Abstract
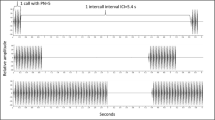
We investigated how communication distance influenced the efficacy of communication by studying the effects of two attributes of male chorus sounds, namely, reduction in sound level and degradation of temporal sound structure, on attraction and accuracy of female phonotaxis in gray treefrogs, Hyla versicolor. For this, we conducted acoustic playback experiments, using synthetic calls and natural calls recorded at increasing distances from a focal male as stimuli. We found that the degradation of temporal structure had a greater effect on signal attractiveness than did the reduction in sound level, and that increasing sound level preferentially affected the attractiveness of proximally recorded calls, with less temporal degradation. Unlike signal attraction, accuracy of female localization increased systematically with the sound level. These results suggest that the degradation of temporal fine structure from both the chorus and signal-environmental effects imposes a limit for effective communication distances for female treefrogs in nature.






Similar content being viewed by others
References
Aertsen AMHJ, Olders JHJ, Johannesma PIM (1981) Spectro-temporal receptive fields of auditory neurons in the grassfrog III. Analysis of the stimulus-event relation for natural stimuli. Hear Res 5:147–178
Bacon SP, Grantham DW (1989) Modulation masking: effects of modulation frequency, depth and noise. J Acoust Soc Am 85:2575–2580
Batschelet E (1981) Circular statistics in biology. Academic Press, London
Beckers O, Schul J (2004) Phonotaxis in Hyla versicolor (Anura, Hylidae): the effect of absolute call amplitude. J Comp Physiol A 190:869–876
Bee MA (2015) Treefrogs as animal models for research on auditory scene analysis and the cocktail party problem. Int J Psychophys 95:216–237
Bee MA, Micheyl C (2008) The cocktail party problem: what is it? How can it be solved? And why should animal behaviorists study it? J Comp Pyschol 122:235–251
Bee MA, Schwartz JJ (2009) Behavioral measures of signal recognition thresholds in frogs in the presence and absence of chorus-shaped noise. J Acoust Soc Am 126:2788–2801
Bee MA, Vélez A, Forester JD (2012) Sound level discrimination by Cope’s gray treefrog (Hyla chrysoscelis) in the presences and absence of chorus-shaped noise. J Acoust Sci Am 135:4188–4195
Brown J, Handford P (2000) Sound design for vocalizations: quality in the woods, consistency in the fields. Condor 102:81–92
Bush SL, Gerhardt HC, Schul J (2002) Pattern recognition and call preferences in treefrogs (Anura: Hylidae): a quantitative analysis using a no-choice paradigm. Anim Behav 63:7–14
Christie K, Schul J, Feng AS (2010) Phonotaxis to male’s calls embedded within a chorus by female gray treefrogs, Hyla versicolor. J Comp Physiol A 196:569–579
Cooke M (2006) A glimpsing model of speech perception in noise. J Acoust Sci Am 119:1562–1573
Diekamp B, Gerhardt HC (1995) Selective phonotaxis to advertisement calls in the gray treefrog Hyla versicolor: behavioral experiments and neurophysiological correlates. J Comp Physiol A 117:173–190
Edwards CJ, Alder TB, Rose GJ (2002) Auditory midbrain neurons that count. Nat Neurosci 5:934–936
Eggermont JJ (1990) Temporal modulation transfer functions for single neurons in the auditory midbrain of the leopard frog. Intensity and carrier-frequency dependence. Hear Res 43:181–198
Fay RR, Simmons AM (1999) The sense of hearing in fishes and amphibians. In: Fay RR, Popper AN (eds) Comparative hearing: fish and amphibians. Springer, New York, pp 269–318
