Abstract
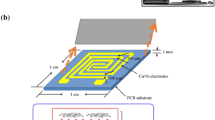
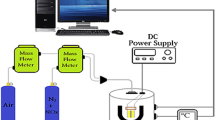
Sulfur dioxide (SO2) is prominent as hazardous gas owing to its unpropitious effects on the ecosystem. In this report, a flexible SO2 gas sensor is reported by solvothermally synthesized crystalline nickel(II)benzenetricarboxylate metal–organic framework (Ni-MOF) modified with hydroxyl group (–OH) activated single wall carbon nanotubes (SWNTs) and multi-walled carbon nanotubes (MWNTs), respectively. Introduction of –OH-SWNTs and –OH–MWNTs played crucial role in the improvement in electrical and morphological properties of Ni-MOF as well as boosted the sensing ability toward SO2 gas at room temperature. The structural and spectroscopy properties of pristine Ni-MOF, Ni-MOF/–OH–SWNTs and Ni-MOF/–OH-MWNTs were studied by X-Ray diffraction (XRD) and Fourier-transform infrared spectroscopy (FTIR), respectively. Atomic force microscopy (AFM) and field emission scanning electron microscope (FESEM) were used for the morphological analysis of synthesized material. The selective response of Ni-MOF/–OH-SWNTs and Ni-MOF/–OH-MWNTs toward SO2, NO2, NH3 and CO analytes (0.5–15 ppm) was withal studied by monitoring the changes in electrical resistance of the material at room temperature. The present study reveals that doping of –OH-SWNTs and –OH-MWNTs into the MOF leads to efficient increment in the sensing characteristics. The composite of Ni-MOF/–OH-SWNTs exhibited better sensing response (10 s) with less recovery time (30 s) for 1 ppm concentration along with considerable sensitivity (0.9784) and selectivity toward SO2 gas.






Similar content being viewed by others
References
S. Kulkarni, Y. Navale, S. Navale, F. Stadler, N. Ramgir, V. Patil, Hybrid polyaniline-WO3 flexible sensor: a room temperature competence towards NH3 gas. Sensors Actuators B Chem. 288, 279–288 (2019)
P.-G. Su, Z.-H. Liao, Fabrication of a flexible single-yarn NH3 gas sensor by layer-by-layer self-assembly of graphene oxide. Mater. Chem. Phys. 224, 349–356 (2019)
Y. Fu, J. Li, H. Xu, SnO2 recycled from tin slime for enhanced SO2 sensing properties by NiO surface decoration. Mater. Sci. Semicond. Process. 114, 105073 (2020)
P.W. Sayyad, S.S. Khan, N.N. Ingle, G.A. Bodkhe, T. Al-Gahouari, M.M. Mahadik, S.M. Shirsat, M.D. Shirsat, Chemiresistive SO 2 sensor: graphene oxide (GO) anchored poly (3, 4-ethylenedioxythiophene): poly (4styrenesulfonate)(PEDOT: PSS). Appl. Phys. A 126, 1–8 (2020)
T. Al-Gahouari, G. Bodkhe, P. Sayyad, N. Ingle, M. Mahadik, S.M. Shirsat, M. Deshmukh, N. Musahwar, M. Shirsat, Electrochemical sensor: L-cysteine induced selectivity enhancement of electrochemically reduced graphene oxide-multiwalled carbon nanotubes hybrid for detection of lead (Pb2+) ions. Front. Mater. 7, 68 (2020)
I. Obot, I.B. Onyeachu, S.A. Umoren, M.A. Quraishi, A.A. Sorour, T. Chen, N. Aljeaban, Q. Wang, High temperature sweet corrosion and inhibition in the oil and gas industry: progress, challenges and future perspectives. J. Petrol. Sci. Eng. 185, 106469 (2020)
M. Mahadik, H. Patil, G. Bodkhe, N. Ingle, P. Sayyad, T. Al-Gahaouri, S.M. Shirsat, M. Shirsat, EDTA modified PANI/GO composite based detection of Hg (II) ions. Front. Mater. 7, 81 (2020)
K. Park, S. Choi, H.Y. Chae, C.S. Park, S. Lee, Y. Lim, H. Shin, J.J. Kim, An energy-efficient multimode multichannel gas-sensor system with learning-based optimization and self-calibration schemes. IEEE Trans. Industr. Electron. 67, 2402–2410 (2019)
Zaza, F.; Pallozzi, V.; Serra, E. Optimization of working conditions for perovskite-based gas sensor devices by multiregression analysis. J. Nanotechnol. 2019.
