Abstract
Objectives
This study was conducted in order to investigate whether there is a correlation between the time-to-enhancement (TTE) in ultrafast MRI and histopathological characteristics of breast cancers.
Methods
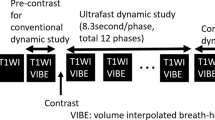
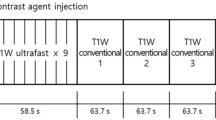
Between January and August 2017, 274 consecutive breast cancer patients (mean age, 53.5 years; range, 25–80 years) who underwent ultrafast MRI and subsequent surgery were included for analysis. Ultrafast MRI scans were acquired using TWIST-VIBE or 4D TRAK-3D TFE sequences. TTE and maximum slope (MS) were derived from the ultrafast MRI. The repeated measures ANOVA, Mann–Whitney U test and Kruskal–Wallis H test were performed to compare the median TTE, MS and SER according to histologic type, histologic grade, ER/PR/HER2 positivity, level of Ki-67 and tumour subtype. For TTE calculation, intraclass correlation coefficient (ICC) was used to evaluate interobserver variability.
Results
The median TTE of invasive cancers was shorter than that of in situ cancers (p < 0.001). In invasive cancers, large tumours showed shorter TTE than small tumours (p = 0.001). High histologic/nuclear grade cancers had shorter TTE than low to intermediate grade cancers (p < 0.001 and p < 0.001). HER2-positive cancers showed shorter TTE than HER2-negative cancers (p = 0.001). The median TTE of cancers with high Ki-67 was shorter than that of cancers with low Ki-67 (p < 0.001). ICC between two readers showed moderate agreement (0.516). No difference was found in the median MS or SER values according to the clinicopathologic features.
Conclusions
The median TTE of breast cancer in ultrafast MRI was shorter in invasive or aggressive tumours than in in situ cancer or less aggressive tumours, respectively.
Key Points
• Invasive breast tumours show a shorter TTE in ultrafast DCE-MRI than in situ cancers.
• A shorter TTE in ultrafast DCE-MRI is associated with breast tumours of a large size, high histologic or nuclear grade, PR negativity, HER2 positivity and high Ki-67 level.




Similar content being viewed by others
Abbreviations
- DCE:
-
Dynamic contrast-enhanced
- ER:
-
Oestrogen receptor
- eTHRIVE:
-
Enhanced T1-weighted high-resolution isotropic volume examination
- HER2:
-
Human epidermal growth factor receptor (HER)-2
- IHC:
-
Immunohistochemistry
- MRI:
-
Magnetic resonance imaging
- MS:
-
Maximum slope
- PR:
-
Progesterone receptor
- ROI:
-
Region of interest
- SER:
-
Signal enhancement ratio
- TFE:
-
Turbo field echo
- TRAK:
-
Time-resolved MR angiography with keyhole
- TTE:
-
Time-to-enhancement
- TWIST:
-
Time-resolved angiography with interleaved stochastic trajectories
- VIBE:
-
Volume-interpolated breath-hold examination
References
Mann RM, Mus RD, van Zelst J, Geppert C, Karssemeijer N, Platel B (2014) A novel approach to contrast-enhanced breast magnetic resonance imaging for screening: high-resolution ultrafast dynamic imaging. Invest Radiol 49:579–585
Herrmann KH, Baltzer PA, Dietzel M et al (2011) Resolving arterial phase and temporal enhancement characteristics in DCE MRM at high spatial resolution with TWIST acquisition. J Magn Reson Imaging 34:973–982
Pineda FD, Medved M, Wang S et al (2016) Ultrafast bilateral DCE-MRI of the breast with conventional Fourier sampling: preliminary evaluation of semi-quantitative analysis. Acad Radiol 23:1137–1144
Platel B, Mus R, Welte T, Karssemeijer N, Mann R (2014) Automated characterization of breast lesions imaged with an ultrafast DCE-MR protocol. IEEE Trans Med Imaging 33:225–232
Mus RD, Borelli C, Bult P et al (2017) Time to enhancement derived from ultrafast breast MRI as a novel parameter to discriminate benign from malignant breast lesions. Eur J Radiol 89:90–96
Abe H, Mori N, Tsuchiya K et al (2016) Kinetic analysis of benign and malignant breast lesions with ultrafast dynamic contrast-enhanced MRI: comparison with standard kinetic assessment. AJR Am J Roentgenol 207:1159–1166
Onishi N, Sadinski M, Gibbs P et al (2019) Differentiation between subcentimeter carcinomas and benign lesions using kinetic parameters derived from ultrafast dynamic contrast-enhanced breast MRI. Eur Radiol. https://doi.org/10.1007/s00330-019-06392-5
Goto M, Sakai K, Yokota H et al (2019) Diagnostic performance of initial enhancement analysis using ultra-fast dynamic contrast-enhanced MRI for breast lesions. Eur Radiol 29:1164–1174
Szabo BK, Aspelin P, Kristoffersen Wiberg M, Tot T, Bone B (2003) Invasive breast cancer: correlation of dynamic MR features with prognostic factors. Eur Radiol 13:2425–2435
Tuncbilek N, Karakas HM, Okten OO (2005) Dynamic magnetic resonance imaging in determining histopathological prognostic factors of invasive breast cancers. Eur J Radiol 53:199–205
Baltzer PA, Vag T, Dietzel M et al (2010) Computer-aided interpretation of dynamic magnetic resonance imaging reflects histopathology of invasive breast cancer. Eur Radiol 20:1563–1571
