Abstract
In this study we estimate relative pollen productivity (RPP) for plant taxa characteristic of human-induced vegetation in ancient cultural landscapes of the low mountain ranges of Shandong province in eastern temperate China. RPP estimates are required to achieve pollen-based reconstructions of Holocene plant cover using modelling approaches based on Prentice’s and Sugita’s theoretical background and models (REVEALS and LOVE). Pollen counts in moss samples and vegetation data from 36 sites were used in the Extended R-Value (ERV) model to estimate the relevant source area of pollen (RSAP) of moss polsters and RPP of major plant taxa. The best results were obtained with the ERV sub-model 3 and Prentice’s taxon-specific method (using a Gaussian Plume dispersal model) to distance weight vegetation data. RSAP was estimated to 145 m using the maximum likelihood method. RPP was obtained for 18 taxa of which two taxa had unreliable RPP (Amaranthaceae/Chenopodiaceae and Vitex negundo). RPPs for Castanea, Cupressaceae, Robinia/Sophora, Aster/Anthemis-type, Cannabis/Humulus, Caryophyllaceae, Brassicaceae and Galium-type are the first ones for China. Trees, except Robinia/Sophora (RPP = 0.78 ± 0.03) have larger RPPs than herbs other than Artemisia (RPP = 24.7 ± 0.36). The RPPs for Quercus, Pinus and Artemisia are comparable with other RPPs obtained in China, the RPPs for Pinus, Quercus, Ulmus, Cyperaceae and Galium-type with the mean RPPs obtained in Europe, and RPP for Cupressaceae with that for Juniperus in Europe. The values for Aster/Anthemis-type, Caryophyllaceae, Asteraceae SF Cichorioideae and Juglans differ from the few RPPs available in China and/or Europe.
Similar content being viewed by others
Introduction
Quantitative reconstructions of anthropogenic land-cover change at the regional to continental spatial scale, especially the relative size and distribution of open and wooded land area, are of great value in answering research questions related to climate change (e.g. Gaillard et al. 2010; Strandberg et al. 2014). In this study, we estimate relative pollen productivity (RPP) of major tree and herb taxa in ancient agrarian landscapes of central and southern Shandong, central eastern China (Fig. 1), as the first step of a larger project using RPPs and Sugita’s REVEALS model (Sugita 2007a) to achieve pollen-based quantitative reconstructions of Holocene regional vegetation cover in northern and temperate China as a contribution to the PAGES LandCover6k initiative (http://www.pastglobalchanges.org/ini/wg/landcover6k/intro). The aim of the initiative is to provide quantitative descriptions of Holocene anthropogenic vegetation cover for climate modelling, with the goal of studying the impact of past land-use change as one of the many forcings of past climate change. Because earlier RPP studies in China were performed in forest, desert, steppe and meadow vegetation with little human impact, there was a need for RPP of cultivated crops, ruderals and taxa characteristic of grazed land.
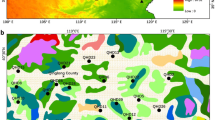
Location of the study area (a) and distribution of the sampling sites (b). a Location of study areas for our study and earlier investigations: a Wang and Herzschuh (2011), b Wu et al. (2013), c Li et al. (2011), d Xu et al. (2014), e Ge et al. (2015), f Li et al. (2015). b. Vegetation map of the study area and location of the study sites for collection of moss polsters (pollen samples) and vegetation surveys. c, d Common cultural landscapes in the study area
RPP can be calculated using the Extended R Value (ERV) model (Parsons and Prentice 1981; Prentice and Parsons 1983; Sugita 1993, 1994) given that modern pollen and related vegetation data are available. In the last decades, many studies have been conducted to estimate RPP for major taxa in various landscapes of Europe (see syntheses in Broström et al. 2008; Mazier et al. 2012): wooded vegetation in UK (Bunting 2003), Estonia (Poska et al. 2011) and Poland (Baker et al. 2016); forest-tundra ecotone in western-central Sweden (Von Stedingk et al. 2008); open and semi-open cultural landscapes of southern Sweden (Sugita et al. 1999; Broström et al. 2004, 2005), Denmark (Nielsen and Sugita 2005), the Swiss Plateau (Soepboer et al. 2007) and the Czech Republic (Abraham and Kozáková 2012); mown meadows and pasture lands in western Norway (Hjelle 1998; Hjelle and Sugita 2012); wooded pasture landscapes of the Jura mountains in Switzerland (Mazier et al. 2008); various vegetation communities in southern Greenland (Bunting et al. 2013a). The impact of vegetation survey methods on the relevant source area of pollen (RSAP) sensu Sugita (1994) and RPP estimates was discussed in Bunting and Hjelle (2010) and a standardized vegetation-survey protocol was proposed by Bunting et al. (2013b). The relatively high number of RPP estimates available in Europe made it possible to test and use the models developed for quantitative reconstruction of vegetation cover such as the Landscape Reconstruction Algorithm (LRA: REVEAL and LOVE models) (Sugita 2007a, b) and the Multiple Scenario Approach (MSA; Bunting and Middleton 2005). REVEALS reconstructions over the Holocene are now available for a large part of Europe (Nielsen and Odgaard 2010; Fyfe et al. 2013; Trondman 2015; Trondman et al. 2016; Marquer et al. 2014). LOVE reconstructions were performed in southern Sweden (Cui et al. 2014; Mazier et al. 2015), Denmark (Overballe-Petersen et al. 2013) and Britain (Fyfe et al. 2013). The MSA was applied in northern Sweden (Karlsson et al. 2008).
Model-based quantitative reconstructions of plant cover are spatially limited due to the unavailability of RPPs in large parts of the world. RPP are available in e.g. northern America for tree taxa (Calcote 1995) and some herb taxa (Commerford et al. 2013), and in southern Africa for a few savanna taxa (Duffin and Bunting 2007). A relatively large number of RPP studies were conducted recently in China. RPPs for the four major taxa (Artemisia, Chenopodiaceae, Cyperaceae, Poaceae) characteristic of the Tibetan Plateau were applied in REVEALS-based reconstructions showing that the proportions of Artemisia and Chenopodiaceae in the modelled vegetation were greatly reduced compared to pollen percentages, while Cyperaceae had a larger relative fraction in the vegetation than in the pollen assemblages (Wang and Herzschuh 2011). Moreover, the REVEALS-based cover of the four taxa resulted in lower values of species-composition turnover than pollen percentages, which indicated that the previously reported vegetation changes might have been overestimated. RPPs for herbs and a few trees and shrubs are available for desert and semi-desert vegetation (Li et al. 2011), steppe and meadow vegetation of northern China (Xu et al. 2014; Ge et al. 2015), and several forest-types of northeastern China (Li et al. 2015) (Fig. 1). So far no attempts have been made to obtain RPP estimates for taxa related to human-influenced landscapes in northeastern China, which hampers reconstruction of cultural landscapes over the Holocene in the area with the most intensive human impact over the past three millennia (Fu 2003). We chose as study area the central and southern low mountains of the Shandong province (Fig. 1) where ancient land-use structures and practices dating to the last 1,000 calendar years are still partly preserved (Xiuqi Fang pers. comm.). Therefore, we assume that this landscape is a better analogue of past agrarian landscapes in terms of vegetation and flora than modern agrarian landscapes. The vegetation of the study region is a mosaic of crop cultivation, weeds and ruderals on terraces, pasture land, and patches of woodland (Fig. 2). The weed and ruderal flora is rich and is assumed to include a mix of species that might have been characteristic of earlier traditional agriculture.
