Abstract
Purpose
The aim of this retrospective study is to analyze the prognostic role and the practical implication of mesenteric lymph nodes (MLN) involvements in advanced ovarian cancer (AOC).
Methods
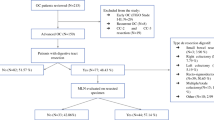
A total of 429 patients with AOC underwent surgery between December 2007 and May 2017. We included in the study 83 patients who had primary (PDS) or interval debulking surgery (IDS) for AOC with bowel resection. Numbers, characteristics and surgical implication of MLN involvement were considered.
Results
Eighty-three patients were submitted to bowel resection during cytoreduction for AOC. Sixty-seven patients (80.7%) underwent primary debulking surgery (PDS). Sixteen patients (19.3%) experienced interval debulking surgery (IDS). 43 cases (51.8%) showed MLN involvement. A statistic correlation between positive MLN and pelvic lymph nodes (PLN) (p = 0.084), aortic lymph nodes (ALN) (p = 0.008) and bowel infiltration deeper than serosa (p = 0.043) was found. A longer overall survival (OS) and disease-free survival was observed in case of negative MLN in the first 20 months of follow-up. No statistical differences between positive and negative MLN in terms of operative complication, morbidity, Ca-125, type of surgery (radical vs supra-radical), length and site of bowel resection, residual disease and site of recurrence were observed.
Conclusions
An important correlation between positive MLN, ALN and PLN was detected; these results suggest a lymphatic spread of epithelial AOC similar to that of primary bowel cancer. The absence of residual disease after surgery is an independent prognostic factor; to achieve this result should be recommended a radical bowel resection during debulking surgery for AOC with bowel involvement.


Similar content being viewed by others
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics: 2011. CA Cancer J Clin 61(2):69–90. https://doi.org/10.3322/caac.20107.Available
Hudson CN (1968) A radical operation for fixed ovarian tumours. BJOG An Int J Obstet Gynaecol 75(11):1155–1160. https://doi.org/10.1111/j.1471-0528.1968.tb02901.x
Scarabelli C, Gallo A, Franceschi S et al. (2000) Primary cytoreductive surgery with rectosigmoid colon resection for patients with advanced epithelial ovarian carcinoma. Cancer 88(2):389–397
Obermair A, Hagenauer S, Tamandl D et al (2001) Safety and efficacy of low anterior en bloc resection as part of cytoreductive surgery for patients with ovarian cancer. Gynecol Oncol 83(1):115–120. https://doi.org/10.1006/gyno.2001.6353
Berretta R, Marchesi F, Volpi L et al (2016) Posterior pelvic exenteration and retrograde total hysterectomy in patients with locally advanced ovarian cancer: clinical and functional outcome. Taiwan J Obstet Gynecol. 55(3):346–350. https://doi.org/10.1016/j.tjog.2016.04.010
Hertel H, Diebolder H, Herrmann J et al (2001) Is the decision for colorectal resection justified by histopathologic findings: a prospective study of 100 patients with advanced ovarian cancer. Gynecol Oncol 83(3):481–484. https://doi.org/10.1006/gyno.2001.6338
Bristow RE, Del Carmen MG, Kaufman HS, Montz FJ (2003) Radical oophorectomy with primary stapled colorectal anastomosis for resection of locally advanced epithelial ovarian cancer. J Am Coll Surg 197(4):565–574. https://doi.org/10.1016/S1072-7515(03)00478-2
Gillette-Cloven N, Burger RA, Monk BJ et al (2001) Bowel resection at the time of primary cytoreduction for epithelial ovarian cancer. J Am Coll Surg 193(6):626–632. https://doi.org/10.1016/S1072-7515(01)01090-0
Gallotta V, Fanfani F, Fagotti A et al (2014) Mesenteric lymph node involvement in advanced ovarian cancer patients undergoing rectosigmoid resection: prognostic role and clinical considerations. Ann Surg Oncol 21(7):2369–2375. https://doi.org/10.1245/s10434-014-3558-0
Gasimli K, Braicu EI, Nassir M et al (2016) Lymph node involvement pattern and survival differences of FIGO IIIC and FIGO IIIA1 ovarian cancer patients after primary complete tumor debulking surgery: a 10-year retrospective analysis of the tumor bank ovarian cancer network. Ann Surg Oncol 23(4):1279–1286. https://doi.org/10.1245/s10434-015-4959-4
Heitz F, Harter P, Alesina PF et al (2016) Pattern of and reason for postoperative residual disease in patients with advanced ovarian cancer following upfront radical debulking surgery. Gynecol Oncol 141(2):264–270. https://doi.org/10.1016/j.ygyno.2016.03.015
Jacquet P, Sugarbaker P (1996) Peritoneal carcinomatosis: principles of management. Perit Carcinomatosis Princ Manag 82:359–374. https://doi.org/10.1007/978-1-4613-1247-5
Esquivel J (2007) Cytoreductive surgery for peritoneal malignancies-development of standards of care for the community. Surg Oncol Clin N Am 16(3):653–666. https://doi.org/10.1016/j.soc.2007.04.015
