Abstract
Objectives
To compare combined percutaneous radiofrequency ablation and ethanol injection (RFA-PEI) with hepatic resection (HR) in the treatment of resectable solitary hepatocellular carcinoma (HCC) with 2.1–5.0 cm diameter.
Methods
From June 2009 to December 2015, 271 patients whom underwent RFA-PEI (n = 141) or HR (n = 130) in three centres were enrolled. The overall survival (OS) and recurrence-free survival (RFS) between groups were compared with Kaplan–Meier method and log-rank tests. Complications, hospital stay and cost were assessed.
Results
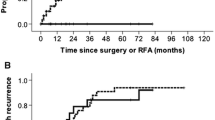
The OS rates at 1, 3 and 5 years were 93.5%, 72.7%, 58.6% in RFA-PEI group and 82.3%, 57.5%, 51.8% in HR group (p = 0.021). The corresponding 1-, 3- and 5-year RFS rates were 65.8%, 41.3%, 34.3% in RFA-PEI group and 50.5%, 33.8%, 28.4% in HR group (p = 0.038). For patients with 2.1–3.0 cm tumours, the 1-, 3- and 5-year OS after RFA-PEI and HR were 98.0%, 82.3%, 74.2% and 89.4%, 65.1%, 61.9%, respectively (p = 0.024). The corresponding RFS were 79.6%, 54.7%, 45.1% in RFA-PEI group, and 57.6%, 43.9%, 31.7% in HR group, respectively (p = 0.020). RFA-PEI was superior to HR in major complication rates, length of hospital stay and cost (all p < 0.001).
Conclusion
RFA-PEI had a survival benefit over HR in the treatment of solitary HCCs, especially for those with 2.1–3.0 cm in diameter.
Key Points
• RFA-PEI provided superior survival to HR in solitary HCC with 2.1–5.0 cm in diameter.
• RFA-PEI is superior to HR in complications, length of hospital stay and cost.
• RFA-PEI might be an alternative treatment for solitary HCC within 5.0 cm in diameter.



Similar content being viewed by others
Abbreviations
- BCLC:
-
Barcelona clinic liver cancer
- CECT:
-
Contrast-enhanced computed tomography
- CEUS:
-
Contrast-enhanced ultrasound
- ECOG:
-
Eastern cooperative oncology group
- HCC:
-
Hepatocellular carcinoma
- HR:
-
Hepatic resection
- LTP:
-
Local tumour progression
- MWA:
-
Microwave ablation
- OS:
-
Overall survival
- PEI:
-
Percutaneous ethanol injection
- RCT:
-
Randomized controlled trial
- RFA:
-
Radiofrequency ablation
- RFS:
-
Recurrence-free survival
- TACE:
-
Transarterial chemoembolization
References
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. CA Cancer J Clin 61:69–90
European Association for the Study of the Liver; European Organisation for Research and Treatment of Cancer (2012) EASL-EORTC clinical practice guidelines: management of hepatocellular carcinoma. J Hepatol 56:908–943
Bruix J, Sherman M (2011) Management of hepatocellular carcinoma: an update. Hepatology 53:1020–1022
O’Leary JG, Lepe R, Davis GL (2008) Indications for liver transplantation. Gastroenterology 134:1764–1776
Bruix J, Reig M, Sherman M (2016) Evidence-based diagnosis, staging, and treatment of patients with hepatocellular carcinoma. Gastroenterology 150:835–853
Chen MS, Li JQ, Zheng Y et al (2006) A prospective randomized trial comparing percutaneous local ablative therapy and partial hepatectomy for small hepatocellular carcinoma. Ann Surg 243:321–328
Feng K, Yan J, Li XW et al (2012) A randomized controlled trial of radiofrequency ablation and surgical resection in the treatment of small hepatocellular carcinoma. J Hepatol 57:794–802
Liang HH, Chen MS, Peng ZW et al (2008) Percutaneous radiofrequency ablation versus repeat hepatectomy for recurrent hepatocellular carcinoma: a retrospective study. Ann Surg Oncol 15:3484–3493
Huang G, Lin M, Xie X et al (2014) Combined radiofrequency ablation and ethanol injection with a multipronged needle for the treatment of medium and large hepatocellular carcinoma. Eur Radiol 24:1565–1571