Landscape of the study region, province of Shandong, China. Low mountains/hills with traditional crop cultivation on terraces (solid line), grazing land and woodlands at higher altitudes, and modern agriculture in the lowlands (dash line). Photo taken by Florence Mazier (University of Toulouse), during field work in May–June 2014
The study area
The Shandong province is one of the most important agricultural provinces of China located in the lower reach of the Yellow River drainage basin in central-eastern China (Fig. 1). The study area is ca. 100 km (longitudinal distance) ×200 km (latitudinal distance) large, and located between the latitudes 35°00′ and 36°30′N and the longitudes 117°00′ and 118°30′E. The elevations are <500 m a.s.l except for some mountains tops >1,000 m a.s.l. high (Fig. 1). The bedrock is characterized by limestones in the low mountain areas up to elevations of ca. 500–1,000 m, while granite and gneisses predominate at higher elevations. The climate of central and southern Shandong belongs to the sub-humid, warm climate zone with mean annual temperatures of 12–14.5 °C and annual precipitations of 700–900 mm occurring mainly between June and September.
The spatial landscape patterns of the low mountains are the result of geological and biogeographical characteristics combined with traditional land-use. The overall landscape structure at a large spatial scale comprises (1) low mountains with cultivated and abandoned terraces with small fields, grazed land, and woodland, and (2) flat, low-lying plains occupied by modern agriculture with large cultivated fields and planted trees (often Populus sp.). These two broad landscape/land-use units are each characterized by a relatively consistent vegetation mosaic in terms of vegetation patch-size and plant-taxa composition (Figs. 1, 2). Therefore, the study area was assumed to be suitable for calculation of pollen productivity estimates using the ERV model (see theoretical background of the ERV model in Methods, below).
Modern high-production agriculture of the plains is dominated by cultivation of Triticum spp. (wheat), Zea mays (maize) and fruit and nut trees including Juglans regia (walnut), Castanea mollissima (chestnut), Diospyros kaki (kaki), Zanthoxylum bungeanum (Chinese pepper), Malus domestica (apple), Pyrus spp. (pear), Crataegus pinnatifida (Chinese hawthorn) and Prunus cerasus (cherry). There are very few weeds and ruderal species growing in and between the fields. Both relatively modern and old-fashioned traditional agriculture are practiced on the terraces of the low mountains. Terraces with cultivated fields are separated by soil slopes or high stone walls. The slopes are covered with herb or tree vegetation, e.g. Poaceae (grasses), Selaginella chinensis (starry spikemoss), Vitex negundo (Chinese chastetree), Artemisa mongolica (besser) and A. annua (sagewort), Lespedeza bicolor and L. tomentosa (bush clovers), Humulus scandens (hop), Asteraceae and Caryophyllaceae. The stone walls may be covered by vegetation including the same taxa as mentioned above. Arachis hypogaea (peanut) and Ipomoea batatas (sweet potato) are the dominant cultivated crops, but the variety of other crops and planted fruit/nut trees is high. Trees and crops may be cultivated in the same fields when trees are still young. Fruit trees and crops are the same as those cultivated in the low lying plains, with the addition of Lonicera japonica (Japanese honeysuckle) cultivated for its flowers (used as “tea”). In the upper part of the mountains, patches of coniferous forest/woodland (planted or natural) and broad-leaved deciduous woodland occur together with shrubs, grasslands and meadows that are often grazed by sheep and goats. Coniferous woodlands/forests are dominated by Pinus densiflora (Japanese red pine), P. tabulaeformis (Chinese red pine), P. thunbergii (Japanese black pine), and Platycladus orientalis (Chinese thuja), while broad-leaved forests mainly consist of Robinia pseudoacacia (Black locust), Quercus acutissima (sawtooth oak) and Q. variabilis (Chinese cork oak), and shrubs including mainly Vitex negundo, Lespedeza spp and Zizyphus jujuba (Chinese date). On the top of the mountains, plant cover is scarce and dominated by Themeda triandra (red oat grass) and Bothriochloa ischaemum (King Ranch bluestem).
Methods
The methodological strategy and flow of methods are shown in Fig. 3. Detailed overviews of the theory of the ERV model and its developments can be found in e.g. Gaillard et al. (2008) and Bunting et al. (2013b). More details on the underlying theory of the ERV model and the methods are presented in the ESM.
Site selection
Because the ERV model assumes that the taxon-specific background term ωi for all sites included in the analysis is constant at the RSAP distance, the results of the maximum likelihood method (likelihood function scores or log likelihood) used to identify the RSAP are very sensitive to the overall vegetation structure and composition in the study region. Moreover, a statistical method such as maximum likelihood requires a random distribution of pollen-vegetation sites in the landscape. Ignoring this rule may lead to results that are difficult to interpret and estimates of RSAP and RPP that are not reliable (Broström et al. 2005). Therefore, sampling sites were randomly placed using ArcView (ArcGIS 10.0) with the rule that the distance between sampling site should be at least 5,000 m in order to avoid auto-correlation (e.g. Bunting et al. 2013b). Of the 60 random sites, 37 were appropriate for data collection, i.e. the areas were reachable by car and the sites were reachable by foot within no more than two hours walk. The 37 sites are located at elevations between 163 and 538 m a.s.l. (Table 1). Field work comprising collection of pollen samples and vegetation surveys around each sampling location was performed in May–June 2013 and 2014.
Pollen data
The pollen sample (moss polster) was collected before doing any vegetation survey. Several moss subsamples were taken within an area of 0.5 m radius at each of the 37 sites. In the laboratory, remaining soil particles were removed from the moss before pollen extraction. The volume of each moss sample was measured by the “water displacement method” and treated with (1) 10% cold HCl, (2) 10% hot KOH, (3) 46% hot HF, and (4) hot acetolysis following a slightly modified version of the acetolysis procedure of Fægri and Iversen (1989). HCl and HF were used to remove any remaining calcareous and mineral fractions from the sample. After the HCl and KOH treatment, the moss was also sieved through a 0.25 mm mesh size to remove any larger mineral or organic remains.
ESM Table 1 presents the pollen-types that were identified in the moss samples, their definition, and harmonization with plant taxa in the vegetation data. Table 2 presents the pollen-types that were finally used for the first, exploratory ERV-model run. Pollen identification was performed using published pollen identification keys and pollen-type descriptions (Punt et al. 1976–2009; Wang 1995; Beug 2004), the pollen atlas of Reille (1995), and reference collections. The results of the pollen analysis are shown in ESM Fig. 1. Taxonomy and nomenclature of pollen-types or groups of pollen-types follow mainly Beug (2004) and Wang (1995) (Table 2; ESM Table 1 and ESM Fig. 1).
Vegetation data
Vegetation data was obtained following the standard protocol published by Bunting et al. (2013b) for Europe. The surveys were performed in May–June 2013 and 2014.
Vegetation survey 0–10 m: Vegetation was surveyed in 1 m2 quadrats, a central one around the site of pollen sampling, and 20 quadrats centred at the distances 1, 2.5, 4.5 and 7.5 m in all four directions N, E, S, W, and 7.5 m in the NE, SE, SW, and NW directions (21 quadrats in total). Plant taxa composition was estimated as total cover in percentage of the quadrat area.
