahora H, Voigt B, Mashimo T (2016) ssODN- mediated knock-in with CRISPR-Cas for large genomic regions in zygotes. Nat Commun 7:10431
Zelensky AN, Schimmel J, Kool H, Kanaar R, Tijsterman M (2017) Inactivation of pol θ and C-NHEJ eliminates off-target integration of exogenous DNA. Nat Commun 8:66
Zetsche B, Gootenberg JS, Abudayyeh OO, Slaymaker IM, Makarova KS, Essletzbichler P, Volz SE, Joung J, van der Oost J, Regev A, Koonin EV, Zhang F (2015) Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-Cas system. Cell 163:759–771
Zhang F (2015) CRISPR-Cas9: prospects and challenges. Hum Gene Ther 26:409–410
Zhang JP, Li XL, Li GH, Chen W, Arakaki C, Botimer GD, Baylink D, Zhang L, Wen W, Fu YW, Xu J, Chun N, Yuan W, Cheng T, Zhang XB (2017) Efficient precise knockin with a double cut HDR donor after CRISPR/Cas9-mediated double-stranded DNA cleavage. Genome Biol 18:35
Acknowledgements
Research in the author’s lab is supported by NIH R01 HG008160, NIH R01 GM121455, and NSF MCB 1607411.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Yamamoto, Y., Gerbi, S.A. Making ends meet: targeted integration of DNA fragments by genome editing. Chromosoma 127, 405–420 (2018). https://doi.org/10.1007/s00412-018-0677-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00412-018-0677-6