Leong LC, Gombos EC, Jagadeesan J, Fook-Chong SM (2015) MRI kinetics with volumetric analysis in correlation with hormonal receptor subtypes and histologic grade of invasive breast cancers. AJR Am J Roentgenol 204:W348–W356
Nam SY, Ko ES, Lim Y et al (2018) Preoperative dynamic breast magnetic resonance imaging kinetic features using computer-aided diagnosis: association with survival outcome and tumor aggressiveness in patients with invasive breast cancer. PLoS One 13:e0195756
Kim JJ, Kim JY, Kang HJ et al (2017) Computer-aided diagnosis-generated kinetic features of breast cancer at preoperative MR imaging: association with disease-free survival of patients with primary operable invasive breast cancer. Radiology 284:45–54
Heacock L, Melsaether AN, Heller SL et al (2016) Evaluation of a known breast cancer using an abbreviated breast MRI protocol: correlation of imaging characteristics and pathology with lesion detection and conspicuity. Eur J Radiol 85:815–823
Heacock L, Lewin AA, Gao Y et al (2018) Feasibility analysis of early temporal kinetics as a surrogate marker for breast tumor type, grade, and aggressiveness. J Magn Reson Imaging 47:1692–1700
Perou CM, Sorlie T, Eisen MB et al (2000) Molecular portraits of human breast tumours. Nature 406:747–752
Wolff AC, Hammond ME, Hicks DG et al (2013) Recommendations for human epidermal growth factor receptor 2 testing in breast cancer: American Society of Clinical Oncology/College of American Pathologists clinical practice guideline update. J Clin Oncol 31:3997–4013
Morris EA, Comstock CE, Lee CH et al (2013) ACR BI-RADS® magnetic resonance imaging. In: ACR BI-RADS® atlas, breast imaging reporting and data system. American College of Radiology, Reston
Mori N, Abe H, Mugikura S et al (2018) Ultrafast dynamic contrast-enhanced breast MRI: kinetic curve assessment using empirical mathematical model validated with histological microvessel density. Acad Radiol. https://doi.org/10.1016/j.acra.2018.08.016
Youden WJ (1950) Index for rating diagnostic tests. Cancer 3:32–35
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159–174
Koo HR, Cho N, Song IC et al (2012) Correlation of perfusion parameters on dynamic contrast-enhanced MRI with prognostic factors and subtypes of breast cancers. J Magn Reson Imaging 36:145–151
Kim JY, Kim SH, Kim YJ et al (2015) Enhancement parameters on dynamic contrast enhanced breast MRI: do they correlate with prognostic factors and subtypes of breast cancers? Magn Reson Imaging 33:72–80
Grimm LJ, Soo MS, Yoon S, Kim C, Ghate SV, Johnson KS (2015) Abbreviated screening protocol for breast MRI: a feasibility study. Acad Radiol 22:1157–1162
Kuhl CK, Schrading S, Strobel K, Schild HH, Hilgers RD, Bieling HB (2014) Abbreviated breast magnetic resonance imaging (MRI): first postcontrast subtracted images and maximum-intensity projection—a novel approach to breast cancer screening with MRI. J Clin Oncol 32:2304–2310
Mango VL, Morris EA, David Dershaw D et al (2015) Abbreviated protocol for breast MRI: are multiple sequences needed for cancer detection? Eur J Radiol 84:65–70
Sheth D, Abe H (2017) Abbreviated MRI and accelerated MRI for screening and diagnosis of breast cancer. Top Magn Reson Imaging 26:183–189
Su MY, Cheung YC, Fruehauf JP et al (2003) Correlation of dynamic contrast enhancement MRI parameters with microvessel density and VEGF for assessment of angiogenesis in breast cancer. J Magn Reson Imaging 18:467–477
Oldrini G, Fedida B, Poujol J et al (2017) Abbreviated breast magnetic resonance protocol: value of high-resolution temporal dynamic sequence to improve lesion characterization. Eur J Radiol 95:177–185
Song T, Laine AF, Chen Q et al (2009) Optimal k-space sampling for dynamic contrast-enhanced MRI with an application to MR renography. Magn Reson Med 61:1242–1248
Funding
This study has received funding by a grant (no. 04-2017-0470) from the Seoul National University Hospital Research Fund.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Nariya Cho MD, PhD, Professor of the Department of Radiology, Seoul National University Hospital.
Conflict of interest
The authors declare that they have no conflict of interest.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was waived by the Institutional Review Board.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• Retrospective
• Observational
• Performed at one institution
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 70.5 kb)
Rights and permissions
About this article
Cite this article
Shin, S.U., Cho, N., Kim, SY. et al. Time-to-enhancement at ultrafast breast DCE-MRI: potential imaging biomarker of tumour aggressiveness. Eur Radiol 30, 4058–4068 (2020). https://doi.org/10.1007/s00330-020-06693-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-020-06693-0