Vegetation survey 10–100 m: the boundaries between vegetation communities were drawn using a compass and measuring distances with a hand-held GPS, a modification of the standard method described in Bunting et al. (2013b) where vegetation communities are drawn while following transects from the pollen sampling site out to 100 m. The latter method is not practical in situations where some bushy or wooded vegetation communities are difficult, if not impossible to walk through. Mapping with GPS is also more precise in terms of the boundaries of the communities. After or during mapping, the cover of plant taxa was estimated for each community using 1 m2 quadrats in open communities, and 6-m radius point surveys in semi-open and forest communities. The major vegetation communities recognized in the field are listed in Table 1.
Vegetation data beyond 100 m: satellite images within an area of 1,500 m radius (distance assumed to be larger than the RSAP) from the centre of the moss sample area were downloaded from Google Earth professional for each study site. Landscape/vegetation features differentiated by their colour in the images were assigned to vegetation/land-use units (in total 18) that were defined on the basis of the vegetation communities identified in the field surveys (ESM Table 2). Once a number of polygons were drawn in each vegetation/land-use unit, a map was created by maximum likelihood classification with ArcView (ArcGIS 10.0) (Figs. 3, 4). These vegetation/land-use maps have a resolution of 25 m2. However, the method used introduces large errors that cannot be quantified unless the classification is checked in field. Moreover, we assume that the taxa composition in each vegetation/land-use unit is homogenous over the entire study area and can be estimated as the mean composition calculated from the vegetation inventories performed in the field within 100 m at each of the study sites. This assumption also leads to errors that are not quantifiable.
Examples of vegetation/land-use maps for nine randomly selected sites created from Google Earth satellite images for each of the 36 sites included in the study. The maps of vegetation/land-use units were created by maximum likelihood classification with ArcView (ArcGIS 10.0). See text for more details. The land-use-type IDs are the same as in Table 3. Land-use-types 2, 3, and 4 are not present in these sites but occur at other sites. Land use-types: 1 Pinus single tree and Pinus woodland; 5 Quercus woodland; 6 Mixed Robinia and Quercus woodland; 7 Robinia woodland; 8 Platycladus single tree and Platycladus forest; 9 Planted Populus; 10 Mixed Populus and Platycladus forest; 11 cultivated trees; 12 cultivated Castanea; 13 open land with Vitex and herbs; 14 crop fields; 15 abandoned fields; 16 field border (trees + crops); 17 rocks; 18 water body
Data handling for vegetation input files in the ERV models: for each set of vegetation data (0–10, 10–100, 100–1,500 m), the mean absolute cover (in m2/m2) of the plant taxa included in the analysis was calculated for each chosen distance increment. In this study we use a 1 m increment consistently within the entire distance of vegetation survey (1,500 m), following, e.g. Sugita et al. (2010) and Mazier et al. (2008). We assume homogeneous vegetation cover in each concentric ring, as it is the assumption for data analysis using ERV models and related pollen-dispersal functions. The computer program used for the ERV model-based analysis requires vegetation datasets in concentric rings with the same width from the pollen-sampling sites out to the maximum distance of the vegetation survey if the RSAP estimate is to be obtained statistically using a moving-window regression approach. Considering the importance of vegetation data close to the pollen-sampling sites, it is justifiable to extract the plant abundance data within a 10-m radius with a 1 m increment, and use the same increment beyond a 10 m radius for consistency. However, the spatial resolution of the vegetation survey data is at the best ca. 5 m × 5 m beyond 10 m. The errors on (1) the vegetation composition and (2) the distances from the pollen sampling point at which the various plant taxa are located increase strongly between the three vegetation datasets. Nevertheless, we assume that compilation of vegetation data in concentric rings of 1 m up to 1,500 m does not alter the plant abundance estimates significantly compared to vegetation data extracted in larger concentric rings of 5–10 m. However, this assumption needs to be tested in future studies.
ERV-model runs
Besides pollen and vegetation data, other input data needed to run the ERV model are fall speed of pollen (FSP) and wind speed. We used a constant wind speed of 3 m s−1, which is assumed to correspond approximatively to the modern mean annual wind speed in continental China. FSP for the 24 selected taxa (ERV.I) was calculated using Stoke’s law (Gregory 1973) and measurements of the pollen grains. For 14 taxa, 30 grains of each taxon were measured on reference slides. These reference slides were prepared from fresh flowers that were dried and washed through a sieve mesh of 0.2 mm. The material <0.2 mm was then acetolysed (Fægri and Iversen 1989), and glycerol was added before preparing the slides. For ten taxa, the measurements are from Beug (2004, four taxa) and Wang (1995, six taxa) (Table 2). The calculated velocities were adjusted by a shape factor following McNown and Malaika (1950).
All sets of ERV-model runs include runs using the three sub-models (see the ERV model description in ESM) and two methods to distance weight vegetation, i.e. the taxon-specific distance-weighting method for bogs proposed by Prentice (1985) (Prentice’s model), and the inverse distance (1/d) method for comparison. These sub-models and distance-weighting methods were implemented in the program ERV.Analysis.v1.3.1.exe (Sugita, unpublished). It should be stressed that this program implements the original Prentice’s model that uses a Gaussian plume diffusion model (GPM) of small particles in the air (Sutton 1953). Recently, Theuerkauf et al. (2016) and Mariani et al. (2016) used a state-of-the-art Lagrangian stochastic dispersal model (LSM) and concluded that the LSM may perform better than the GPM when applied for REVEALS-based reconstructions of past regional vegetation in northern Germany (Theuerkauf et al. 2016) or for ERV-based estimates of RPPs in Australia (Mariani et al. 2016) (see also discussion below).
A first selection of taxa was made with the following criteria: each selected taxon (1) is present in both pollen and vegetation data, (2) is represented as pollen in a minimum of ten sites, and (3) is characterized by between-sample variation in pollen percentages and vegetation abundances. This set of taxa was used to perform the first set of ERV-model runs (ERV.I). Poaceae was chosen as reference taxon (see the ERV model theory in ESM). It is the most common reference taxon used in studies of RPPs in open and semi-open vegetation because it is one of the most common pollen-types in such vegetation, and has often a close-to-linear pollen-vegetation relationship. In our case, although there is a large deviation of the values around the ideal linear relationship, Poaceae has the highest number of values and best spread of these values along the gradient (see results below and Fig. 5). The RSAP was calculated with the moving-window regression method (Sugita in Gaillard et al. 2008) using a window of 100 m.
Plots of the log likelihood function scores using the three ERV sub-models and two methods of vegetation distance weighting, the taxon-specific and the inverse distance (1/d) methods, based on pollen data from moss samples and vegetation data within 1,500 m radius around the pollen sample at 36 random sites
The results of ERV.I were used to (1) make a first assessment of the log-likelihood curve and identify the RSAP, (2) plot the pollen/vegetation relationships from the three sub-models at the RSAP distance, and (3) evaluate these relationships. The taxa with non-linear relationships were excluded and a second set of ERV-model runs using the same procedure as ERV.I was performed (ERV.II).
The results of ERV.II provided a theoretically correct curve of log-likelihood values, i.e. the values increased with distance and reached an asymptote. If at this stage (ERV.II) the log likelihood curve does not behave in a theoretically correct way, further ERV-model runs with different selections of sites and/or taxa should be performed. Awkward behaviours of log likelihood curves may be due to the selection of taxa with non-linear pollen-vegetation relationships and/or too large between-site differences in regional vegetation (unpublished data, pers. comm. from several collaborators). It is recommended to delete site outliers in terms of regional vegetation composition and structure, and pollen-types for which the pollen-vegetation relationship is too far from a linear relationship.