Zhang YJ, Liang HH, Chen MS et al (2007) Hepatocellular carcinoma treated with radiofrequency ablation with or without ethanol injection: a prospective randomized trial. Radiology 244:599–607
Vallone P, Catalano O, Izzo F, Siani A (2006) Combined ethanol injection therapy and radiofrequency ablation therapy in percutaneous treatment of hepatocellular carcinoma larger than 4 cm. Cardiovasc Intervent Radiol 29:544–551
Bruix J, Sherman M, Llovet JM et al (2001) Clinical management of hepatocellular carcinoma. Conclusions of the Barcelona-2000 EASL conference. European Association for the Study of the Liver. J Hepatol 35:421–430
Bruix J, Sherman M, Practice Guidelines Committee, American Association for the Study of Liver Diseases (2005) Management of hepatocellular carcinoma. Hepatology 42:1208–1236
Kuang M, Lu MD, Xie XY et al (2009) Ethanol ablation of hepatocellular carcinoma up to 5.0 cm by using a multipronged injection needle with high-dose strategy. Radiology 253:552–561
Yin XY, Xie XY, Lu MD et al (2009) Percutaneous thermal ablation of medium and large hepatocellular carcinoma: long-term outcome and prognostic factors. Cancer 115:1914–1923
Chen MH, Yang W, Yan K et al (2004) Large liver tumors: protocol for radiofrequency ablation and its clinical application in 110 patients–mathematic model, overlapping mode, and electrode placement process. Radiology 232:260–271
Strasberg SM (2005) Nomenclature of hepatic anatomy and resections: a review of the Brisbane 2000 system. J Hepato-Biliary-Pancreat Surg 12:351–355
Peng ZW, Zhang YJ, Chen MS et al (2013) Radiofrequency ablation with or without transcatheter arterial chemoembolization in the treatment of hepatocellular carcinoma: a prospective randomized trial. J Clin Oncol 31:426–432
Couinaud C (1956) Contribution of anatomical research to liver surgery. Fr Med 19:5–12
Ahmed M, Solbiati L, Brace CL et al (2014) Image-guided tumor ablation: standardization of terminology and reporting criteria—a 10-year update. Radiology 273:241–260
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Omary RA, Bettmann MA, Cardella JF et al (2003) Quality improvement guidelines for the reporting and archiving of interventional radiology procedures. J Vasc Interv Radiol 14:S293–S295
Goldberg SN, Kruskal JB, Oliver BS, Clouse ME, Gazelle GS (2000) Percutaneous tumor ablation: increased coagulation by combining radio-frequency ablation and ethanol instillation in a rat breast tumor model. Radiology 217:827–831
Wong SN, Lin CJ, Lin CC, Chen WT, Cua IH, Lin SM (2008) Combined percutaneous radiofrequency ablation and ethanol injection for hepatocellular carcinoma in high-risk locations. AJR Am J Roentgenol 190:W187–W195
Mizukoshi E, Yamashita T, Arai K et al (2013) Enhancement of tumor-associated antigen-specific T cell responses by radiofrequency ablation of hepatocellular carcinoma. Hepatology 57:1448–1457
Zerbini A, Pilli M, Fagnoni F et al (2008) Increased immunostimulatory activity conferred to antigen-presenting cells by exposure to antigen extract from hepatocellular carcinoma after radiofrequency thermal ablation. J Immunother 31:271–282
Katz SC, Shia J, Liau KH et al (2009) Operative blood loss independently predicts recurrence and survival after resection of hepatocellular carcinoma. Ann Surg 249:617–623
Shiba H, Ishida Y, Wakiyama S et al (2009) Negative impact of blood transfusion on recurrence and prognosis of hepatocellular carcinoma after hepatic resection. J Gastrointest Surg 13:1636–1642