Feng AS, Schul J (2007) Sound processing in real-world environments. In: Narins PM, Feng AS, Fay RR, Popper AN (eds) Hearing and sound communication in amphibians. Springer, New York, pp 323–350
Feng AS, Hall JC, Siddique S (1991) Coding of temporal parameters of complex sounds by frog auditory nerve fibers. J Neurophysiol 65:424–445
Fuzessery ZM, Feng AS (1982) Frequency selectivity in the anuran auditory midbrain: Single unit responses to single and multiple tone stimulation. J Comp Physiol A 146:471–484
Fuzessery ZM, Feng AS (1983) Frequency selectivity in the anuran medulla: Excitatory and inhibitory tuning properties of single neurons in the dorsal medullary and superior olivary nuclei. J Comp Physiol A 150:107–119
Gall MD, Wilczynski W (2014) Prior experience with conspecific signals enhances auditory midbrain responsiveness to conspecific vocalizations. J Exp Biol 217:1977–1982
Gall MD, Wilczynski W (2016) The effects of call-like masking diminish after nightly exposure to conspecific choruses in green treefrogs (Hyla cinerea). J Exp Biol 219:1295–1302
Gall MD, Bee MA, Baugh AT (2019) The difference a day makes: breeding remodels hearing hormones and behavior in female Cope’s gray treefrogs (Hyla chrysoscelis). Horm Behav 108:62–72
Gayou DC (1984) Effects of temperature on the mating call of Hyla versicolor. Copeia 1984:733–738
Gerhardt HC (1991) Female mate choice in treefrogs: static and dynamic acoustic criteria. Anim Behav 42:615–635
Gerhardt HC (2008) Phonotactic selectivity in two cryptic species of gray treefrogs: effects of differences in pulse rate, carrier frequency and playback level. J Exp Biol 211:2609–2616
Gerhardt HC, Doherty JA (1988) Acoustic communication in the gray treefrog, Hyla versicolor: evolutionary and neurobiological implications. J Comp Physiol A162:261–278
Gerhardt HC, Klump GM (1988) Phonotactic responses and selectivity of barking treefrogs (Hyla gratiosa) to chorus sounds. J Comp Physiol A 163:795–802
Gerhardt HC, Schul J (1999) A quantitative analysis of behavioral selectivity for pulse rise-time in the gray treefrog, Hyla versicolor. J Comp Physiol A 185:33–40
Gerhardt HC, Watson GF (1995) Within-male variability in call properties and female choice in the gray treefrog. Anim Behav 50:1187–1191
Gerhardt HC, Dyson ML, Tanner SD (1996) Dynamic properties of the advertisement calls of gray tree frogs: patterns of variability and female choice. Behav Ecol 7:7–18
Gerhardt HC, Tanner SD, Corrigan CM, Walton HC (2000) Female preference functions based on call duration in the gray tree frog (Hyla versicolor). Behav Ecol 11:663–669
Goense JBM, Feng AS (2012) Effects of noise bandwidth and amplitude modulation on masking in frog auditory midbrain neurons. PLoS One 7:e31589
Gooler DM, Feng AS (1992) Temporal coding in the frog auditory midbrain: the influence of duration and rise-fall time on the processing of complex amplitude-modulated stimuli. J Neurophysiol 67:1–22
Hall JC, Feng AS (1986) Neural analysis of temporally patterned sounds in the frog’s thalamus: processing of pulse duration and pulse repetition rate. Neurosci Lett 63:215–220
Hall JC, Feng AS (1988) Influence of envelope rise time on neural responses in the auditory system of anurans. Hear Res 36:261–276
Kime NM, Turner WR, Ryan MJ (2000) The transmission of advertisement calls in Central American frogs. Behav Ecol 11:71–83
Kuczynski MC, Vélez A, Schwartz JJ, Bee MA (2010) Sound transmission and the recognition of temporally degraded sexual advertisement signals in Cope’s gray treefrog (Hyla chrysoscelis). J Exp Biol 213:2840–2850
Lang F (2000) Acoustic communication distances of a Gomphocerine grasshopper. Bioacoustics 10:233–258