Results
Pollen data
A total of 78 pollen-types were found in the 37 moss samples, of which 71 were found as plant taxa in the vegetation surveys (listed in ESM Table 1). Ephedra, Euphorbiaceae, Larix, Nitraria, Tilia, Typha and Valerianaceae were found only as pollen-types. One moss sample had a very low diversity of pollen-types and bad preservation of the pollen grains and was therefore excluded from further analysis. The 28 most common pollen taxa are presented in the percentage pollen diagram (ESM Fig. 1). Of those 28 taxa, Paulownia, Zanthoxylum-type, Betula and Salix were deleted before the first set of ERV runs (ERV.I) because they had gradients of pollen and vegetation values that were too poor. Of those 24 taxa, six additional taxa were found to have a relationship of the ERV-adjusted pollen and vegetation data that again was too poor. Therefore, the second set of ERV runs (ERV.II) was performed with 18 taxa.
Vegetation data
The raw vegetation survey data are not presented in this paper. A PCA of (1) the distance-weighted, harmonized vegetation (DWHV) data (in m2/m2) (see methods above), and (2) both pollen and DWHV data together did not exhibit clear outlier sites except for one site with very low pollen concentration (results not shown).
Relevant source area of pollen
Log-likelihood increases with distance until maximum values within 100 to 200 m distance whatever the combination of sub-model and distance-weighting method. However, it reaches an asymptote with relatively constant values beyond ca. 150 m only when sub-model 3 is used (Fig. 6). When sub-models 1 and 2 are used, the log likelihood values decrease after the maximum values, most significantly when sub-model 2 is applied. The distance-weighting methods used have little influence on the log likelihood curve and the RSAP distance. The RSAP calculated with the moving-window regression is 145 m when sub-model 3 and Prentice’s model are used.
Plots of the pollen-vegetation relationship at the distance corresponding to the radius of the relevant source area of pollen (RSAP = 145 m) as calculated using the ERV sub-model 3 and the taxon-specific vegetation distance-weighting method (Prentice’s model). Upper panel: original pollen proportion and vegetation absolute abundance. Lower panel: relative pollen loading and absolute distance-weighted vegetation abundance
Pollen-vegetation relationships
The scatter plots of the original (percentages) and ERV-adjusted pollen and vegetation values using sub-model 3 and Prentice’s model are shown in Fig. 6 and ESM Fig. 2. Sub-model 1 (results not shown) and 3 produced comparable results in terms of the pollen-vegetation relationship. Pinus exhibits the relationship that is closest to an ideal linear relationship, followed by Artemisia, Quercus, Poaceae, Cyperaceae and Asteraceae SF Cichorioideae (Fig. 6). Several taxa have relationships characterized by many low values and few high values of pollen and vegetation (i.e. Castanea, Juglans, Platycladus, Robinia/Sophora), or have strongly deviating high values with either high pollen values corresponding to low vegetation values or the inverse (i.e. Amaranth./Chenop., Cannabis/Humulus, Castanea, Brassicaceae, Galium-type, Ulmus, Vitex) (ESM Fig. 2).
Relative pollen productivity estimates
The RPP estimates for 18 taxa and their standard errors (SEs) calculated using the three ERV sub-models and two alternative distance-weighting methods are shown in Fig. 7 and Table 4. ERV sub-model 2 often produces lower RPPs than sub-models 1 and 3, and sub-model 3 higher values than sub-model 1 and 2. The ranking of the RPPs is the same for all combinations of ERV sub-model and distance-weighting method, i.e. for tree and shrub taxa Castanea > Pinus > Quercus > Cupressaceae > Ulmus > Juglans > Vitex negundo, with Ulmus having a RPP of 1, and for herb taxa Artemisia > Cannabis/Humulus > Aster/Anthemis-type > Galium-type > Poaceae > Brassicaceae > Caryophyllaceae > Asteraceae SF Cichorioideae > Cyperaceae > Amaranthaceae/Chenopodiaceae, with Artemisia and Cannabis/Humulus having higher RPPs than Castanea. Trees, except Robinia/Sophora (RPP = 0.78 ± 0.03), have larger RPP estimates than herbs, except Artemisia (RPP = 24.7). Unfortunately, although cereal pollen was found in many of the moss samples (ESM Table 2), the pollen-vegetation relationship was too poor to provide a reliable RPP estimate.
Estimates of relative pollen productivity (RPP) with standard deviation for 18 taxa estimated with the three ERV sub-models and two vegetation-distance weighting methods, Prentice’s model and the inverse distance (1/d), based on pollen data from moss samples and vegetation data within 1,500 m radius around the pollen sample at 36 random sites
Discussion
In this study we used the original model of Prentice (1985) that uses a Gaussian plume diffusion model (Sutton 1953; see methods above). Recently, Theuerkauf et al. (2016) used a state-of-the-art Lagrangian stochastic dispersal model (LSM) and compared the model outcomes with those based on a conventional Gaussian plume dispersal model (GPM). In the LSM turbulence implies that pollen fall-speed has little impact on the dispersal pattern, whereas fall speed is a major factor influencing dispersal in the GPM. These authors also showed that the REVEALS model (Sugita 2007a) performed better in reconstructing regional plant cover in NE Germany when applied with the LSM rather than with the GPM. Mariani et al. (2016) found that RPPs and RSAP obtained using the LSM appeared to be more realistic when compared to the results from the GPM in the wind- and animal-pollinated vegetation mosaics of western Tasmania. The discussion below is conducted having in mind that the use of the GPM is one of the potential methodological factors influencing our results. A comparison of RPP obtained when using the LSM with the RPP obtained in this study is in progress.
Pollen-vegetation relationship
It is common in RPP studies based on field data that few taxa have their pollen-vegetation data closely fitting to the theoretical ERV-model linear relationship, in particular in half-open and open landscapes in Europe (e.g. Hjelle 1998; Broström et al. 2004; Mazier et al. 2008; Poska et al. 2011) and in China (e.g. Xu et al. 2014; Ge et al. 2015). The latter might be due to (1) the generally low pollen productivity combined with restricted dispersion of herb pollen (except for Artemisia, Poaceae, Cyperaceae and Asteraceae SF Cichorioideae), leading to low values in the pollen data corresponding to high values in the vegetation data (e.g. Brassicaceae, Galium-type), and/or (2) the generally effective dispersion of tree pollen (except for Ulmus and the cultivated tree Juglans) and the generally scattered occurrence of such trees in the landscape, producing high values in the pollen data that may correspond to either high or low values in the vegetation data (e.g. Castanea, Robinia/Sophora). Further, the cultural landscape in our study region is characterized by a high cover of cultivated soils with shifting cover of crops, weeds and ruderals over the years. However, we assume that the cover of the weed taxa for which RPP was estimated (e.g. Artemisia, Humulus, Poaceae, Asteraceae SF Asterioideae, Asteraceae SF Cichorioideae) at the time of the surveys is a fair approximation of the mean cover of these taxa over a 1–3 year period of pollen deposition in the moss sample, even though the weeds may have changed their spatial distribution over the years. Other factors influencing the pollen-vegetation relationship may be stochastic pollen dispersal by insects rather than wind transport or pollen transported in clumps rather than as single grains, given that the ERV model assumes dispersal of single grains by wind and uses a Gaussian plume model (GPM) rather than a Lagrangian model (LSM).
