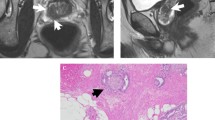
Abstract
Objective
The aim of this study was to evaluate three indirect MRI signs for predicting extraprostatic disease in patients referred to radical prostatectomy: index tumor volume (MTV), apparent diffusion coefficient (ADC) and tumor contact length (TCL).
Materials and methods
This prospective study included 183 patients with biopsy proven prostate cancer. In all patients the MTV (ml), ADC (× 10−5 mm2/s) and TCL (mm) of the index tumor were registered at the preoperative MRI. Whole-mounted microscopical examination classified each patient as having either localized- or extraprostatic disease. The Youden index was used to identify the optimal cut-off values for predicting extraprostatic disease. Univariate regression analyses were conducted to estimate the odds ratio (OR) with 95% confidence intervals (CI). Results were stratified upon zonal location of the index tumor.
Results
Extraprostatic disease was identified in 103 (56%) patients. The risk of extraprostatic disease was nine times higher in peripheral zone tumors with ADC ≤ 89 (OR 9.1, 95% CI 4.2–19.6), five times higher in MTV ≥ 0.9 ml (OR 5.5, 95% CI 2.6–11.4) and five times higher in case of TCL ≥ 14 mm (OR 4.9, 95% CI 2.3–10.2). None of the indirect MRI signs could predict extraprostatic disease for transition zone tumors.
Conclusion
The MTV, ADC and TCL are all significant predictors of extraprostatic disease for peripheral zone tumors, while none of the indirect signs were useful for transition zone tumors.






Similar content being viewed by others
References
Rud E, Klotz D, Rennesund K et al (2014) Preoperative magnetic resonance imaging for detecting uni- and bilateral extraprostatic disease in patients with prostate cancer. World J Urol. https://doi.org/10.1007/s00345-014-1362-x
Obek C, Louis P, Civantos F, Soloway MS (1999) Comparison of digital rectal examination and biopsy results with the radical prostatectomy specimen. JURO 161:494–498 (discussion 498–9)
de Rooij M, Hamoen EHJ, Witjes JA et al (2015) Accuracy of magnetic resonance imaging for local staging of prostate cancer: a diagnostic meta-analysis. Eur Urol. https://doi.org/10.1016/j.eururo.2015.07.029
Weinreb JC, Barentsz JO, Choyke PL et al (2016) PI-RADS prostate imaging—reporting and data system: 2015, version 2. Eur Urol 69:16–40. https://doi.org/10.1016/j.eururo.2015.08.052
Yu KK, Hricak H, Alagappan R et al (1997) Detection of extracapsular extension of prostate carcinoma with endorectal and phased-array coil MR imaging: multivariate feature analysis. Radiology 202:697–702. https://doi.org/10.1148/radiology.202.3.9051019
Outwater EK, Petersen RO, Siegelman ES et al (1994) Prostate carcinoma: assessment of diagnostic criteria for capsular penetration on endorectal coil MR images. Radiology 193:333–339. https://doi.org/10.1148/radiology.193.2.7972739
Kim KH, Lim SK, Shin T-Y et al (2013) Tumor volume adds prognostic value in patients with organ-confined prostate cancer. Ann Surg Oncol 20:3133–3139. https://doi.org/10.1245/s10434-013-3016-4
Rud E, Klotz D, Rennesund K et al (2014) Detection of the index tumor and tumor volume in prostate cancer using T2w and DW MRI alone. BJU Int. https://doi.org/10.1111/bju.12637
Baco E, Rud E, Vlatkovic L et al (2014) Predictive value of magnetic resonance imaging determined tumor contact length for extra-capsular extension of prostate cancer. J Urol. https://doi.org/10.1016/j.juro.2014.08.084