L. Zhu, W. Zeng, Room-temperature gas sensing of ZnO-based gas sensor: a review. Sens. Actuators A 267, 242–261 (2017)
K. Eslami Jahromi, Q. Pan, A. Khodabakhsh, C. Sikkens, P. Assman, S.M. Cristescu, P.M. Moselund, M. Janssens, B.E. Verlinden, F.J. Harren, A broadband mid-infrared trace gas sensor using supercontinuum light source: applications for real-time quality control for fruit storage. Sensors 19, 2334 (2019)
Vashpanov, Y.; Son, J.-Y.; Heo, G.; Kwack, K.-D. In Tilte2020; IOP Publishing.
M.D. Shirsat, M.A. Bangar, M.A. Deshusses, N.V. Myung, A. Mulchandani, Polyaniline nanowires-gold nanoparticles hybrid network based chemiresistive hydrogen sulfide sensor. Appl. Phys. Lett. 94, 083502 (2009)
Y. Seekaew, A. Wisitsoraat, D. Phokharatkul, C. Wongchoosuk, Room temperature toluene gas sensor based on TiO2 nanoparticles decorated 3D graphene-carbon nanotube nanostructures. Sensors Actuators B Chem. 279, 69–78 (2019)
Y. Deng, Sensing Mechanism and Evaluation Criteria of Semiconducting Metal Oxides Gas Sensors (Springer, In Semiconducting Metal Oxides for Gas Sensing, 2019), pp. 23–51
H. Kharat, K. Kakde, P. Savale, K. Datta, P. Ghosh, M. Shirsat, Synthesis of polypyrrole films for the development of ammonia sensor. Polym. Adv. Technol. 18, 397–402 (2007)
F. Loghin, A. Abdellah, A. Falco, M. Becherer, P. Lugli, A. Rivadeneyra, Time stability of carbon nanotube gas sensors. Measurement 136, 323–325 (2019)
P.W. Sayyad, N.N. Ingle, T. Al-Gahouari, M.M. Mahadik, G.A. Bodkhe, S.M. Shirsat, M.D. Shirsat, Sensitive and selective detection of Cu2+ and Pb2+ ions using Field Effect Transistor (FET) based on L-Cysteine anchored PEDOT: PSS/rGO composite. Chem. Phys. Lett. 761, 138056 (2020)
M. Patil, V. Ganbavle, K. Rajpure, H. Deshmukh, S. Mujawar, Fast response and highly selective nitrogen dioxide gas sensor based on Zinc Stannate thin films. Mater. Sci. Energy Technol. 3, 36–42 (2020)
G. Neri, A. Bonavita, C. Milone, S. Galvagno, Role of the Au oxidation state in the CO sensing mechanism of Au/iron oxide-based gas sensors. Sensors Actuators B Chem. 93, 402–408 (2003)
R.K. Jha, V. Singh, J. Sinha, S. Avasthi, N. Bhat, CVD grown cuprous oxide thin film based high performance chemiresistive ammonia gas sensors. IEEE Sens. J. 19, 11759–11766 (2019)
M.A. Deshmukh, H.K. Patil, G.A. Bodkhe, M. Yasuzawa, P. Koinkar, A. Ramanavicius, S. Pandey, M.D. Shirsat, EDA modified PANI/SWNTs nanocomposite for determination of Ni (II) metal ions. Colloids Surf. A 537, 303–309 (2018)
M.A. Deshmukh, H.K. Patil, G.A. Bodkhe, M. Yasuzawa, P. Koinkar, A. Ramanaviciene, M.D. Shirsat, A. Ramanavicius, EDTA-modified PANI/SWNTs nanocomposite for differential pulse voltammetry based determination of Cu (II) ions. Sensors Actuators B Chem. 260, 331–338 (2018)