Relevant source area of pollen (RSAP)
Our results confirm that when absolute vegetation data are used sub-model 3 is the most appropriate model to use.
The RSAP is affected by a variety of factors including size and type of sediment basin and spatial distribution of taxa and vegetation patches in the vegetation/landscape studied. The effect of basin size on the RSAP has been tested in many previous studies showing that the RSAP increases with basin size. For instance, in North America, RSAPs of 50–100 m were estimated for moss samples in small forest hollows, 300–400 m for surface sediments from small lakes, and 600–800 m for sediments from middle sized lakes (Sugita 1993). In Europe, RSAP for moss polster samples was estimated to be 400 m in the open and semi-open cultural landscapes of southern Sweden (Broström et al. 2005), ca. 300 m in the pasture woodland landscape of the Jura Mountains (Mazier et al. 2008), 1,050 m in the modern agricultural landscape of central Bohemia (Abraham and Kozáková 2012), and 1,000 m in the boreal forest landscapes of northern Finland (Räsänen et al. 2007). The variability of the RSAP for moss polsters in Europe is most probably due to differences between studied landscapes in terms of size and spatial distribution of vegetation patches. The latter has been shown to strongly influence the RSAP (Sugita et al. 1999; Bunting et al. 2004; Nielsen and Sugita 2005; Gaillard et al. 2008; Hellman et al. 2009a; Poska et al. 2011). Hellman et al. (2009a, b) tested these effects using simulated hypothetical landscapes and found that the influence of vegetation spatial structure was due to its effect on the spatial distribution of the taxa involved, i.e. the more fine-grained and homogenous spatial distribution of the taxa, the smaller the RSAP. The RSAP is also influenced by the plant taxa characteristic of the studied landscape. All-herb vegetation usually has smaller RSAP than all-tree vegetation, while mixed herb-tree vegetation may have larger RSAP than all-herb or all-tree vegetation (Bunting and Gaillard in Gaillard et al. 2008; Bunting and Hjelle 2010). Finally, the RSAP estimate may be influenced by the dispersal model used (Mariani et al. 2016).
In the case of our study region, we expected a higher RSAP than the estimated 145 m because of the large degree of openness and the relative scarcity of tree patches at the regional spatial scale. However, the satellite maps for extraction of vegetation data beyond 100 m were generated at a resolution of 25 m2, which created a high-resolution patchiness of vegetation/land-use-types comparable to the patchiness recorded within 100 m, which could explain the relatively small RSAP. The RSAP values obtained in earlier studies in China are generally larger, but few of the published studies are based on pollen data from moss polsters. Li et al. (2015) estimated a RSAP of 2,000–2,500 m for moss polsters in the woodlands of the Changbai Mountains in north eastern China. The large RSAP probably reflects the heterogeneity of the woodland landscape in the study region. Interestingly, the RSAP of 145 m in the cultural landscape of Shandong is comparable to the RSAP of moss polsters in the cultural landscapes of southern Sweden (400 m; Broström et al. 2005) and the Swiss and French Jura Mountains (ca. 300 m; Mazier et al. 2008). Therefore, it would be worth testing whether the RSAP in other cultural landscapes of the northern hemisphere temperate zone is generally of such magnitude.
Relative pollen productivity (RPP) estimates
In this study, few of the pollen-vegetation relationships as corrected by the ERV model are perfectly linear (see discussion on pollen-vegetation relationships above). It is unclear what effect the poorer relationships in our dataset had on the resulting RPP estimates. Estimates of RPP values can be tested by using them in pollen-based reconstruction of modern or historical plant cover and comparing the results with modern or historical plant cover (e.g. Nielsen 2004; Hellman et al. 2008). Such a test is in progress for the Shandong province and will be published elsewhere.
The reliability of the RPP estimates can also be assessed on the basis of the standard deviation (SD) calculated in the ERV analysis (see details in the ESM, description of methods). If the SD is larger than the RPP value, it implies that the estimated RPP is not different from zero and should be considered as unreliable (Sugita, personal communication). In our study, the RPP estimates for Vitex negundo (0.0025 ± 0.0184) and Amaranthaceae/Chenopodiaceae (0.18 ± 0.16) are considered as unreliable. Another way to evaluate the RPP estimates is to compare the results from the ERV sub-models 1 and 3. These sub-models have usually provided similar values (e.g. Broström et al. 2004; Mazier et al. 2008). In our study, when sub-models 1 and 3 are used, the taxa for which the RPPs are most similar are: Artemisia, Aster/Anthemis-type, Caryophyllaceae, Asteraceae SF Cichorioideae, Cyperaceae, Cannabis/Humulus, Pinus, Quercus, Robinia/Sophora, Ulmus. In contrast, the RPPs (sub-model 3; sub-model 1) obtained for Castanea (11.49; 4.63), Brassicaceae (0.87; 0.14), Galium-type (1.23; 0.32), Juglans (0.3; 0.96), and Cupressaceae (1.11; 0.51) are significantly different depending on the sub-model used, which could suggest that these values should be considered with care.
It is expected that the taxon-specific distance weighting (dw) method using a GPM will produce different RPP estimates than the 1/d dw method. If GPM is applied, the 1/d method may lead to an overestimation of the RPP of taxa with large and heavy grains, while it will underestimate the RPP of taxa with small and light grains (Broström et al. 2004; Nielsen 2004; Soepboer et al. 2007; Mazier et al. 2008; Poska et al. 2011). However, using a LSM may result in smaller differences between RPP estimates obtained applying the taxon-specific or 1/d dw methods because FSP has less influence on pollen dispersal in LSM than in GPM (Theuerkauf et al. 2016).
A comparison of RPPs in China and Europe
Of the RPP estimates available from published studies in northern and temperate China (Li et al. 2011, 2015; Wang and Herzschuh 2011; Xu et al. 2014; Ge et al. 2015), the ones that best can be compared with the RPPs obtained in this study are those of Ge et al. (2015) from the meadow and steppe landscapes of Inner Mongolia, and of Li et al. (2015) from the woodlands of the Changbai mountains, because the methods used in these investigations are comparable to ours. However, Ge et al. (2015) have used pollen from soil samples and Li et al. (2015) did not apply the random sampling approach and the standardized strategy of vegetation data collection of Bunting et al. (2013b). There are only six taxa for which this study’s RPP values can be compared with other published values in China, i.e. Artemisia, Cyperaceae, Galium-type, Juglans, Quercus, and Ulmus (Table 5). Several additional RPP studies have been performed in meadows, steppes areas, and woodland but were still unpublished at the time of the present study. In this discussion, we also compare the Chinese RPP values with those obtained in Europe (Mazier et al. 2012, RPP dataset standard 2).
Artemisia has a RPP of 24.7 in this study and 19.33 in Inner Mongolia (Ge et al. 2015). Some Artemisia species are growing in the two environments, others are characteristic of either cultural landscapes or meadows and steppes. In spite of the different species involved, the RPPs are comparable. In contrast, the RPPs of Artemisia from western Europe are 10× lower (Mazier et al. 2012). However, the species represented in Europe are different. The RPP estimate of Cyperaceae in our study (0.21) is much lower than the value obtained in Inner Mongolia (8.9; Ge et al. 2015), but closer to the estimate from Europe (0.87). Cyperaceae include many different genera and species and therefore comparison is problematic.
