Abstract
Background
We report on our multicentre evaluation of the occurrence and management of complications with the ATOMS device (A.M.I., Austria).
Patients and methods
Between 10/09 and 09/18, a total of 187 patients with persistent postoperative stress urinary incontinence received an ATOMS device in Madrid (n = 101) or Halle (n = 86). This prospective evaluation was carried out consecutively. In addition to complications, patient age, BMI, comorbidities and previous treatments for prostate cancer and urinary incontinence were examined. Statistical analysis was performed with GraphPad Prism 7® (GraphPad Software, Inc., La Jolla, USA); p < 0.05 was considered significant.
Results
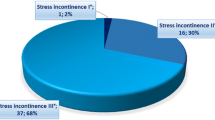
The average age of the overall population was 69.4 years, with a mean CCI of 4.3. In total, 18.2% of the patients had diabetes mellitus, 21.9% were irradiated, and 14.1% and 22.9% underwent previous surgery for urinary incontinence and urethral stricture, respectively. The overall success rate of the device was 80.2%. A total of 51 postoperative complications (51/187, 27.3%) were observed, with 18 grade I (9.6%), 1 grade II (0.5%), 3 grade IIIa (1.6%) and 29 grade IIIb (15.5%) complications. The most serious postoperative complications were primary wound infection (8/187, 4.3%) and long-term cutaneous erosion with subsequent infection (15/187, 8.02%). The main risk factors for complications were previous radiotherapy (p = 0.003) and surgery for urethral stricture (p = 0.017). No other parameters were significant in bivariate analysis (p > 0.05).
Conclusion
The most severe complications of ATOMS implantation were primary wound infection and cutaneous erosion, especially in the presence of previous radiotherapy. Particular focus on performing hygienic and accurate implantation techniques is recommended.


Similar content being viewed by others
References
Seweryn J, Bauer W, Ponholzer A, Schramek P (2012) Initial experience and results with a new adjustable transobturator male system for the treatment of stress urinary incontinence. J Urol 187(3):956–961
Hoda MR, Primus G, Fischereder K, Von Heyden B, Mohammed N, Schmid N, Moll V, Hamza A, Karsch JJ, Brössner C, Fornara P, Bauer W (2013) Early results of a european multicentre experience with a new self-anchoring adjustable transobturator system for treatment of stress urinary incontinence in men. BJU Int 111:296–303
Friedl A, Mühlstädt S, Zachoval R, Giammò A, Kivaranovic D, Rom M, Fornara P, Brössner C (2017) Long-term outcome of the adjustable transobturator male system (ATOMS): results of a European multicentre study. BJU Int 119(5):785–792
Mühlstädt S, Friedl A, Zachoval R, Mohammed N, Schumann A, Theil G, Fornara P (2019) An overview of the ATOMS generations: port types, functionality and risk factors. World J Urol 37(8):1679–1686
Angulo JC, Cruz F, Esquinas C, Arance I, Manso M, Rodríguez A, Pereira J, Ojea A, Carballo M, Rabassa M, Teyrouz A, Escribano G, Rodríguez E, Teba F, Celada G, Madurga B, Álvarez-Ossorio JL, Marcelino JP, Martins FE (2018) Treatment of male stress urinary incontinence with the adjustable transobturator male system: outcomes of a multi-center Iberian study. Neurourol Urodyn 37(4):1458–1466
Esquinas C, Angulo JC (2019) Effectiveness of adjustable transobturator male system (ATOMS) to treat male stress incontinence: a systematic review and meta-analysis. Adv Ther 36(2):426–441
Friedl A, Mühlstädt S, Rom M, Kivaranovic D, Mohammed N, Fornara P, Brössner C (2017) Risk factors for treatment failure with the adjustable transobturator male system incontinence device: Who will succeed, who will fail? Results of a multicenter study. Urology 90:189–194
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications. Ann Surg 240(2):205–213
Fuchs JS, Shakir N, McKibben MJ, Scott JM, Morey AF (2018) Prolonged duration of incontinence for men before initial anti-incontinence surgery: an opportunity for improvement. Urology 119:149–154
Hüsch T, Kretschmer A, Thomsen F, Kronlachner D, Kurosch M, Obaje A, Anding R, Kirschner-Hermanns R, Pottek T, Rose A, Olianas R, Lusuardi L, Friedl A, Homberg R, Pfitzenmaier J, Queissert F, Naumann CM, Schweiger J, Wotzka C, Nyarangi-Dix J, Brehmer B, Abdunnur R, Loertzer H, Ulm K, Hübner W, Bauer RM, Haferkamp A (2018) The AdVance and AdVanceXP male sling in urinary incontinence: is there a difference? World J Urol 36(10):1657–1662
Mühlstädt S, Hüsch T, Bauer RM (2019) Differentiated surgical treatment of male stress urinary incontinence-between intention and reality? Urol A. https://doi.org/10.1007/s00120-019-0947-9(Epub ahead of print)