Leary CJ, Edwards CJ, Rose GJ (2008) Midbrain auditory neurons integrate excitation and inhibition to generate duration selectivity: an in vivo whole-cell patch study in anurans. J Neurosci 28: 5481–5493
Lee N, Ward JL, Vélez A, Micheyl C, Bee MA (2017) Frogs exploit statistical regularities in noisy acoustic scenes to solve cocktail-party-like problems. Curr Biol 24:743–750
Luther D, Gentry K (2013) Sources of background noise and their influence in vertebrate acoustic communication. Behaviour 150:1045–1068
Marshall VT, Schwartz JJ, Gerhardt HC (2006) Effects of heterospecific call overlap on the phonotactic behaviour of gray treefrogs. Anim Behav 72:449–459
Naguib M (2003) Reverberation of rapid and slow trills: implications for signal adaptations to long-range communication. J Acoust Soc Am 1131(3):1749–1756
Narins PM, Feng AS, Fay RR, Popper AN (2007) Hearing and sound communication in amphibians. Springer, New York
Ptacek MB, Gerhardt HC, Sage RD (1994) Speciation by polyploidy in treefrogs: multiple origins of the tetraploid, Hyla versicolor. Evolution 48:898–908
Richards DG, Wiley RH (1980) Reverberations and amplitude fluctuations in the propagation of sounds in a forest: implications for animal communication. Am Nat 115:381–399
Rose GJ, Hanson JL, Leary CJ, Graham JA, Alluri RK, Vasquez-Opazo GA (2015) Species-specificity of temporal processing in the auditory midbrain of gray treefrogs: interval-counting neurons. J Comp Physiol A 201:485–503
Ryan MJ, Keddy-Hector A (1992) Directional patterns of female mate choice and the role of sensory biases. Am Nat 139:S4–S35
Ryan MJ, Sullivan BK (1989) Transmission effects on patterns of amplitude modulation in the advertisement calls of two toads, Bufo woodhousii and Bufo valliceps. Ethology 80:182–189
Schrode KM, Bee MA (2015) Evolutionary adaptations for the temporal processing of natural sounds by the anuran peripheral auditory system. J Exp Biol 218:837–848
Schul J, Bush SL (2002) Non-parallel coevolution of sender and receiver in the acoustic communication system of treefrogs. Proc R Soc B 269:1847–1852
Schwartz JJ (1993) Male calling behavior, female discrimination and acoustic interference in the neotropical treefrog Hyla microcephala under realistic acoustic conditions. Behav Ecol Sociobiol 32:401–414
Schwartz JJ, Bee MA (2013) Anuran signal production in noisy environments. In: Brumm H (ed) Animal communication and noise. Springer, New York, pp 91–132
Schwartz JJ, Marshall VT (2006) Forms of call overlap and their impact on advertisement call attractiveness to females of the gray treefrog, Hyla versicolor. Bioacoustics 16:39–56
Schwartz JJ, Buchanan BW, Gerhardt HC (2001) Female mate choice in the gray treefrog (Hyla versicolor) in three experimental environments. Behav Ecol Sociobiol 49:443–455
Schwartz JJ, Huth K, Hunce R, Lentine B (2010) Effect of anomalous pulse timing on call discrimination by females of the gray treefrog (Hyla versicolor): behavioral correlates of neurobiology. J Exp Biol 213:2066–2072
Schwartz JJ, Crimarco NC, Bregman Y, Umeoji KR (2013) An investigation of the functional significance of responses of the gray treefrog (Hyla versicolor) to chorus noise. J Herp 47:354–360
Swanson EM, Tekman S, Bee MA (2007) Do female frogs exploit inadvertant social information to locate breeding aggregations? Can J Zool 85:921–932
Vélez A, Bee MA (2010) Signal recognition by frogs in the presence of temporally fluctuating chorus-shaped noise. Behav Ecol Sociobiol 64:1695–1709
Vélez A, Bee MA (2011) Dip listening and the cocktail party problem in grey treefrogs: signal recognition in temporally fluctuating noise. Anim Behav 82:1319–1327