Our RPP value for Amaranthaceae/Chenopodiaceae has a large error estimate (0.18 ± 0.16) and, therefore, is not recommended for use in reconstructions. The value from Inner Mongolia is, in contrast, very high (21). In Europe, the only value available so far is 4.28 (±0.27) (Abraham and Kozáková 2012). The large differences between RPP estimates might be due to different species and vegetation-types between regions. Our value and that from Europe were obtained from studies in cultural landscapes of the temperate zone, while the very high value from Inner Mongolia is from meadow and steppe environments. In our study, Chenopodiaceae were represented by a few ruderals growing in cultivated fields, or cultivated crops (Spinacia oleracea), i.e. often removed before flowering. In Europe, the family is represented mainly by Chenopodium species growing in cultivated fields and along tracks and roads.
The value obtained in this study for Galium-type (1.23) is the first one in China and can only be compared with that of Europe (2.61).
In order to compare our RPP estimates for trees relative to Poaceae with those relative to Quercus (Li et al. 2015), we used the mean value of all Quercus RPP relative to Poaceae available in China to transform the values for Juglans and Ulmus (relative to Quercus) from Li et al. (2015) to values relative to Poaceae. We also reran the ERV-model with our data using Quercus as reference to check that differences between the two studies for RPP published values of Juglans and Ulmus were comparable to those found using the method above. The values for Quercus RPP relative to Poaceae are very close between our study (4.89), the mean for China (5.19) and the mean for Europe (5.83), which is reassuring. On the other hand, our RPP value for Juglans (0.3) is very low compared to that (10.06) of Li et al. (2015). This might be due to poor representation of pollen in the moss samples from this cultivated tree in our study area (i.e. the trees were seldom abundant and often young with perhaps a relatively poor flowering), while it is growing naturally in the woodlands of the Changbai Mountains and can be assumed to have a more abundant flowering if the trees are old. We therefore conclude that the RPP we obtained for Juglans is certainly not appropriate to use for pollen-based reconstruction of woodlands. As for Quercus, the RPP value for Ulmus in our study area (1.00) is close to the mean value from Europe (1.27), but it is much lower that the value from Li et al. (2015). This might again be related to the differences in species and landscapes between Europe and Shandong (human-induced vegetation and low abundance of Ulmus in both cases), and the Changbai mountains (dense woodlands and high abundance). Our RPP for Pinus (8.96) is similar to the European mean RPP (6.38). However, it should be stressed that the values of Pinus RPP in Europe vary a lot between latitudes and landscape-types.
Methodological differences between studies for vegetation data collection is another reason for significant differences in RPP estimates (Bunting and Hjelle 2010). Vegetation-survey strategies vary a lot between the published studies available from China. Differences in estimates of fall speed of pollen (FSP) for the same taxon can also have a significant effect on RPP estimates when the distance weighting method uses a Gaussian plume model (Theuerkauf et al. 2016). When FSP is calculated using Sutton’s equation the pollen grains of species belonging to a particular pollen taxon need to be measured. Pollen taxa can include a large number of species, such as Asteraceae SF Cichorioideae or Cyperaceae, therefore only some species are selected for measurements. The size of pollen grains will also differ depending on their origin (lake sediment samples, mosses or fresh plant material) and on the preparation methods and embedding (glycerine, silicone oil or others). For instance, the FSP of Quercus (0.026 m/s) in this study is similar to Gregory’s (1973) value, but it is lower than the value used in Europe (0.035 m/s) (Mazier et al. 2012), and higher than the value (0.018 m/s) provided by Li et al. (2015). The effect of these differences in FSP on the RPP values has not been tested in this study.
Conclusions
This is the first RPP study performed in cultural landscapes in China. Therefore, it is also a test of whether the methods and models used are appropriate in landscapes strongly impacted by humans. The theory of ERV models is based on a number of assumptions that are best met in natural vegetation or human-induced vegetation with relatively low fragmentation, i.e. vegetation with homogenous species composition and spatial distribution. Cultural landscapes are often very fragmented and may be characterized by sharp changes in species composition over a number of spatial scales. Nevertheless, several RPP studies in the cultural landscapes of Europe showed that it was possible to estimate relevant source area of pollen (RSAP) and RPPs in this kind of landscapes (e.g. Broström et al. 2005; Hellman et al. 2009a, b; Mazier et al. 2012). In our study, we find that the ERV models and the methods used for vegetation data collection are also appropriate for the highly fragmented and human-impacted cultural landscape of the Shandong province, which suggests that the methodology and models used are robust.
In this study, nine RPP estimates are new for China (Aster/Anthemis-type, Cannabis/Humulus, Caryophyllaceae, Castanea, Brassicaceae, Cupressaceae, Galium-type, Robinia/Sophora and Vitex negundo), and six are new for the world (Cannabis/Humulus, Castanea, Brassicaceae, Cupressaceae, Robinia/Sophora and Vitex negundo). RPP estimates for common taxa such as Quercus, Pinus and Artemisia are reasonably consistent in temperate China. Moreover, the values for Quercus, Pinus, Ulmus, Cyperaceae and Galium-type are comparable with the mean RPP in northern and central Europe. The values for Aster/Anthemis-type, Caryophyllaceae, Asteraceae SF Cichorioideae and Juglans differ from the few estimates available for China and/or Europe. Studies in Europe have shown that trees generally have higher RPP values than herbs (e.g. Broström et al. 2008; Mazier et al. 2012) and, therefore, pollen percentages or accumulation rates (PAR) overestimate the cover of trees in past vegetation (e.g. Trondman 2015). In this study we find that, as in Europe, trees (except Robinia/Sophora, 0.78 ± 0.03) have larger RPP estimates than herbs, except Artemisia (24.7 ± 0.36).
The taxa for which the RPP estimates are the first values published for China are common today in the cultural landscapes/human-induced vegetation of temperate north-western China. Therefore, these RPP estimates are of great value for pollen-based reconstructions of past human-induced vegetation cover in China. The latter is particularly valuable to test hypotheses related to land cover-climate interactions, archaeological questions, and plant diversity/vegetation dynamics in the past (e.g. Gaillard et al. 2010; Fyfe et al. 2013; Marquer et al. 2014; Hultberg et al. 2015). Although we could not estimate RPPs for cultivated crops, the RPPs for common ruderals in traditional cultivated land such as Artemisia, Aster/Anthemis-type, Caryophyllaceae, Asteraceae SF Cichorioideae, Cannabis/Humulus and Brassicaceae can be used to achieve an approximate quantification of cultivated land using Sugita’s (2007a, b) REVEALS and LOVE models.
New useful RPP to estimate the cover of woodland are those for Robinia/Sophora and Cupresssaceae. Robinia was introduced to China between CE 1877–1878 from Japan. However, the pollen morphology of Robinia is very similar to that of Sophora, an indigenous taxon in China. Unfortunately, palynologists have seldom separated this pollen-type. A separation of Robinia/Sophora would be important in the future, as well as identification of other pollen-types in the Fabaceae family. The RPP obtained for Cupressaceae is based on pollen and vegetation data from a single genus, Platycladus spp. It is an indigenous taxon, although it is/has also been planted widely in the mountains above the cultivated terraces in recent years. The pollen morphology of Platycladus is hard to separate from other Cupressaceae, and is seldom separated as a genus in pollen records. Therefore, our RPP for Cupressaceae should be used with care when the species involved are not known. However, it is interesting to note that the RPP estimate for Cupressaceae in China is comparable to the estimate for Juniperus in Europe.