Angulo JC, Arance I, Ojea A, Carballo M, Rodríguez A, Pereira J, Rebassa M, Teyrouz A, Escribano G, Teba F, Madurga B, Martins FE, Cruz F (2019) Patient satisfaction with adjustable transobturator male system in the Iberian multicenter study. World J Urol. https://doi.org/10.1007/s00345-019-02639-4(Epub ahead of print)
Mühlstädt S, Friedl A, Mohammed N, Schumann A, Weigand K, Kawan F, Göllert C, Kahlert C, Theil G, Fischer K, Fornara P (2017) Five-year experience with the adjustable transobturator male system for the treatment of male stress urinary incontinence: a single-center evaluation. World J Urol 35(1):145–151
Bauer RM, Soljanik I, Füllhase C, Buchner A, May F, Stief CG, Gozzi C (2011) Results of the AdVance transobturator male sling after radical prostatectomy and adjuvant radiotherapy. Urology 77(2):474–479
Bauer RM, Rutkowski M, Kretschmer A, Casuscelli J, Stief CG, Huebner W (2015) Efficacy and complications of the adjustable sling system ArgusT for male incontinence: results of a prospective 2-center study. Urology 85(2):316–320
Léon P, Chartier-Kastler E, Rouprêt M, Ambrogi V, Mozer P, Phé V (2015) Long-term functional outcomes after artificial urinary sphincter implantation in men with stress urinary incontinence. BJU Int 115(6):951–957
Kretschmer A, Hüsch T, Thomsen F, Kronlachner D, Obaje A, Anding R, Pottek T, Rose A, Olianas R, Friedl A, Hübner W, Homberg R, Pfitzenmaier J, Grein U, Queissert F, Naumann CM, Schweiger J, Wotzka C, Nyarangi-Dix JN, Hofmann T, Seiler R, Haferkamp A, Bauer RM, Debates On Male Incontinence (DOMINO)-Project (2016) Complications and short-term explantation rate following artificial urinary sphincter implantation: results from a large middle European multi-institutional case series. Urol Int 97(2):205–211
Lee D, Chang J, Zimmern PE (2017) Iatrogenic pelvic pain: surgical and mesh complications. Phys Med Rehabil Clin N Am 28(3):603–619
Rice NT, Hu Y, Slaughter JC, Ward RM (2013) Pelvic mesh complications in women before and after the 2011 FDA public health notification. Female Pelvic Med Reconstr Surg 19(6):333–338
Rehder P, Haab F, Cornu JN, Gozzi C, Bauer RM (2012) Treatment of postprostatectomy male urinary incontinence with the transobturator retroluminal repositioning sling suspension: 3-year follow up. Eur Urol 62:140–145
Kowalik CG, DeLong JM, Mourtzinos AP (2015) The advance transobturator male sling for post-prostatectomy incontinence: subjective and objective outcomes with 3 years follow up. Neurourol Urodyn 34(3):251–254
Bauer RM, Grabbert MT, Klehr B, Gebhartl P, Gozzi C, Homberg R, May F, Rehder P, Stief CG, Kretschmer A (2017) 36-month data for the AdVance XP® male sling: results of a prospective multicentre study. BJU Int 119(4):626–630
Hübner WA, Gallistl H, Rutkowski M, Huber ER (2010) Adjustable bulbourethral male sling: experience after 101 cases of moderate-to-severe male stress urinary incontinence. BJU Int 107:777–782
Sousa-Escandón A, Cabrera J, Mantovani F, Moretti M, Ioanidis E, Kondelidis N, Neymeyer J, Noguera R (2007) Adjustable suburethral sling (male Remeex system) in the treatment of male stress urinary incontinence: a multicentric European study. Eur Urol 52(5):1473–1479
Bauer RM, Rutkowski M, Kretschmer A, Casuscelli J, Stief CG, Huebner W (2015) Efficacy and complications of the adjustable sling system ArgusT for male incontinence: results of a prospective 2-center study. Urology 85(2):316–320
Romano SV, Huebner W, Rocha FT, Vaz FP, Muller V, Nakamura F (2015) A transobturator adjustable system for male incontinence: 30-month follow-up of a multicenter study. Int Braz J Urol 40:781–789
Zheng X, Narayanan S, Sunkari VG, Eliasson S, Botusan IR, Grünler J, Catrina AI, Radtke F, Xu C, Zhao A, Ekberg NR, Lendahl U, Catrina SB (2019) Triggering of a Dll4-Notch1 loop impairs wound healing in diabetes. Proc Natl Acad Sci USA 116(14):6985–6994
Cornel EB, Elzevier HW, Putter H (2010) Can advance transobturator sling suspension cure male urinary postoperative stress incontinence? J Urol 183:1459–1463
Author information
Authors and Affiliations
Contributions
SM: protocol development, data collection and analysis, and manuscript writing, JCA: data collection and analysis, and manuscript writing. NM: manuscript editing. AS: manuscript editing. PF: protocol development and manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
Ethical approval was obtained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mühlstädt, S., Angulo, J.C., Mohammed, N. et al. Complications of the urinary incontinence system ATOMS: description of risk factors and how to prevent these pitfalls. World J Urol 38, 1795–1803 (2020). https://doi.org/10.1007/s00345-019-02962-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-019-02962-w