Abstract
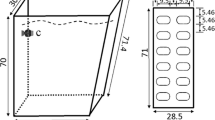
Turbulent mixing, in particular on a small scale, affects the growth of microalgae by changing diffusive sublayers and regulating nutrient fluxes of cells. We tested the nutrient flux hypothesis by evaluating the cellular stoichiometry and phosphorus storage of microalgae under different turbulent mixing conditions. Aphanizomenon flos-aquae were cultivated in different stirring batch reactors with turbulent dissipation rates ranging from 0.001 51 m2/s3 to 0.050 58 m2/s3, the latter being the highest range observed in natural aquatic systems. Samples were taken in the exponential growth phase and compared with samples taken when the reactor was completely stagnant. Results indicate that, within a certain range, turbulent mixing stimulates the growth of A. flos-aquae. An inhibitory effect on growth rate was observed at the higher range. Photosynthesis activity, in terms of maximum effective quantum yield of PSII (the ratio of Fv/Fm) and cellular chlorophyll a, did not change significantly in response to turbulence. However, Chl a/C mass ratio and C/N molar ratio, showed a unimodal response under a gradient of turbulent mixing, similar to growth rate. Moreover, we found that increases in turbulent mixing might stimulate respiration rates, which might lead to the use of polyphosphate for the synthesis of cellular constituents. More research is required to test and verify the hypothesis that turbulent mixing changes the diffusive sublayer, regulating the nutrient flux of cells.
Similar content being viewed by others
References
Ayata S D, Lévy M, Aumont O, Resplandy L, Tagliabue A, Sciandra A, Bernard O. 2014. Phytoplankton plasticity drives large variability in carbon fixation efficiency. Geophys. Res. Lett., 41(24): 8994–9000.
Dickman E M, Vanni M J, Horgan M J. 2006. Interactive effects of light and nutrients on phytoplankton stoichiometry. Oecologia, 149(4): 676–689.
Ebert U, Arrayás M, Temme N, Sommeijer B, Huisman J. 2001. Critical conditions for phytoplankton blooms. Bull. Math. Biol., 63(6): 1095–1124.
Eixler S, Karsten U, Selig U. 2006. Phosphorus storage in Chlorella vulgaris (Trebouxiophyceae, Chlorophyta) cells and its dependence on phosphate supply. Phycologia, 45(1): 53–60.
Eixler S, Selig U, Karsten U. 2005. Extraction and detection methods for polyphosphate storage in autotrophic planktonic organisms. Hydrobiologia, 533(1-3): 135–143.
Elliott J A. 2010. The seasonal sensitivity of Cyanobacteria and other phytoplankton to changes in flushing rate and water temperature. Global Change Biol., 16(2): 864–876.
Estrada M, Berdalet E. 1997. Phytoplankton in a turbulent world. Scientia Marina, 61 (S1): 125–140.
Gallardo Rodríguez J J, Sánchez Mirón A, García Camacho F, Cerón García M C, Belarbi E H, Chisti Y, Molina Grima E. 2009. Causes of shear sensitivity of the toxic dinoflagellate Protoceratium reticulatum. Biote chnology Progress, 25(3): 792–800.
Guillard R R L. 1973. Division rates. In: Stein J R ed. Handbook of Phycological Methods. I. Culture Methods and Growth Measurements. Cambridge University Press, Cambridge. p.289–312.
Halsey K H, Jones B M. 2015. Phytoplankton strategies for photosynthetic energy allocation. Annu. Rev. Mar. Sci., 7(1): 265–297.
Halsey K H, Milligan A J, Behrenfeld M J. 2014. Contrasting strategies of photosynthetic energy utilization drive lifestyle strategies in ecologically important picoeukaryotes. Metabolites, 4(2): 260–280.
Hondzo M, Lyn D. 1999. Quantified small-scale turbulence inhibits the growth of a green alga. Freshwater Biol ogy, 41(1): 51–61.
Hondzo M, Wüest A. 2009. Do Microscopic organisms feel turbulent flows? Environ. Sci. Technol., 43(3): 764–768.
Huisman J, Arrayás M, Ebert U, Sommeijer B. 2002. How do sinking phytoplankton species manage to persist? Am. Nat., 159(3): 245–254.
Karp-Boss L, Boss E, Jumars P A. 1996. Nutrient fluxes to planktonic osmotrophs in the presence of fluid motion. Oceanogr. Mar. Biol. Annu. Rev, 34: 71–107.
Klausmeier C A, Litchman E, Daufresne T, Levin S A. 2008. Phytoplankton stoichiometry. Ecol. Res., 23(3): 479–485.