Vélez A, Bee MA (2013) Signal recognition by green treefrogs (Hyla cinereai) and Cope’s gray treefrogs (Hyla chrysoscelis) in naturally fluctuating noise. J Comp Psychol 127:166–178
Vélez A, Höbel G, Gordon NH, Bee MA (2012) Dip listening or modulation masking? Call recognition by green treefrogs (Hyla cinerea) in temporally fluctuating noise. J Comp Physiol A 198:891–904
Vélez A, Linehan-Skillings BJ, Gu Y, Sun Y, Bee MA (2013a) Pulse-number discrimination by Cope’s gray treefrog (Hyla chrysoscelis) in modulated and unmodulated noise. J Acoust Sci Am 134:3079–3089
Vélez A. Schwartz JJ, Bee MA (2013b) Anural acoustic signal perception in noisy environments. In: Brumm H (ed) Animal communication and noise. Springer, New York, pp 133–185
Vélez A, Gordon N, Bee MA (2017) The signal in noise: acoustic information for soundscape orientation in two North American tree frogs. Behav Ecol 28(3):844–853
Vestergaard MD, Fyson NRC, Patterson RD (2011) The mutual roles of temporal glimpsing and vocal characteristics in cocktail-party listening. J Acoust Sci Am 130:429–439
Walkowiak W (1984) Neuronal correlates of the recognition of pulsed sound signals in the grass frog. J Comp Physiol A 155:57–66
Ward JL, Buerkle NP, Bee MA (2013a) Spatial release from masking improves sound pattern discrimination along a biologically relevant pulse-rate continuum in gray treefrogs. Hear Res 306:63–75
Ward JL, Love EK, Vélez A, Buerkle NP, O’Bryan LR, Bee MA (2013b) Multitasking males and multiplicative females: dynamic signaling and receiver preferences in Cope’s grey treefrog. Anim Behav 86:231–243
Wiley RH (2017) How noise determines the evolution of communication. Anim Behav 124:307–313
Wollerman L (1999) Acoustic interference limits call detection in a Neotropical frog Hyla ebraccata. Anim Behav 57:529–536
Wollerman L, Wiley RD (2002) Background noise from a natural chorus alters female discrimination of male calls in a Neotropical frog. Anim Behav 63:15–22
Zelick RD, Narins PM (1982) Analysis of acoustically evoked call suppression behaviour in a neotropical treefrog. Anim Behav 30:728–733
Zhang H, Feng AS (1998) Sound direction modifies the inhibitory as well as the excitatory frequency tuning characteristics of single neurons in the frog torus semicircularis (inferior colliculus). J Comp Physiol A 182:725–735
Acknowledgements
We thank the University of Missouri-Columbia and the Missouri Dept. of Conservation for site access and capture permits. Carl Gerhardt provided advice and material support, and Oliver Beckers, Sarah Bush, Noah Gordon and Sarah Humfeld provided valuable advice and comments. Paul Kimball, Stephanie Kleyman, and Leah Reynolds aided in animal collection and running experiments. Atulya Iyengar provided valuable spectral analysis advice, and Daniel Eberl and Bernd Fritsch provided indispensable advice, comments, and feedback on the manuscript. Research funding was provided by a National Science Foundation Collaborative Research in Computational Neuroscience (CRCNS-0422073) Grant to ASF and a National Science Foundation Grant (IOB-0445286) to JS. All experimental procedures were approved by the Institutional Animal Care and Use Committee of the University of Illinois.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Christie, K.W., Schul, J. & Feng, A.S. Differential effects of sound level and temporal structure of calls on phonotaxis by female gray treefrogs, Hyla versicolor. J Comp Physiol A 205, 223–238 (2019). https://doi.org/10.1007/s00359-019-01325-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-019-01325-5