Abstract
Purpose
There are limited data regarding postoperative complications and autoimmune reactions caused by surgery in early-stage cervical cancer patients who underwent laparoscopic radical resection (LRR). This study aimed to investigate the therapeutic effect of LRR of cervical cancer patients and its effect on cytokines.
Methods
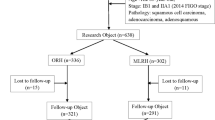
168 patients with cervical cancer were enrolled. The patients were divided into open group and laparoscopic group according to the random number table method, with 84 cases in each group. The surgical-related indexes and the incidence of complications of the two groups were observed, and the IFN-γ, TNF, and IL-1/2/4/6/8/10/12 levels in peripheral blood were compared before and after surgery in both groups.
Results
The operation time of the patients in the laparoscopic group was significantly shorter than that in the open group (119.56 ± 45.26 vs. 206.36 ± 54.39, P < 0.01). The intraoperative blood loss in the laparoscopic group was significantly less than that in the open group (155.29 ± 57.58 vs. 529.58 ± 162.4, P < 0.01). The postoperative visual analog scale (VAS) score was also significantly lower than that in the open group (3.65 ± 0.88 vs. 6.32 ± 1.12, P < 0.01). There was no significant difference in the incidence of complications between the two groups. The degree of inflammatory cytokines changes caused by LRR was less than that of open radical surgery (P < 0.001).
Conclusions
LRR surgery has less stress on patients with early cervical cancer than open surgery within 5 days after surgery, which has certain reference value for early cervical cancer treatment.
Similar content being viewed by others
References
Wu J, Ye T, Lv J, He Z, Zhu J (2019) Laparoscopic nerve-sparing radical hysterectomy vs laparoscopic radical hysterectomy in cervical cancer: a systematic review and meta-analysis of clinical efficacy and bladder dysfunction. J Minim Invasive Gynecol 26(3):417–426.e416. https://doi.org/10.1016/j.jmig.2018.10.012
Wu Y, Chen Y, Li L, Yu G, Zhang Y, He Y (2006) Associations of high-risk HPV types and viral load with cervical cancer in China. J Clin Virol Off Publ Pan Am Soc Clin Virol 35(3):264–269. https://doi.org/10.1016/j.jcv.2005.07.011
Jemal A, Siegel R, Xu J, Ward E (2010) Cancer statistics. CA Cancer J Clin 60(5):277–300. https://doi.org/10.3322/caac.20073
Diaz-Padilla I, Monk BJ, Mackay HJ, Oaknin A (2013) Treatment of metastatic cervical cancer: future directions involving targeted agents. Crit Rev Oncol Hematol 85(3):303–314. https://doi.org/10.1016/j.critrevonc.2012.07.006
Sobiczewski P, Bidzinski M, Derlatka P, Panek G, Danska-Bidzinska A, Gmyrek L, Michalski W (2009) Early cervical cancer managed by laparoscopy and conventional surgery: comparison of treatment results. Int J Gynecol Cancer Off J Int Gynecol Cancer Soc 19(8):1390–1395. https://doi.org/10.1111/IGC.0b013e3181ba5e88
Lee CL, Wu KY, Huang KG, Lee PS, Yen CF (2010) Long-term survival outcomes of laparoscopically assisted radical hysterectomy in treating early-stage cervical cancer. Am J Obstet Gynecol 203(2):165.e161–167. https://doi.org/10.1016/j.ajog.2010.02.027
Yan X, Li G, Shang H, Wang G, Chen L, Han Y (2009) Complications of laparoscopic radical hysterectomy and pelvic lymphadenectomy–experience of 117 patients. Int J Gynecol Cancer Off J Int Gynecol Cancer Soc 19(5):963–967. https://doi.org/10.1111/IGC.0b013e3181a79430
Luster AD (1998) Chemokines–chemotactic cytokines that mediate inflammation. N Engl J Med 338(7):436–445. https://doi.org/10.1056/nejm199802123380706
Tedgui A, Mallat Z (2006) Cytokines in atherosclerosis: pathogenic and regulatory pathways. Physiol Rev 86(2):515–581. https://doi.org/10.1152/physrev.00024.2005
Dranoff G (2004) Cytokines in cancer pathogenesis and cancer therapy. Nat Rev Cancer 4(1):11–22. https://doi.org/10.1038/nrc1252
Chopra V, Dinh TV, Hannigan EV (1998) Circulating serum levels of cytokines and angiogenic factors in patients with cervical cancer. Cancer Invest 16(3):152–159. https://doi.org/10.3109/07357909809050029
Smyth MJ, Cretney E, Kershaw MH, Hayakawa Y (2004) Cytokines in cancer immunity and immunotherapy. Immunol Rev 202:275–293. https://doi.org/10.1111/j.0105-2896.2004.00199.x
Yamamura M, Modlin RL, Ohmen JD, Moy RL (1993) Local expression of antiinflammatory cytokines in cancer. J Clin Invest 91(3):1005–1010. https://doi.org/10.1172/jci116256
