Abstract
Purpose
The purpose of the study was to investigate the association between gene phosphate and tensin homolog (PTEN) single nucleotide polymorphisms (SNPs) and risk of developing polycystic ovary syndrome (PCOS) in South Indian women. PTEN is one of the most important tumor suppressor genes that regulate cell proliferation, migration, and death. It is also involved in the maintenance of genome stability. PCOS is one of the most common endocrine disorders among women of reproductive age. It is a heterogeneous syndrome characterized by abnormal reproductive cycles, irregular ovulation, hormonal imbalance, hyperandrogenism, acne and hirsutism.
Research question
What is the association status of PTEN SNPs with PCOS?
Methods
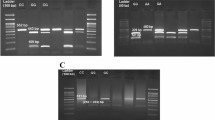
A total of 240 subjects were recruited in this case–control study comprising 110 patients with PCOS and 130 individuals without PCOS. All the subjects were of South Indian origin. The genotyping of PTEN SNPs (rs1903858 A/G, rs185262832G/A and rs10490920T/C) was carried out on DNA from subjects by polymerase chain reaction (PCR) and sequencing analysis. Haplotype frequencies for multiple loci and the standardized disequilibrium coefficient (D′) for pairwise linkage disequilibrium (LD) were surveyed by Haploview Software.
Results
Our results showed significant increase in the frequencies of rs1903858 A/G (P = 0.0016), rs185262832 G/A (P = 0.0122) and rs10490920 T/C (P = 0.0234) genotypes and alleles in cases compared to controls.
Conclusion
The PTEN (rs1903858A/G, rs185262832G/A and rs10490920T/C) gene polymorphisms may constitute an inheritable risk factor for PCOS in South Indian women.


Similar content being viewed by others
References
Krul-Poel YH, Agca R, Lips P, van Wijland H, Stam F, Simsek S (2015) Vitamin D status is associated with skin autofluorescence in patients with type 2 diabetes mellitus: a preliminary report. Cardiovasc Diabetol 16(14):89
Lei L, Ding L, Su J, Liu M, Shi Q, Zhou J, Sun H, Yan G (2017) Attenuated expression of MTR in both prenatally androgenized mice and women with the hyperandrogenic phenotype of PCOS. PLoS One 12(12):e0187427
Zhao H, Zhou D, Chen Y, Liu D, Chu S, Zhang S (2017) Beneficial effects of Heqi san on rat model of polycystic ovary syndrome through the PI3K/AKT pathway. Daru 25(1):21
Kim J, Mersereau JE, Khankari N, Bradshaw PT, McCullough LE, Cleveland R, Shantakumar S, Teitelbuam SL, Neugut AI, Senie RT, Gammon MD (2016) Polycystic ovarian syndrome (PCOS), related symptoms/sequelae, and breast cancer risk in a population-based case-control study. Cancer Causes Control. 27(3):403–414
Karadeniz M, Erdogan M, Berdeli A (2007) The progesterone receptor PROGINS polymorphism is not related to oxidative stress factors in women with polycystic ovary syndrome. Cardiovasc Diabetol 6:29
Mary MJ, Saravanan L, Deecaraman M, Vijayalakshmi M, Umashankar V, Sailaja J (2017) Polymorphism of the PAI-1gene (4G/5G) may be linked with Polycystic Ovary Syndrome and associated pregnancy disorders in South Indian Women. Bioinformation 13(5):149–153
Siddamalla S, Reddy TV, Govatati S, Erram N, Deenadayal M, Shivaji S, Bhanoori M (2018) Vitamin D receptor gene polymorphisms and risk of polycystic ovary syndrome in South Indian women. Gynecol Endocrinol 34(2):161–165
Siddamalla S, Reddy TV, Govatati S, Guruvaiah P, Deenadayal M, Shivaji S, Bhanoori M (2018) Influence of tumour suppressor gene (TP53, BRCA1 and BRCA2) polymorphisms on polycystic ovary syndrome in South Indian women. Eur J Obstet Gynecol Reprod Biol 227:13
Reddy TV, Govatati S, Deenadayal M, Sisinthy S, Bhanoori M (2017) Impact of mitochondrial DNA copy number and displacement loop alterations on polycystic ovary syndrome risk in south Indian women. Mitochondrion 17:1567–7249
Guruvaiah P, Govatati S, Reddy TV (2016) Analysis of Connexin37 gene C1019T polymorphism and PCOS susceptibility in South Indian population: case-control study. Eur J Obstet Gynecol Reprod Biol 196:17–20