Nakazawa T, Kokubu S, Shibuya A et al (2007) Radiofrequency ablation of hepatocellular carcinoma: correlation between local tumor progression after ablation and ablative margin. AJR Am J Roentgenol 188:480–488
Poon RT, Fan ST, Tsang FH, Wong J (2002) Locoregional therapies for hepatocellular carcinoma: a critical review from the surgeon's perspective. Ann Surg 235:466–486
Shi M, Guo RP, Lin XJ et al (2007) Partial hepatectomy with wide versus narrow resection margin for solitary hepatocellular carcinoma: a prospective randomized trial. Ann Surg 245:36–43
Ye JZ, Miao ZG, Wu FX, Zhao YN, Ye HH, Li LQ (2012) Recurrence after anatomic resection versus nonanatomic resection for hepatocellular carcinoma: a meta analysis. Asian Pac J Cancer Prev 13:1771–1777
Feng X, Su Y, Zheng S et al (2017) A double blinded prospective randomized trial comparing the effect of anatomic versus non-anatomic resection on hepatocellular carcinoma recurrence. HPB (Oxford) 19:667–674
Kaibori M, Kon M, Kitawaki T et al (2017) Comparison of anatomic and non-anatomic hepatic resection for hepatocellular carcinoma. J Hepatobiliary Pancreat Sci 24:616–626
Morimoto M, Numata K, KondouM NA, Morita S, Tanaka K (2010) Midterm outcomes in patients with intermediate-sized hepatocellular carcinoma: a randomized controlled trial for determining the efficacy of radiofrequency ablation combined with transcatheterarterial chemoembolization. Cancer 116:5452–5460
Iezzi R, Pompili M, La Torre MF et al (2015) Radiofrequency ablation plus drug-eluting beads transcatheter arterial chemoembolization for the treatment of single large hepatocellualr carcinoma. Dig Liver Dis 47:242–248
Maluccio M, Covey AM, Gandhi R et al (2005) Comparison of survival rates after bland arterial embolization and ablation versus surgical resection for treating solitary hepatocellular carcinoma up to 7 cm. J Vasc Interv Radiol 16:955–961
Elnekave E, Erinjeri JP, Brown KT et al (2013) Long-term outcomes comparing surgery to embolization-ablation for treatment of solitary HCC<7 cm. Ann Surg Oncol 20:2881–2886
Xu Y, Shen Q, Liu P et al (2017) Microwave ablation for the treatment of hepatocellular carcinoma that met up-to-seven criteria: feasibility, local efficacy and long-term outcomes. Eur Radiol 27:3877–3887
Ma S, Ding M, Li J et al (2017) Ultrasound-guided percutaneous microwave ablation for hepatocellular carcinoma: clinical outcomes and prognostic factors. J Cancer Res Clin Oncol 143:131–142
Funding
This study has received funding by the National Natural Science Foundation of China (No. 81272312; No. 81301842), Pearl River S&T Nova Program of Guangzhou, China (No. 2014J2200087) and Guangdong Medical Science and Technology Foundation (No. 20161192364982).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Ming Kuang.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Statistics and biometry
One of the authors (Bin Li) has significant statistical expertise.
Informed consent
Written informed consent was obtained from all subjects (patients) in this study.
Ethical approval
Institutional review board approval was obtained.
Methodology
• retrospective
• observational
• multicentre study
Electronic supplementary material
ESM 1
(DOCX 92 kb)
Rights and permissions
About this article
Cite this article
Chen, S., Peng, Z., Lin, M. et al. Combined percutaneous radiofrequency ablation and ethanol injection versus hepatic resection for 2.1–5.0 cm solitary hepatocellular carcinoma: a retrospective comparative multicentre study. Eur Radiol 28, 3651–3660 (2018). https://doi.org/10.1007/s00330-018-5371-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-018-5371-9