W. Zhang, S. Cao, Z. Wu, M. Zhang, Y. Cao, J. Guo, F. Zhong, H. Duan, D. Jia, High-performance gas sensor of Polyaniline/Carbon nanotube composites promoted by interface engineering. Sensors 20, 149 (2020)
T. Seesaard, T. Kerdcharoen, C. Wongchoosuk, Hybrid Materials with Carbon Nanotubes for Gas Sensing (Elsevier, In Semiconductor Gas Sensors, 2020), pp. 185–222
H.K. Patil, M.A. Deshmukh, G.A. Bodkhe, S.M. Shirsat, K. Asokan, M.D. Shirsat, Reinforcement of polyaniline and poly-(o-toluidine) with SWNTs and tuning of their physicochemical properties by heavy ion beams. Appl. Phys. A 124, 491 (2018)
Reed, D.; Xiao, D. J.; Jiang, H. Z.; Chakarawet, K.; Oktawiec, J.; Long, J. R. Biomimetic O2 adsorption in an iron metal–organic framework for air separation. Chem. Sci. 2020.
G.A. Bodkhe, M.A. Deshmukh, H.K. Patil, S.M. Shirsat, V. Srihari, K.K. Pandey, G. Panchal, D.M. Phase, A. Mulchandani, M.D. Shirsat, Field effect transistor based on proton conductive metal organic framework (CuBTC). J. Phys. D Appl. Phys. 52, 335105 (2019)
D. Li, M. Kassymova, X. Cai, S.-Q. Zang, H.-L. Jiang, Photocatalytic CO2 reduction over metal-organic framework-based materials. Coord. Chem. Rev. 412, 213262 (2020)
N. Ingle, P. Sayyad, G. Bodkhe, M. Mahadik, A.-G. Theeazen, S. Shirsat, M.D. Shirsat, ChemFET Sensor: nanorods of nickel-substituted Metal-Organic framework for detection of SO 2. Appl. Phys. A 126, 1–9 (2020)
Q. Yang, B. Wang, Y. Chen, Y. Xie, J. Li, An anionic In (III)-based metal-organic framework with Lewis basic sites for the selective adsorption and separation of organic cationic dyes. Chin. Chem. Lett. 30, 234–238 (2019)
Souza, B. E.; Donà, L.; Titov, K.; Bruzzese, P.; Zeng, Z.; Zhang, Y.; Babal, A. S.; Möslein, A. F.; Frogley, M. D.; Wolna, M. Elucidating the drug release from metal-organic framework nanocomposites via in situ synchrotron microspectroscopy and theoretical modelling. ACS Appl. Mater. Interfaces 2020.
Connolly, B. M.; Madden, D. G.; Wheatley, A. E.; Fairen-Jimenez, D. Shaping the future of fuel: Monolithic metal-organic frameworks for high-density gas storage. J. Am. Chem. Soci. 2020.
S.G. Surya, S. Bhanoth, S.M. Majhi, Y.D. More, V.M. Teja, K.N. Chappanda, A silver nanoparticle-anchored UiO-66 (Zr) metal–organic framework (MOF)-based capacitive H 2 S gas sensor. CrystEngComm 21, 7303–7312 (2019)
X. Chen, Y. Lyu, Z. Wang, X. Qiao, B.C. Gates, D. Yang, Tuning Zr12O22 node defects as catalytic sites in the metal-organic framework hcp UiO-66. ACS Catalysis 10, 2906–2914 (2020)
Farha, O. K.; Eryazici, I.; Jeong, N. C.; Hauser, B. G.; Wilmer, C. E.; Sarjeant, A. A.; Snurr, R. Q.; Nguyen, S. T.; Yazaydın, A. O. z. r.; Hupp, J. T. Metal–organic framework materials with ultrahigh surface areas: is the sky the limit? Journal of the American Chemical Society 2012, 134, 15016–15021.