du Bois A, Kristensen G, Ray-Coquard I, Reuss A, Pignata S, Colombo N, Denison U, Vergote I, Del Campo JM, Ottevanger P, Heubner M, Minarik T, Sevin E, de Gregorio N, Bidzinski M, Pfisterer J, Malander S, Hilpert F, Mirza MR, Scambia G, Meier W, Nicoletto MO, Bjorge L, Lortholary A, Sailer MO, Merger M, Harter P, AGO Study Group led Gynecologic Cancer Intergroup/European Network of Gynaecologic Oncology Trials Groups Intergroup Consortium (2016) Standard first-line chemotherapy with or without nintedanib for advanced ovarian cancer (AGO-OVAR 12): a randomised, double-blind, placebo-controlled phase 3 trial. Lancet Oncol 17(1):78–89. https://doi.org/10.1016/S1470-2045(15)00366-6
El-Deeb NM, El-Adawi HI, El-Wahab AEA, Haddad AM, El Enshasy HA, He YW, Davis KR (2019) Modulation of NKG2D, KIR2DL and cytokine production by glucan enhances natural killer cell cytotoxicity toward cancer cells. Front Cell Dev Biol 7:165. https://doi.org/10.3389/fcell.2019.00165
Woda A, Picard P, Dutheil F (2016) Dysfunctional stress responses in chronic pain. Psychoneuroendocrinology 71:127–135. https://doi.org/10.1016/j.psyneuen.2016.05.017
De Guise S, Levin M, Jasperse L, Risatti G, Wells RS (2019) T Helper cell subsets and their functions in common bottlenose dolphins (Tursiops truncatus). Front Immunol 10:1578. https://doi.org/10.3389/fimmu.2019.01578
Povoleri GA, Scottà C, Nova-Lamperti EA, John S, Lombardi G, Afzali B (2013) Thymic versus induced regulatory T cells - who regulates the regulators? Front Immunol 4:169. https://doi.org/10.3389/fimmu.2013.00169
van Zyl-Smit RN, Binder A, Meldau R, Semple PL, Evans A, Smith P, Bateman ED, Dheda K (2014) Cigarette smoke impairs cytokine responses and BCG containment in alveolar macrophages. Thorax 69(4):363–370. https://doi.org/10.1136/thoraxjnl-2013-204229
Sprague AH, Khalil RA (2009) Inflammatory cytokines in vascular dysfunction and vascular disease. Biochem Pharmacol 78(6):539–552. https://doi.org/10.1016/j.bcp.2009.04.029
Han P, Hodge G (1999) Intracellular cytokine production and cytokine receptor interaction of cord mononuclear cells: relevance to cord blood transplantation. Br J Haematol 107(2):450–457. https://doi.org/10.1046/j.1365-2141.1999.01696.x
Yokota S, Geppert TD, Lipsky PE (1988) Enhancement of antigen- and mitogen-induced human T lymphocyte proliferation by tumor necrosis factor-alpha. J Immunol 140(2):531–536
Wang YZ, Deng L, Xu HC, Zhang Y, Liang ZQ (2015) Laparoscopy versus laparotomy for the management of early stage cervical cancer. BMC Cancer 15:928. https://doi.org/10.1186/s12885-015-1818-4
Ramirez PT, Frumovitz M, Pareja R, Lopez A, Vieira M, Ribeiro R, Buda A, Yan X, Shuzhong Y, Chetty N, Isla D, Tamura M, Zhu T, Robledo KP, Gebski V, Asher R, Behan V, Nicklin JL, Coleman RL, Obermair A (2018) Minimally invasive versus abdominal radical hysterectomy for cervical cancer. N Engl J Med 379(20):1895–1904. https://doi.org/10.1056/NEJMoa1806395
Funding
This study was supported by Health Science and Technology Plan Projects of Yunnan Province (Grant Nos.: 2016NS195 and 2016NS196), Provincial Innovation Team Project of Human Assisted Reproductive Technology Research (Grant No.: 2017HC009), Martin Expert Workstation (Grant No.: 2018IC106), Special Project of Yunnan "Ten Thousand Plan · Famous Doctor" (Grant No.: YNWR-MY-2018–014), Clinical Medical Center of Yunnan Provincial Health Commission (Grant No.: ZX2019-01–01), and National Natural Science Foundation of China (Grant No.: 31700798).
Author information
Authors and Affiliations
Contributions
QX: Data collection and analysis, Manuscript writing and revision. MFD: Data analysis, Literature search, Manuscript writing and revision. WD: Data analysis, Literature search, Manuscript revision. DHY: Data analysis, Literature search, Data visualization, Manuscript review. JZ: Data collection, Literature search, Manuscript review and validation. JL: Data analysis, Manuscript revision and validation. LR: Data analysis, Manuscript revision and validation, Funding acquisition, Project administrator. YF: Study design, Data collection, Manuscript writing and revision, Funding acquisition, Project administrator.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the First People’s Hospital of Yunnan Province, the Affiliated Hospital of Kunming University of Science and Technology.
Informed consent
Written informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, Q., Dong, M., Dong, W. et al. Postoperative comparison of laparoscopic radical resection and open abdominal radical hysterectomy for cervical cancer patient. Arch Gynecol Obstet 302, 473–479 (2020). https://doi.org/10.1007/s00404-020-05606-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-020-05606-2