Leupold M, Hindersin S, Gust G, Kerner M, Hanelt D. 2013. Influence of mixing and shear stress on Chlorella vulgaris, Scenedesmus obliquus, and Chlamydomonas reinhardtii. J. Appl. Phycol., 25(2): 485–495.
Litchman E, Klausmeier C A. 2008. Trait-based community ecology of phytoplankton. Ann. Rev. Ecol. Evol. Syst., 39(1): 615–639.
Maxwell K, Johnson G N. 2000. Chlorophyll fluorescence—a practical guide. J. Exp. Bot., 51(345): 659–668.
Michels M H A, van der Goot A J, Norsker N H, Wijffels R H. 2010. Effects of shear stress on the microalgae Chaetoceros muelleri. Bioproc. Biosyst. Eng., 33(8): 921–927.
Montechiaro F, Giordano M. 2010. Compositional homeostasis of the dinoflagellate Protoceratium reticulatum grown at three different pCO2. J. Plant Physiol., 167(2): 110–113.
O’Halloran I P, Cade-Menun B J. 2008. Total and organic phosphorus. In: Carter M R, Gregorich E G eds. Soil Sampling and Methods of Analysis. 2 nd edn. CRC Press, Boca Raton.
Padisák J, Köhler J, Hoeg S. 1999. Effect of changing flushing rates on development of late summer Aphanizomenon and Microcystis populations in a shallow lake, Müggelsee, Berlin, Germany. In: Tundisi J G, Straškraba M eds. Theoretical Reservoir Ecology and Its Applications. Backhuys Publishers, Leiden. p.411–424.
Parkinson J A, Allen S E. 1975. A wet oxidation procedure suitable for the determination of nitrogen and mineral nutrients in biological material. Commun. Soil Sci. Plan t Anal., 6(1): 1–11.
Powell N, Shilton A N, Pratt S, Chisti Y. 2008. Factors influencing luxury uptake of phosphorus by microalgae in waste stabilization ponds. Environ. Sci. Technol., 42(16): 5958–5962.
Powell N, Shilton A, Chisti Y, Pratt S. 2009. Towards a luxury uptake process via microalgae-defining the polyphosphate dynamics. Water Res., 43(17): 4207–4213.
Reynolds C S. 2006. The Ecology of Phytoplankton. Cambridge University Press, Cambridge.
Rothschild B J, Osborn T R. 1988. Small-scale turbulence and plankton contact rates. J. Plankton Res., 10(3): 465–474.
Sterner R W, Elser J J. 2002. Ecological Stoichiometry: The Biology of Elements from Molecules to the Biosphere. Princeton University Press, Princeton.
Thomas W H, Gibson C H. 1990. Effects of small-scale turbulence on microalgae. J. Appl. Phycol., 2(1): 71–77.
Thomas W H, Vernet M, Gibson C H. 1995. Effects of smallscale turbulence on photosynthesis, pigmentation, cell division, and cell size in the marine dinoflagellate Gomaulax polyedra (Dinophyceae). J. Phycol., 31(1): 50–59.
Tripathi K, Sharma N K, Kageyama H, Takabe T, Rai A K. 2013. Physiological, biochemical and molecular responses of the halophilic cyanobacterium Aphanothece halophytica to Pi-deficiency. Eur. J. Phycol., 48(4): 461–473.
Warnaars T A, Hondzo M. 2006. Small-scale fluid motion mediates growth and nutrient uptake of Selenastrum capricornutum. Freshwater Biol ogy, 51(6): 999–1015.
Wintermans J F G M, de Mots A. 1965. Spectrophotometric characteristics of chlorophylls a and b and their phenophytins in ethanol. Biochimica et Biophysica Acta (BBA)-Biophysics including Photosynthesis, 109(2): 448–453.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by the National Natural Science Foundation of China (Nos. 51309220, 51679226), the National Key SandT Project on Water Pollution Control and Treatment (Nos. 2014ZX07104-006, 2015ZX07103-007), and the Western Action Program funded by the Chinese Academy of Sciences (No. KZCX2-XB3-14)
Rights and permissions
About this article
Cite this article
Li, Z., Xiao, Y., Yang, J. et al. Response of cellular stoichiometry and phosphorus storage of the cyanobacteria Aphanizomenon flos-aquae to small-scale turbulence. Chin. J. Ocean. Limnol. 35, 1409–1416 (2017). https://doi.org/10.1007/s00343-017-6178-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00343-017-6178-2