C.R. Wade, M. Dincă, Investigation of the Synthesis, Activation, and Isosteric Heats of CO2 Adsorption of the Isostructural Series of Metal-Organic Frameworks M3 (BTC)2 (M= Cr (Fe, Ni, Cu, Mo, Ru), (2012).
Song, Y.; Khudozhitkov, A. E.; Lee, J.; Kang, H.; Kolokolov, D. I.; Stepanov, A. G.; Yoon, M. Transformation of a proton insulator to a conductor via reversible amorphous to crystalline structure transformation of MOFs. Chem. Commun. 2020.
Q.-Q. Huang, Y.-J. Lin, R. Zheng, W.-H. Deng, C. Kashi, P.N. Kumar, G.-E. Wang, G. Xu, Tunable electrical conductivity of a new 3D MOFs: Cu-TATAB. Inorg. Chem. Commun. 105, 119–124 (2019)
T.T. Tung, M.T. Tran, J.-F. Feller, M. Castro, T. Van Ngo, K. Hassan, M.J. Nine, D. Losic, Graphene and metal organic frameworks (MOFs) hybridization for tunable chemoresistive sensors for detection of volatile organic compounds (VOCs) biomarkers. Carbon 159, 333–344 (2020)
O. Yaghi, H. Li, T. Groy, Construction of porous solids from hydrogen-bonded metal complexes of 1, 3, 5-benzenetricarboxylic acid. J. Am. Chem. Soc. 118, 9096–9101 (1996)
N. Ingle, S. Mane, P. Sayyad, G. Bodkhe, T. Al-Gahouari, M. Mahadik, S. Shirsat, M.D. Shirsat, Sulfur dioxide (SO 2) detection using composite of Nickel benzene carboxylic (Ni 3 BTC 2) and OH-functionalized single walled carbon nanotubes (OH-SWNTs). Front. Mater. 7, 93 (2020)
H. Hu, B. Zhao, M.E. Itkis, R.C. Haddon, Nitric acid purification of single-walled carbon nanotubes. J. Phys. Chem. B 107, 13838–13842 (2003)
L.-N. Jin, Q. Liu, W.-Y. Sun, Room temperature solution-phase synthesis of flower-like nanostructures of [Ni3 (BTC) 2· 12H2O] and their conversion to porous NiO. Chin. Chem. Lett. 24, 663–667 (2013)
Lestari, W.; Winarni, I.; Rahmawati, F. In Tilte2017; IOP Publishing.
L. Xing, F. Du, J.-J. Liang, Y.-S. Chen, Q.-L. Zhou, Preparation of Pt/SWNTs for heterogeneous asymmetric hydrogenation of ethyl pyruvate. J. Mol. Catal. A: Chem. 276, 191–196 (2007)
S. Liang, G. Li, R. Tian, Multi-walled carbon nanotubes functionalized with a ultrahigh fraction of carboxyl and hydroxyl groups by ultrasound-assisted oxidation. J. Mater. Sci. 51, 3513–3524 (2016)
P.-C. Ma, M.-Y. Liu, H. Zhang, S.-Q. Wang, R. Wang, K. Wang, Y.-K. Wong, B.-Z. Tang, S.-H. Hong, K.-W. Paik, Enhanced electrical conductivity of nanocomposites containing hybrid fillers of carbon nanotubes and carbon black. ACS Appl. Mater. Interfaces. 1, 1090–1096 (2009)
H.H. Choi, J. Lee, K.-Y. Dong, B.-K. Ju, W. Lee, Gas sensing performance of composite materials using conducting polymer/single-walled carbon nanotubes. Macromol. Res. 20, 143–146 (2012)
Eletskii, A. V.; Knizhnik, A. A.; Potapkin, B. V. e.; Kenny, J. M. Electrical characteristics of carbon nanotube-doped composites. Phys.-Uspekhi 2015, 58, 209.
