Abstract
Objectives
To demonstrate, in patients with cystic fibrosis (CF), the correlation between three-dimensional dynamic oxygen-enhanced magnetic resonance imaging (OE-MRI) measurements and computed tomography Brody score (CF-CT) and lung function testing (LFT).
Methods
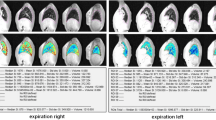
Twenty-one patients (median age, 25 years; female, n = 8) with a range of CF lung disease and five healthy volunteers (median age, 31 years; female, n = 2) underwent OE-MRI performed on a 1.5-T MRI scanner. Coronal volumes were acquired while patients alternately breathed room air and 100% oxygen. Pre-oxygen T1 was measured. Dynamic series of T1-weighted volumes were then obtained while breathing oxygen. T1-parameter maps were generated and the following OE-MRI parameters were measured: oxygen uptake (ΔPO2max), wash-in time and wash-out time. High-resolution CT and LFT were performed. The relationship between CF-CT, LFT and OE-MRI parameters were evaluated using Pearson correlation for the whole lung and regionally.
Results
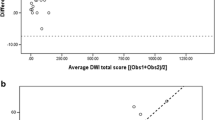
Mean CF-CT was 24.1±17.1. Mean ΔPO2max and mean wash-in as well as skewness of wash-out showed significant correlation with CF-CT (ΔPO2max: r = -0.741, p < 0.001; mean wash-in: r = 0.501, p = 0.017; skewness of wash-out: r = 0.597, p = 0.001). There was significant correlation for the whole lung and regionally between LFT parameters and OE-MR (ΔPO2max: r = 0.718, p < 0.001; wash-in: r = -0.576, p = 0.003; wash-out skewness: r = -0.552, p = 0.004).
Conclusions
Functional lung imaging using OE-MRI has the capability to assess the severity of CF lung disease and shows a significant correlation with LFT and CF-CT.
Key points
• Oxygen-enhanced MRI might play a future role in evaluation and follow-up of cystic fibrosis.
• Heterogeneity of parameter maps reflects localised functional impairment in cystic fibrosis.
• Avoidance of cumulative radiation burden in CF is feasible using OE-MRI.



Similar content being viewed by others
Change history
12 June 2018
The original version of this article, published on 13 April 2018, unfortunately contained a mistake.
Abbreviations
- ΔPO2max :
-
Delta partial pressure of oxygen in parenchyma
- AT-CT:
-
Subscore for air trapping
- BR-CT:
-
Subscore for bronchial changes
- CF:
-
Cystic fibrosis
- CF-CT:
-
Computed tomography Brody score
- ECFS:
-
CT Standards of Care of the European CF Society
- FEV1 :
-
Forced expiratory volume in 1 s
- FVC:
-
Forced vital capacity
- HRCT:
-
High-resolution computed tomography
- LFT:
-
Lung function test
- MMEF:
-
Maximum mid-expiratory flow
References
Smyth AR, Bell SC, Bojcin S, Bryon M, Duff A, Flume P et al (2014) European Cystic Fibrosis Society Standards of Care: Best Practice guidelines. J Cyst Fibros 13:S23–S42
Marshall H, Horsley A, Taylor CJ, Smith L, Hughes D, Horn FC et al (2017) Detection of early subclinical lung disease in children with cystic fibrosis by lung ventilation imaging with hyperpolarised gas MRI. Thorax 72:760–762
van Beek EJ, Wild JM, Kauczor HU, Schreiber W, Mugler JP 3rd, de Lange EE (2004) Functional MRI of the lung using hyperpolarized 3-helium gas. J Magn Reson Imaging 20:540–554
Schreiber WG, Morbach AE, Stavngaard T, Gast KK, Herweling A, Sogaard LV et al (2005) Assessment of lung microstructure with magnetic resonance imaging of hyperpolarized Helium-3. Respir Physiol Neurobiol 148:23-42
Heussel CP, Gast KK, Dahmen A, Morbach AE, Kauczor HU, Schreiber WG et al (2005) Hyperpolarized (3)helium gas for functional magnetic resonance imaging of the lung. Med Klin (Munich) 100:413–424
Flors L, Mugler JP 3rd, de Lange EE, Miller GW, Mata JF, Tustison N et al (2016) Hyperpolarized gas magnetic resonance lung imaging in children and young adults. J Thorac Imaging 31:285–295
