Abstract
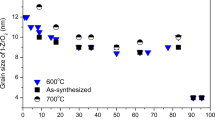
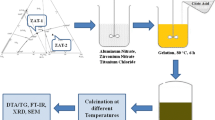
The present work deals with the preparation and characterization of Al2O3/ZnO composites under the effect of structural and temperature changes. A sol–gel method of preparing Al2O3/ZnO composites with different concentrations of ZnO and Al2O3 was used. Materials with the extremely high Al2O3 concentration (80 mol%) have been simply prepared by the sol–gel technique. 27Al NMR spectroscopy, X-ray diffraction (XRD), scanning and transmission electron microscopy (SEM and TEM) examined the structural features of all the samples. 27Al NMR showed a significant improvement in the product composition with the change of the Al2O3 to ZnO molar ratios. The main units that existed in the Al2O3 rich sample (80 mol%) are the structured groups AlO6, AlO5 and AlO4. The main unit in the lower Al2O3 concentration (20 mol%) is only AlO4 coordination. Through adding Al2O3, the crystalline configuration in the pure ZnO matrix is fully transformed into one of the amorphous forms. The amorphous composition of Al2O3/ZnO is retransformed into crystalline forms after calcination. The presence of different Al2O3 coordination such as AlO6, AlO5 and AlO4 is considered as the main cause for mismatching between different structural units that leads to transformation of crystalline into non-crystalline structure.







Similar content being viewed by others
References
M. Dehbashi, M. Aliahmad, M.R. Mohammad Shafiee, M. Ghashang, Synth. React. Inorg. Met.-Org. Nano-Met. Chem. 43, 1301–1306 (2013)
L. He, H. Cheng, G. Liang, Y. Yu, F. Zhao, Appl. Catal. A Gen. 452, 88–93 (2013)
P. Wei, D. Zhu, S. Huang, W. Zhou, F. Luo, Appl. Surf. Sci. 285, 577–582 (2013)
A. Aslani, A. Bazmandegan-Shamili, S. Barzegar, Phys. B 405, 3585–3589 (2010)
Q. Chen, S. Zheng, S. Yang, W. Li, X. Song, B. Cao, J Sol-Gel Sci Technol 61, 501–508 (2012). https://doi.org/10.1007/s10971-011-2651-0
M. Tavoosi, F. Karimzadeh, M.H. Enayati, Mater. Lett. 62, 282–285 (2008)
D. Jiang, K. Thomson, Scr. Mater. 56, 959–962 (2007)
K.E. Thomson, D. Jiang, Acta Mater. 60, 622–632 (2012)
S.I. Cha, K.T. Kim, Scr. Mater. 53, 793–797 (2005)
S. Maensiri, P. Laokul, Mater. Sci. Eng. A 447, 44–50 (2007)
V. Trombini, E.M.J.A. Pallone, R. Tomasi, Mater. Sci. Eng. A 501, 26–29 (2009)
R.M. Rahimi, H.R. Rezoie, A. Nemati Ceram. Int. 37, 1681–2168 (2011)
S.K.C. Pillai, B. Baron, M.J. Pomeroy, J. Eur. Ceram. Soc. 24, 3317–3326 (2004)
Y. Sun, W. Xiong, C. Li Trans, Nonferrous. Met. Soc. China 20, 624–631 (2010)
L.P. Dail, H. Deng, J. Zang, J. Mater. Sci: Mater. Electron. 19, 727–734 (2008)
F. Ruske, A. Pflug, V. Sittinger, Thin Solid Films 14, 4472–4477 (2008)
M. Ranjbar, G. DehghanNoudeh, M.A. Hashemipour, I. Monamadzadeh, J. Artif. Cells Nanomed. Biotechnol. Int. J. 47, 201–209 (2019)
P. Puspitasari and Andoko, ISSN 1819–6608 ARPN. J. Eng. Appl. Sci. 11(2):1317–1320 (2016)
M. Karpuraranjith, S. Thambidurai, Int. J. Biol. Macromol. 104, 1753–1761 (2017)
J.A. Johnson, J.K.R. Weber, A.I. Kolesnikov, S. Schweizer, J. Phys. Condens. Matter. 21(37), 3751031 (2009)
G. El-Damrawi, A.K. Hassan, A. Shahboub, Magn. Reson. Solids 20, 18202 (2018)
Y. Fujimoto, M. Nakatsuka, J. Non-Cryst. Solids 352, 2254–2258 (2006)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
El Damrawi, G., Salaheldin, H. & Abdelghany, M. Structural investigations on inorganic Al2O3–ZnO composites. Appl. Phys. A 127, 145 (2021). https://doi.org/10.1007/s00339-020-04268-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00339-020-04268-w