Lv J, Zhu Q, Jia X, Yu N, Li Q (2016) In vitro and in vivo effects of tumor suppressor gene PTEN on endometriosis: an experimental study. Med Sci Monit 16(22):3727–3736
Sharma I, Singh D (2012) Conjugated linoleic acids attenuate FSH- and IGF1-stimulated cell proliferation; IGF1, GATA4, and aromatase expression; and estradiol-17β production in buffalo granulosa cells involving PPARγ, PTEN, and PI3K/Akt. Reproduction 144(3):373–383
Wang Y, Zhao S, Zhu L, Zhang Q, Ren Y (2018) MiR-19a negatively regulated the expression of PTEN and promoted the growth of ovarian cancer cells. Gene 378–1119(18):30553–30555
Pulido R (2018) PTEN inhibition in human disease therapy. Molecules 23(2):285
Govatati S, Kodati VL, Deenadayal M, Chakravarty B, Shivaji S, Bhanoori M (2014) Mutations in the PTEN tumor gene and risk of endometriosis: a case–control study. Hum Reprod 29(2):324–336
Sun L, Liu J, Yuan Q, Xing C, Yuan Y (2014) Association between PTEN Gene IVS4 polymorphism and risk of cancer: a meta-analysis. PLoS One 9(6):98851
Zou W, Wang B, Wang J, Zhang Z, Xu X, Chen B, Ma X, Cao Y (2015) No association between polymorphisms in PTEN and primary ovarian insufficiency in a Han Chinese population. Reprod Biol Endocrinol 17(13):62
Kim SS, Eun JW, Cho HJ, Lee H-Y, Seo CW, Lee GH, Yoon SY, Noh CK, Cho SW, Cheong JY (2019) Effect of PTEN Polymorphism on the Development of Hepatitis B Virus-associated Hepatocellular Carcinoma. J Liver Cancer 19(1):46–54
Jing F, Mao Y, Zhang Z, Li Y, Cai S, Li Q, Ma X, Jin M, Chen K (2014) The association of phosphatase and tensin homolog deleted on chromosome 10 polymorphisms and lifestyle habits with colorectal cancer risk in a Chinese population. Tumour Biol 35(9):9233–9240
Ding J, Gao Y, Liu R, Xu F, Liu H (2011) Association of PTEN polymorphisms with susceptibility to hepatocellular carcinoma in a Han Chinese population. DNA Cell Biol 30(4):229–234
Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group (2004) Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome. Fertil Steril 81:19–25
Bhanoori M, Deenadayal M, Kennedy S (2007) The G2964A 3′-untranslated region polymorphism of the signal transducer and activator of transcription 6 gene is associated with endometriosis in South Indian women. Hum Reprod 22(4):1026–1030
Barrett JC, Fry B, Maller J (2005) Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21:263–265
Biau DJ, Jolles BM, Porcher R (2010) P value and the theory of hypothesis testing: an explanation for new researchers. Clin Orthop Relat Res 468(3):885–892
Ghaleh TD, Skandari SS, Najafipour R, Rashvand Z, Darabi M, Sahmani M (2018) Association of − 604G/A and − 501A/C Ghrelin and obestatin prepropeptide gene polymorphisms with Polycystic Ovary Syndrome. Biochem Genet 56(1–2):116–127
Liu J, Visser-Grieve S, Boudreau J, Yeung B, Lo S, Chamberlain G, Yu F, Sun T, Papanicolaou T, Lam A, Yang X, Chin-Sang I (2014) Insulin activates the insulin receptor to downregulate the PTEN tumour suppressor. Oncogene 33(29):3878–3885
Chen CY, Chen J, He L, Stiles BL (2018) PTEN: tumor suppressor and metabolic regulator. Front Endocrinol (Lausanne) 9(9):338
Shafiee MN, Seedhouse C, Mongan N, Chapman C, Deen S, Abu J, Atiomo W (2016) Up-regulation of genes involved in the insulin signalling pathway (IGF1, PTEN and IGFBP1) in the endometrium may link polycystic ovarian syndrome and endometrial cancer. Mol Cell Endocrinol 15(424):94–101
Lerer-Serfaty G, Samara N, Fisch B, Shachar M, Kossover O, Seliktar D, Ben-Haroush A, Abir R (2013) Attempted application of bioengineered/biosynthetic supporting matrices with phosphatidylinositol-trisphosphate-enhancing substances to organ culture of human primordial Follicles. J Assist Reprod Genet 30(10):1279–1288