Edelman RR, Hatabu H, Tadamura E, Li W, Prasad PV (1996) Noninvasive assessment of regional ventilation in the human lung using oxygen-enhanced magnetic resonance imaging. Nat Med 2:1236-1239
Jakob PM, Wang T, Schultz G, Hebestreit H, Hebestreit A, Hahn D (2004) Assessment of human pulmonary function using oxygen-enhanced T(1) imaging in patients with cystic fibrosis. Magn Reson Med 51:1009–1016
Naish JH, Parker GJ, Beatty PC, Jackson A, Young SS, Waterton JC et al (2005) Improved quantitative dynamic regional oxygen-enhanced pulmonary imaging using image registration. Magn Reson Med 54:464–469
Brody AS, Klein JS, Molina PL, Quan J, Bean JA, Wilmott RW (2009) High-resolution computed tomography in young patients with cystic fibrosis: distribution of abnormalities and correlation with pulmonary function tests. J Pediatr 145:32–38
Meyers CR, Blesh TE (1962) Measurement in physical education. Ronald Press, New York
Zhang WJ, Niven RM, Young SS, Liu YZ, Parker GJ, Naish JH (2015) Dynamic oxygen-enhanced magnetic resonance imaging of the lung in asthma—initial experience. Eur J Radiol 84:318–326
Chen Q, Jakob PM, Griswold MA, Levin DL, Hatabu H, Edelman RR (1998) Oxygen enhanced MR ventilation imaging of the lung. MAGMA 7:153–161
Ohno Y, Hatabu H, Takenaka D, Adachi S, Van Cauteren M, Sugimura K (2001) Oxygen-enhanced MR ventilation imaging of the lung. AJR Am J Roentgenol 177:185–194
Ohno Y, Hatabu H, Takenaka D, Van Cauteren M, Fujii M, Sugimura K (2002) Dynamic oxygen-enhanced MRI reflects diffusing capacity of the lung. Magn Reson Med 47:1139–1144
Ramsey BW (1996) Management of pulmonary disease in patients with cystic fibrosis. N Engl J Med 335:179–188
Schaefer JF, Hector A, Schmidt K, Teufel M, Fleischer S, Graepler-Mainka U et al (2018) A semiquantitative MRI-Score can predict loss of lung function in patients with cystic fibrosis: Preliminary results. Eur Radiol 28:74–78
Ciet P, Bertolo S, Ros M, Andrinopoulou ER, Tavano V, Lucca F et al (2017) Detection and monitoring of lung inflammation in cystic fibrosis during respiratory tract exacerbation using diffusion-weighted magnetic resonance imaging. Eur Respir J 50(1). https://doi.org/10.1183/13993003.01437-2016
Grasemann H, Ciet P, Amin R, McDonald N, Klingel M, Tiddens H et al (2017) Changes in magnetic resonance imaging scores and ventilation inhomogeneity in children with cystic fibrosis pulmonary exacerbations. Eur Respir J 50(2). https://doi.org/10.1183/13993003.00244-2017
Acknowledgements
Support and the provision of research features from Philips Clinical Science is gratefully acknowledged.
Funding
This study has received funding by the Swiss Lung association (Lungenliga Schweiz).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Prof. Thomas Frauenfelder.
Conflict of interest
The authors Dr. Martini, Ms. Gygax, Dr. Benden and Dr. Frauenfelder declare no relationships with any companies, whose products or services may be related to the subject matter of the article.
The authors of this manuscript declare relationships with the following companies: Dr. Morgan reports personal fees from Bioxydyn Ltd, during the conduct of the study; personal fees from GSK, outside the submitted work.
Dr. Parker reports personal fees from Bioxydyn Ltd, during the conduct of the study. In addition, Dr. Parker has a patent PCT/GB2008/001390 issued, and a patent PCT/GB2010/001989 issued.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional Review Board approval was obtained.
Methodology
• prospective
• diagnostic or prognostic study
• performed at one institution
Electronic supplementary material
ESM 1
(DOCX 74 kb)
Rights and permissions
About this article
Cite this article
Martini, K., Gygax, C.M., Benden, C. et al. Volumetric dynamic oxygen-enhanced MRI (OE-MRI): comparison with CT Brody score and lung function in cystic fibrosis patients. Eur Radiol 28, 4037–4047 (2018). https://doi.org/10.1007/s00330-018-5383-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-018-5383-5