References
Abraham V, Kozáková R (2012) Relative pollen productivity estimates in the modern agricultural landscape of Central Bohemia (Czech Republic). Rev Palaeobot Palynol 179:1–12
Baker AG, Keczyński A, Bhagwat SA, Willis KJ, Latałowa M (2016) Pollen productivity estimates from old-growth forest strongly differ from those obtained in cultural landscapes: evidence from the Białowieża National Park, Poland. Holocene 26:80–92
Beug H-J (2004) Leitfaden der Pollenbestimmung für Mitteleuropa und angrenzende Gebiete. Pfeil, München
Broström A, Sugita S, Gaillard M-J (2004) Pollen productivity estimates for the reconstruction of past vegetation cover in the cultural landscape of southern Sweden. Holocene 14:368–381
Broström A, Sugita S, Gaillard M-J, Pilesjö P (2005) Estimating the spatial scale of pollen dispersal in the cultural landscape of southern Sweden. Holocene 15:252–262
Broström A, Nielsen AB, Gaillard M-J et al (2008) Pollen productivity estimates of key European plant taxa for quantitative reconstruction of past vegetation: a review. Veget Hist Archaeobot 17:461–478
Bunting MJ (2003) Pollen-vegetation relationships in non-arboreal moorland taxa. Rev Palaeobot Palynol 125:285–298
Bunting MJ, Hjelle KL (2010) Effect of vegetation data collection strategies on estimates of relevant source area of pollen (RSAP) and relative pollen productivity estimates (relative PPE) for non-arboreal taxa. Veget Hist Archaeobot 19:365–374
Bunting MJ, Middleton D (2005) Modelling pollen dispersal and deposition using HUMPOL software, including simulating windroses and irregular lakes. Rev Palaeobot Palynol 134:185–196
Bunting MJ, Gaillard MJ, Sugita S, Middleton R, Broström A (2004) Vegetation structure and pollen source area. Holocene 14:651–660
Bunting MJ, Schofield JE, Edwards KJ (2013a) Estimates of relative pollen productivity (RPP) for selected taxa from southern Greenland: a pragmatic solution. Rev Palaeobot Palynol 190:66–74
Bunting MJ, Farrell M, Broström A et al (2013b) Palynological perspectives on vegetation survey: a critical step for model-based reconstruction of Quaternary land cover. Quat Sci Rev 82:41–55
Calcote R (1995) Pollen source area and pollen productivity: evidence from forest hollows. J Ecol 83:591–602
Commerford JL, McLauchlan KK, Sugita S (2013) Calibrating vegetation cover and grassland pollen assemblages in the Flint Hills of Kansas, USA. Am J Plant Sci 4:10
Cui QY, Gaillard MJ, Lemdahl G, Stenberg L, Sugita S, Zernova G (2014) Historical land-use and landscape change in southern Sweden and implications for present and future biodiversity. Ecol Evol 4:3,555-3,570
Duffin KI, Bunting MJ (2007) Relative pollen productivity and fall speed estimates for southern African savanna taxa. Veget Hist Archaeobot 17:507–525
Fægri K, Iversen J (1989) Textbook of pollen analysis. Wiley, Chichester
Fu C (2003) Potential impacts of human-induced land cover change on East Asia monsoon. Glob Planet Chang 37:219–229
Fyfe RM, Twiddle C, Sugita S et al (2013) The Holocene vegetation cover of Britain and Ireland: overcoming problems of scale and discerning patterns of openness. Quat Sci Rev 73:132–148
Gaillard M-J, Sugita S, Bunting MJ et al (2008) The use of modelling and simulation approach in reconstructing past landscapes from fossil pollen data: a review and results from the POLLANDCAL network. Veget Hist Archaeobot 17:419–443
Gaillard M-J, Sugita S, Mazier F et al (2010) Holocene land-cover reconstructions for studies on land cover-climate feedbacks. Clim Past 6:483–499
Ge Y, Li Y, Yang X et al (2015) Relevant source area of pollen and relative pollen productivity estimates in Bashang steppe. Quat Sci 35:934–945 (Chinese with English abstract)
Gregory PH (1973) Spores: their properties and sedimentation in still air. Microbiology of the atmosphere. A plant science monograph. Leonard Hill, Aylesbury
Hellman S, Gaillard M-J, Broström A, Sugita S (2008) The REVEALS model, a new tool to estimate past regional plant abundance from pollen data in large lakes: validation in southern Sweden. J Quat Sci 23:21–42
Hellman S, Bunting MJ, Gaillard M-J (2009a) Relevant source area of pollen in patchy cultural landscapes and signals of anthropogenic landscape disturbance in the pollen record: a simulation approach. Rev Palaeobot Palynol 153:245–258
Hellman S, Gaillard M-J, Bunting JM, Mazier F (2009b) Estimating the relevant source area of pollen in the past cultural landscapes of southern Sweden—a forward modelling approach. Rev Palaeobot Palynol 153:259–271
Hjelle K (1998) Herb pollen representation in surface moss samples from mown meadows and pastures in western Norway. Holocene 7:79–96
Hjelle KL, Sugita S (2012) Estimating pollen productivity and relevant source area of pollen using lake sediments in Norway: how does lake size variation affect the estimates? Holocene 22:313–324
Hultberg T, Gaillard M-J, Grundmann B, Lindbladh M (2015) Reconstruction of past landscape openness using the landscape reconstruction algorithm (LRA) applied on three local pollen sites in a southern Swedish biodiversity hotspot. Veget Hist Archaeobot 24:253–266
Karlsson H, Bunting MJ, Middleton R (2008) Quantitative estimation of pre-settlement forest cover using the multiple scenario approach at a Stállo settlement site in the Swedish Scandes. In: Karlsson H (ed) Vegetation changes and forest-line positions in the Swedish Scandes during late Holocene. Diss. (sammanfattning/summary). Sveriges lantbruksuniv. Acta Universitatis agriculturae Sueciae, Umeå
Li Y, Bunting MJ, Xu Q et al (2011) Pollen-vegetation-climate relationships in some desert and desert-steppe communities in northern China. Holocene 21:997–1,010
Li Y, Nielsen AB, Zhao X et al (2015) Pollen production estimates (PPEs) and fall speeds for major tree taxa and relevant source areas of pollen (RSAP) in Changbai Mountain, northeastern China. Rev Palaeobot Palynol 216:92–100
Mariani M, Connor SE, Theuerkauf M, Kuneš P, Fletcher MS (2016) Testing quantitative pollen dispersal models in animal-pollinated vegetation mosaics: An example from temperate Tasmania, Australia. Quat Sci Rev 154:214–225
Marquer L, Gaillard M-J, Sugita S et al (2014) Holocene changes in vegetation composition in northern Europe: why quantitative pollen-based vegetation reconstructions matter. Quat Sci Rev 90:199–216
Mazier F, Broström A, Gaillard M-J et al (2008) Pollen productivity estimates and relevant source area of pollen for selected plant taxa in a pasture woodland landscape of the Jura Mountains (Switzerland). Veget Hist Archaeobot 17:479–495
Mazier F, Gaillard M-J, Kuneš P et al (2012) Testing the effect of site selection and parameter setting on REVEALS-model estimates of plant abundance using the Czech Quaternary Palynological Database. Rev Palaeobot Palynol 187:38–49