Rosenkrantz AB, Shanbhogue AK, Wang A et al (2016) Length of capsular contact for diagnosing extraprostatic extension on prostate MRI: assessment at an optimal threshold. J Magn Reson Imaging 43:990–997. https://doi.org/10.1002/jmri.25040
Bratan F, Melodelima C, Souchon R et al (2014) How accurate is multiparametric MR imaging in evaluation of prostate cancer volume? Radiology. https://doi.org/10.1148/radiol.14140524
Hambrock T, Somford DM, Huisman HJ et al (2011) Relationship between apparent diffusion coefficients at 3.0-T MR imaging and Gleason grade in peripheral zone prostate cancer. Radiology 259:453–461. https://doi.org/10.1148/radiol.11091409
Itou Y, Nakanishi K, Narumi Y et al (2011) Clinical utility of apparent diffusion coefficient (ADC) values in patients with prostate cancer: can ADC values contribute to assess the aggressiveness of prostate cancer? J Magn Reson Imaging 33:167–172. https://doi.org/10.1002/jmri.22317
Rud E, Baco E, Eggesbø HB (2012) MRI and ultrasound-guided prostate biopsy using soft image fusion. Anticancer Res 32:3383–3389
Magi-Galluzzi C, Evans AJ, Delahunt B et al (2010) International Society of Urological Pathology (ISUP) consensus conference on handling and staging of radical prostatectomy specimens. Working group 3: extraprostatic extension, lymphovascular invasion and locally advanced disease. Mod Pathol 24:26–38. https://doi.org/10.1038/modpathol.2010.158
Lim C, Flood TA, Hakim SW et al (2016) Evaluation of apparent diffusion coefficient and MR volumetry as independent associative factors for extra-prostatic extension (EPE) in prostatic carcinoma. J Magn Reson Imaging 43:726–736. https://doi.org/10.1002/jmri.25033
Kongnyuy M, Sidana A, George AK et al (2017) Tumor contact with prostate capsule on magnetic resonance imaging: a potential biomarker for staging and prognosis. Urol Oncol 35:30.e1–30.e8. https://doi.org/10.1016/j.urolonc.2016.07.013
Granja MF, Pedraza CM, Flórez DC et al (2017) Predicting extracapsular involvement in prostate cancer through the tumor contact length and the apparent diffusion coefficient. Radiologia 59:313–320. https://doi.org/10.1016/j.rx.2017.03.003
Ranstam J (2008) Analysis units. Acta Radiol 49:371–372. https://doi.org/10.1080/02841850801977312
Woo S, Cho JY, Kim SY, Kim SH (2015) Extracapsular extension in prostate cancer: added value of diffusion-weighted MRI in patients with equivocal findings on T2-weighted imaging. Am J Roentgenol 204:W168–W175. https://doi.org/10.2214/AJR.14.12939
Kim CK, Park SY, Park JJ, Park BK (2014) Diffusion-weighted MRI as a predictor of extracapsular extension in prostate cancer. Am J Roentgenol 202:W270–W276. https://doi.org/10.2214/AJR.13.11333
Rosenkrantz AB, Chandarana H, Gilet A et al (2013) Prostate cancer: utility of diffusion-weighted imaging as a marker of side-specific risk of extracapsular extension. J Magn Reson Imaging 38:312–319. https://doi.org/10.1002/jmri.23972
Lawrence EM, Gallagher FA, Barrett T et al (2014) Preoperative 3-T diffusion-weighted MRI for the qualitative and quantitative assessment of extracapsular extension in patients with intermediate- or high-risk prostate cancer. Am J Roentgenol 203:W280–W286. https://doi.org/10.2214/AJR.13.11754
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Rud, E., Diep, L. & Baco, E. A prospective study evaluating indirect MRI-signs for the prediction of extraprostatic disease in patients with prostate cancer: tumor volume, tumor contact length and tumor apparent diffusion coefficient. World J Urol 36, 629–637 (2018). https://doi.org/10.1007/s00345-018-2171-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-018-2171-4