S. Thirumalairajan, K. Girija, V.R. Mastelaro, N. Ponpandian, Surface morphology-dependent room-temperature LaFeO3 nanostructure thin films as selective NO2 gas sensor prepared by radio frequency magnetron sputtering. ACS Appl. Mater. Interfaces. 6, 13917–13927 (2014)
S. Mao, G. Lu, J. Chen, Nanocarbon-based gas sensors: progress and challenges. J. Mater. Chem. A 2, 5573–5579 (2014)
Yang, J.; Cheng, F.; Zhu, Z.; Feng, J.; Xue, M.; Meng, Z.; Qiu, L. Enhanced gas sensor based on SiO2@ mesoporous MCM-41 core-shell nanocomposites for SO2 visual detection. Analyst 2020.
Y. Liu, X. Xu, Y. Chen, Y. Zhang, X. Gao, P. Xu, X. Li, J. Fang, W. Wen, An integrated micro-chip with Ru/Al2O3/ZnO as sensing material for SO2 detection. Sensors Actuators B: Chem. 262, 26–34 (2018)
Q. Zhou, W. Zeng, W. Chen, L. Xu, R. Kumar, A. Umar, High sensitive and low-concentration sulfur dioxide (SO2) gas sensor application of heterostructure NiO-ZnO nanodisks. Sensors Actuators B Chem. 298, 126870 (2019)
W. Li, J. Guo, L. Cai, W. Qi, Y. Sun, J.-L. Xu, M. Sun, H. Zhu, L. Xiang, D. Xie, UV light irradiation enhanced gas sensor selectivity of NO2 and SO2 using rGO functionalized with hollow SnO2 nanofibers.Sensors Actuators B Chem. 290, 443–452 (2019)
Su, H. C.; Tran, T.-T.; Bosze, W.; Myung, N. V. Chemiresistive sensor arrays for detection of air pollutants based on carbon nanotubes functionalized with porphyrin and phthalocyanine derivatives. Sensors Actuators Reports 2020, 100011.
U. Chaitra, A.M. Ali, A.E. Viegas, D. Kekuda, K.M. Rao, Growth and characterization of undoped and aluminium doped zinc oxide thin films for SO2 gas sensing below threshold value limit. Appl. Surf. Sci. 496, 143724 (2019)
G. Jung, Y. Jeong, Y. Hong, M. Wu, S. Hong, W. Shin, J. Park, D. Jang, J.-H. Lee, SO2 gas sensing characteristics of FET-and resistor-type gas sensors having WO3 as sensing material. Solid-State Electronics 165, 107747 (2020)
M. Drobek, J.-H. Kim, M. Bechelany, C. Vallicari, A. Julbe, S.S. Kim, MOF-based membrane encapsulated ZnO nanowires for enhanced gas sensor selectivity. ACS Appl. Mater. Interfaces. 8, 8323–8328 (2016)
W. Zhou, Y.-P. Wu, J. Zhao, W.-W. Dong, X.-Q. Qiao, D.-F. Hou, X. Bu, D.-S. Li, Efficient gas-sensing for formaldehyde with 3D hierarchical Co3O4 derived from Co5-based MOF microcrystals. Inorg. Chem. 56, 14111–14117 (2017)
Acknowledgements
The authors are thankful to the Inter University Accelerator Center (IUAC), New Delhi, for providing financial and material science beamline with FESEM facilities through IUAC-UGC project having UFR-62320 & UFR-62321. Also thankful to DST-SERB (sanction no. EEQ/2017/000645), UGC-DAE CSR (RRCAT) Indore (Project No. CSR-IC-BL66/CSR-183/2016-17/847), UGC-SAP programme (F.530/16/DRS-1/2016 (SAP-II), dt. 16-04-2016), DST-FIST (Project No. No. SR/FST/PSI-210/2016(C) dtd. 16/12/2016), Rashtria Uchachatar Shiksha Abhiyan (RUSA), Government of Maharashtra for providing characterization facilities. Also thankful to Dr. Saif A. Khan, IUAC, New Delhi.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ingle, N., Sayyad, P., Deshmukh, M. et al. A chemiresistive gas sensor for sensitive detection of SO2 employing Ni-MOF modified –OH-SWNTs and –OH-MWNTs. Appl. Phys. A 127, 157 (2021). https://doi.org/10.1007/s00339-021-04288-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-021-04288-0