Reddy P, Liu L, Adhikari D, Jagarlamudi K, Rajareddy S, Shen Y, Du C, Tang W, Hämäläinen T, Peng SL, Lan ZJ, Cooney AJ, Huhtaniemi I, Liu K (2008) Oocyte-specific deletion of Pten causes premature activation of the primordial follicle pool. Science 319(5863):611–613
Lan ZJ, Krause MS, Redding SD, Li X, Wu GZ, Zhou HX, Bohler HC, Ko C, Cooney AJ, Zhou J, Lei ZM (2017) Selective deletion of Pten in theca-interstitial cells leads to androgen excess and ovarian dysfunction in mice. Mol Cell Endocrinol 15(444):26–37
Zhao Y, Song W, Wang Z, Wang Z, Jin X, Xu J, Bai L, Li Y, Cui J, Cai L (2018) Resveratrol attenuates testicular apoptosis in type 1 diabetic mice: Role of Akt-mediated Nrf2 activation and p62-dependent Keap1 degradation. Redox Biol 14:609–617
Zhou W, Shao H, Zhang D, Dong J, Cheng W, Wang L, Teng Y, Yu Z (2015) PTEN signaling is required for the maintenance of spermatogonial stem cells in mouse, by regulating the expressions of PLZF and UTF1. Cell Biosci 28(5):42
Mandal RK, Akhter N, Irshad M, Panda AK, Ali A, Haque S (2015) Association of the PTEN IVS4 (rs3830675) gene polymorphism with reduced risk of cancer: evidence from a meta-analysis. Asian Pac J Cancer Prev 16(3):897–902
Slattery ML, John EM, Torres-Mejia G, Lundgreen A, Herrick JS, Baumgartner KB, Hines LM, Stern MC, Wolff RK (2012) Genetic variation in genes involved in hormones, inflammation and energetic factors and breast cancer risk in an admixed population. Carcinogenesis 33(8):1512–1521
Hosgood HD III, Menashe I, He X, Chanock S, Lan Q (2009) PTEN identified as important risk factor of chronic obstructive pulmonary disease. Respir Med 103:1866–1870
Treloar SA, Zhao ZZ, Le L, Zondervan KT, Martin NG, Kennedy S, Nyholt DR, Montgomery GW (2007) Variants in EMX2 and PTEN do not contribute to risk of endometriosis. Mol Hum Reprod 13(8):587–594
Xie CC, Lu L, Sun J, Zheng SL, Isaacs WB, Gronberg H, Xu J (2011) Germ-line sequence variants of PTEN do not have an important role in hereditary and non-hereditary prostate cancer susceptibility. J Hum Genet 56(7):496–502
Pezzolesi MG, Li Y, Zhou XP, Pilarski R, Shen L, Eng C (2006) Mutation-positive and mutation-negative patients with Cowden and Bannayan–Riley–Ruvalcaba syndromes associated with distinct 10q haplotypes. Am J Hum Genet 79(5):923–934
Acknowledgements
Swapna Siddamalla would like to thank University Grants Commission (UGC-BSR-RFMS), India, for providing a Senior Research Fellowship (SRF).
Funding
This study was supported in part by grants from the Department of Science and Technology (DST), SERB-(Lr No: SR/FT/LS-188/2009) and DST-PURSE-II Programme (DST Sanction No. SR/PURSE Phase 2/32(G)), India to Dr. Manjula Bhanoori.
Author information
Authors and Affiliations
Contributions
SS: execution of experiments, analysis and interpretation of data, statistical analysis and drafting of manuscript. SG: acquisition of data. VKV: data analysis. EN: data analysis. MD: acquisition of data. SS: analysis and interpretation of data, drafting of manuscript. MB: conception and design of study, analysis and interpretation of data, statistical analysis, drafting of manuscript. All authors will have seen and agreed to the ‘Author Contribution’ statement.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests.
Ethical approval
The study was approved by the ethical committee and review board of Centre for Cellular and Molecular biology (CCMB), Hyderabad. In the study all the participants were of South Indian origin (Dravidian linguistic group).
Informed consent
Informed written consent form was obtained from all subjects prior to participation in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Siddamalla, S., Govatati, S., Venu, V.K. et al. Association of genetic variations in phosphatase and tensin homolog (PTEN) gene with polycystic ovary syndrome in South Indian women: a case control study. Arch Gynecol Obstet 302, 1033–1040 (2020). https://doi.org/10.1007/s00404-020-05658-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-020-05658-4