Mazier F, Broström A, Bragée P, Fredh D, Stenberg L, Thiere G, Sugita S, Hammarlund D (2015) Two hundred years of land-use change in the South Swedish Uplands: comparison of historical map-based estimates with a pollen-based reconstruction using the landscape reconstruction algorithm. Veget Hist Archaeobot 24:555–570
McNown JS, Malaika J (1950) Effects of particle shape on settling velocity at low Reynolds numbers. Am Geophys Union Trans 31:74–82
Nielsen AB (2004) Modelling pollen sedimentation in Danish lakes at c. AD 1800: an attempt to validate the POLLSCAPE model. J Biogeogr 31:1,693-1,709
Nielsen AB, Odgaard BV (2010) Quantitative landscape dynamics in Denmark through the last three millennia based on the landscape reconstruction algorithm approach. Veget Hist Archaeobot 19:375–387
Nielsen AB, Sugita S (2005) Estimating relevant source area of pollen for small Danish lakes around AD 1800. Holocene 15:1,006–1,020
Overballe-Petersen MV, Nielsen AB, Bradshaw RHW (2013) Quantitative vegetation reconstruction from pollen analysis and historical inventory data around a Danish small forest hollow. J Veget Sci 24:755–771
Parsons RW, Prentice IC (1981) Statistical approaches to R-values and the pollen—vegetation relationship. Rev Palaeobot Palynol 32:127–152
Poska A, Meltsov V, Sugita S, Vassiljev J (2011) Relative pollen productivity estimates of major anemophilous taxa and relevant source area of pollen in a cultural landscape of the hemi-boreal forest zone (Estonia). Rev Palaeobot Palynol 167:30–39
Prentice IC (1985) Pollen representation, source area, and basin size: Toward a unified theory of pollen analysis. Quat Res 23:76–86
Prentice IC, Parsons RW (1983) Maximum likelihood linear calibration of pollen spectra in terms of forest composition. Biometrics 39:1,051–1,057
Punt W et al (1976–2009) The Northwest European Pollen Flora (NEPF) Vol I (1976), Vol II (1980), Vol III (1981), Vol IV (1984) Vol V (1988), Vol VI (1991), Vol VII (1996), Vol VIII (2003), Vol IX (2009). Elsevier, Amsterdam
Räsänen S, Suutari H, Nielsen AB (2007) A step further towards quantitative reconstruction of past vegetation in Fennoscandian boreal forests: Pollen productivity estimates for six dominant taxa. Rev Palaeobot Palynol 146:208–220
Reille M (1995) Pollen et spores d’Europe et d’Afrique du Nord. Suppl 1. Laboratoire de Botanique Historique et Palynologie, Marseille
Soepboer W, Sugita S, Lotter AF et al (2007) Pollen productivity estimates for quantitative reconstruction of vegetation cover on the Swiss Plateau. Holocene 17:65–77
Strandberg G, Kjellström E, Poska A et al (2014) Regional climate model simulations for Europe at 6 and 0.2 k bp: sensitivity to changes in anthropogenic deforestation. Clim Past 10:661–680
Sugita S (1993) A model of pollen source area for an entire lake surface. Quat Res 39:239–244
Sugita S (1994) Pollen representation of vegetation in Quaternary sediment: theory and method in patchy vegetation. Ecology 82:881–897
Sugita S (2007a) Theory of quantitative reconstruction of vegetation I: pollen from large sites REVEALS regional vegetation composition. Holocene 17:229–241
Sugita S (2007b) Theory of quantitative reconstruction of vegetation II: all you need is LOVE. Holocene 17:243–257
Sugita S, Gaillard MJ, Broström A (1999) Landscape openness and pollen records: a simulation approach. Holocene 9:409–421
Sugita S, Parshall T, Calcote R, Walker K (2010) Testing the Landscape Reconstruction Algorithm for spatially explicit reconstruction of vegetation in northern Michigan and Wisconsin. Quat Res 74:289–300
Sutton OG (1953) Micrometeorology. McGraw-Hill, New York
Theuerkauf M, Couwenberg J, Kuparinen A, Liebscher V (2016) A matter of dispersal: REVEALSinR introduces state-of-the-art dispersal models to quantitative vegetation reconstruction. Veget Hist Archaeobot 25:541–553
Trondman A-K (2015) Pollen-based quantitative reconstructions of Holocene regional vegetation cover (plant-functional-types and land-cover-types) in Europe suitable for climate modelling. Glob Chang Biol 21:676–697. doi:10.1111/gcb.12737
Trondman A-K, Gaillard M-J, Sugita S et al (2016) Are pollen records from small sites appropriate for REVEALS model-based quantitative reconstructions of past regional vegetation? An empirical test in southern Sweden. Veget Hist Archaeobot 25:131–151
Von Stedingk H, Fyfe RM, Allard A (2008) Pollen productivity estimates from the forest–tundra ecotone in west-central Sweden: implications for vegetation reconstruction at the limits of the boreal forest. Holocene 18:323–332
Wang F (1995) Pollen flora of China. Science Press, Beijing (Chinese)
Wang Y, Herzschuh U (2011) Reassessment of Holocene vegetation change on the upper Tibetan Plateau using the pollen-based REVEALS model. Rev Palaeobot Palynol 168:31–40
Wu J, Ma YZ, Sang YL, Meng HW, Hu CL (2013) Quantitative reconstruction of palaeovegetation and development of the R-value model: an application of R-value and ERV model in Xinglong Mountain natural protection region. Quat Sci 33:554–564 (Chinese with English abstract)
Xu Q, Cao X, Tian F et al (2014) Relative pollen productivities of typical steppe species in northern China and their potential in past vegetation reconstruction. Science China, Serie D, Earth Sciences 57:1,254–1,266
Acknowledgements
This study would not have been possible without the help of numerous colleagues and students helping to collect data in field, Carole Cugny, Elodie Faure, and Laurent Marquer (Toulouse University, France), and Huishuang Mu, Panpan Zhang, Shengrui Zhang, Jie Li, Yang Li and Yanan Hu (Hebei Normal University, Shijiazhuang, China). We thank Qiao-Yu Cui (Academy of Sciences, Beijing, China) for her useful comments on an earlier version of the manuscript. The project was supported by a doctoral student grant for Furong Li from the China Scholarship Council (Grant number 201206180028), funds from the Faculty of Health and Life Sciences of Linnaeus University, Kalmar Sweden (https://lnu.se/en/meet-linnaeus-university/Organisation/faculty-of-health-and-life-sciencesnew-page/), the Swedish Strategical Research Area “ModElling the Regional and Global Ecosystem, MERGE” (http://www.merge.lu.se/) and PAGES (http://www.pastglobalchanges.org). The study is a contribution to the PAGES LandCover6k working group (http://www.pastglobalchanges.org/ini/wg/landcover6k/intro).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by F. Bittmann.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Li, F., Gaillard, MJ., Sugita, S. et al. Relative pollen productivity estimates for major plant taxa of cultural landscapes in central eastern China. Veget Hist Archaeobot 26, 587–605 (2017). https://doi.org/10.1007/s00334-017-0636-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00334-017-0636-9