Vizzielli G, Costantini B, Tortorella L et al (2016) A laparoscopic risk-adjusted model to predict major complications after primary debulking surgery in ovarian cancer: a single-institution assessment. Gynecol Oncol 142(1):19–24. https://doi.org/10.1016/j.ygyno.2016.04.020
Petrillo M, Vizzielli G, Fanfani F et al (2015) Definition of a dynamic laparoscopic model for the prediction of incomplete cytoreduction in advanced epithelial ovarian cancer: proof of a concept. Gynecol Oncol 139(1):5–9. https://doi.org/10.1016/j.ygyno.2015.07.095
Fagotti A, Ferrandina G, Fanfani F et al (2006) A laparoscopy-based score to predict surgical outcome in patients with advanced ovarian carcinoma: a pilot study. Ann Surg Oncol 13(8):1156–1161. https://doi.org/10.1245/ASO.2006.08.021
Gouy S, Goetgheluck J, Uzan C, Duclos J, Duvillard P, Morice P (2012) Prognostic factors for and prognostic value of mesenteric lymph node involvement in advanced-stage ovarian cancer. Eur J Surg Oncol 38(2):170–175. https://doi.org/10.1016/j.ejso.2011.10.006
Mita MT, Marchesi F, Cecchini S et al (2016) Prognostic assessment of gastric cancer: retrospective analysis of two decades. Acta Biomed 87(2):205–211. http://www.ncbi.nlm.nih.gov/pubmed/27649005. Accessed 2016
Riediger H, Keck T, Wellner U et al (2009) The lymph node ratio is the strongest prognostic factor after resection of pancreatic cancer. J Gastrointest Surg. 13(7):1337–1344. https://doi.org/10.1007/s11605-009-0919-2
Sun Z, Xu Y, Li DM et al (2010) Log odds of positive lymph nodes: a novel prognostic indicator superior to the number-based and the ratio-based n category for gastric cancer patients with R0 resection. Cancer 116(11):2571–2580. https://doi.org/10.1002/cncr.24989
Wang X, Appleby DH, Zhang X, Gan L, Wang JJ, Wan F (2013) Comparison of three lymph node staging schemes for predicting outcome in patients with gastric cancer. Br J Surg 100(4):505–514. https://doi.org/10.1002/bjs.9014
Dvoretsky PM, Richards KA, Angel C et al. (1998) Distribution of disease at autopsy in 100 women with ovarian cancer. Hum Pathol 19(1):57–63
O’Hanlan KA, Kargas S, Schreiber M et al (1995) Ovarian carcinoma metastases to gastrointestinal tract appear to spread like colon carcinoma: implications for surgical resection. Gynecol Oncol 59(2):200–206. https://doi.org/10.1006/gyno.1995.0008
Petru, Lax, Holzer et al (1998) Mesenteric and mesocolic lymph node metastases from ovarian carcinoma: a clinicopathological analysis. Int J Gynecol Cancer 8(2):119–123. https://doi.org/10.1046/j.1525-1438.1998.09794.x
Salani R, Diaz-Montes T, Giuntoli RL, Bristow RE (2007) Surgical management of mesenteric lymph node metastasis in patients undergoing rectosigmoid colectomy for locally advanced ovarian carcinoma. Ann Surg Oncol 14(12):3552–3557. https://doi.org/10.1245/s10434-007-9565-7
Ong MLH, Schofield JB (2016) Assessment of lymph node involvement in colorectal cancer. World J Gastrointest Surg 8(3):179. https://doi.org/10.4240/wjgs.v8.i3.179
Amin MB, Greene FL, Edge SB et al (2017) The eighth edition AJCC cancer staging manual: continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin 67(2):93–99. https://doi.org/10.3322/caac.21388
Author information
Authors and Affiliations
Contributions
RB and VAC conceived the study, participated in its design and coordination and drafted the manuscript; MM, GS, MDS, MM, FM and GG participated in the design and interpretation of the data; VC and LV participated in the design and coordination of the study and performed the measurement; MR participated in the design of the study and performed the statistical analysis; MC conceived of the study, and participated in its design and coordination and helped to draft the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Author Capozzi Vito Andrea declare that he has no conflict of interest. Author Roberto Berretta declare that he has no conflict of interest. Author Giulio Sozzi declare that he has no conflict of interest. Author Lavinia Volpi declare that she has no conflict of interest. Author Valentina Ceni declare that she has no conflict of interest. Author Mauro Melpignano declare that he has no conflict of interest. Author Giovanna Giordano declare that she has no conflict of interest. Author Federico Marchesi declare that he has no conflict of interest. Author Michela Monica declare that she has no conflict of interest. Author Maurizio Di Serio declare that he has no conflict of interest. Author Marcello Ceccaroni declare that he has no conflict of interest. Author Matteo Riccò declare that he has no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Berretta, R., Capozzi, V.A., Sozzi, G. et al. Prognostic role of mesenteric lymph nodes involvement in patients undergoing posterior pelvic exenteration during radical or supra-radical surgery for advanced ovarian cancer. Arch Gynecol Obstet 297, 997–1004 (2018). https://doi.org/10.1007/s00404-018-4675-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00404-018-4675-y