Abstract
Objectives
To achieve multicentre external validation of the Herder and Bayesian Inference Malignancy Calculator (BIMC) models.
Methods
Two hundred and fifty-nine solitary pulmonary nodules (SPNs) collected from four major hospitals which underwent 18-FDG-PET characterization were included in this multicentre retrospective study. The Herder model was tested on all available lesions (group A). A subgroup of 180 SPNs (group B) was used to provide unbiased comparison between the Herder and BIMC models. Receiver operating characteristic (ROC) area under the curve (AUC) analysis was performed to assess diagnostic accuracy. Decision analysis was performed by adopting the risk threshold stated in British Thoracic Society (BTS) guidelines.
Results
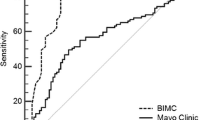
Unbiased comparison performed In Group B showed a ROC AUC for the Herder model of 0.807 (95 % CI 0.742–0.862) and for the BIMC model of 0.822 (95 % CI 0.758–0.875).
Conclusions
Both the Herder and the BIMC models were proven to accurately predict the risk of malignancy when tested on a large multicentre external case series. The BIMC model seems advantageous on the basis of a more favourable decision analysis.
Key Points
• The Herder model showed a ROC AUC of 0.807 on 180 SPNs.
• The BIMC model showed a ROC AUC of 0.822 on 180 SPNs.
• Decision analysis is more favourable to the BIMC model.


Similar content being viewed by others
References
Trotman-Dickenson B, Baumert B (2003) Multidetector-row CT of the solitary pulmonary nodule. In: Seminars in roentgenology, vol. 38, No. 2. WB Saunders, pp 158–167
Hansell DM, Bankier AA, MacMahon H, McLoud TC, Muller NL, Remy J (2008) Fleischner society: glossary of terms for thoracic imaging 1. Radiology 246:697–722
Swensen SJ, Silverstein MD, Edell ES, Trastek VF, Aughenbaugh GL, Ilstrup DM, Schleck CD (1999) Solitary pulmonary nodules: clinical prediction model versus physicians. In: Mayo Clinic Proceedings, vol. 74, No. 4. Elsevier, pp 319–329
Nakamura K, Yoshida H, Engelmann R et al (2000) Computerized analysis of the likelihood of malignancy in solitary pulmonary nodules with use of artificial neural networks 1. Radiology 214:823–830
Gurney JW (1993) Determining the likelihood of malignancy in solitary pulmonary nodules with Bayesian analysis. Part I. Theory. Radiology 186:405–413
Detterbeck FC, Mazzone PJ, Naidich DP, Bach PB (2013) Screening for lung cancer: diagnosis and management of lung cancer: American College of Chest Physicians evidence-based clinical practice guidelines. CHEST J 143:e78S–e92S
Callister MEJ, Baldwin DR, Akram AR et al (2015) British Thoracic Society guidelines for the investigation and management of pulmonary nodules. Thorax 70:ii1–ii54
McWilliams A, Tammemagi MC, Mayo JR, Roberts H, Liu G, Soghrati K, … Lam S (2013) Probability of cancer in pulmonary nodules detected on first screening CT. N Engl J Med 369(10):910–919
Herder GJ, van Tinteren H, Golding RP, Kostense PJ, Comans EF, Smit EF et al (2005) Clinical prediction model to characterize pulmonary nodules: validation and added value of 18F-fluorodeoxyglucose positron emission tomography. CHEST J 128:2490–2496
Perandini S, Soardi GA, Motton M, Dallaserra C, Montemezzi S (2014) Limited value of logistic regression analysis in solid solitary pulmonary nodules characterization: a single‐center experience on 288 consecutive cases. J Surg Oncol 110:883–887
Perandini S, Soardi GA, Motton M, Montemezzi S (2015) Critique of Al-Ameri et al.(2015)-Risk of malignancy in pulmonary nodules: a validation study of four prediction models. Lung cancer (Amsterdam, Neth) 90:118
Isbell JM, Deppen S, Putnam JB Jr et al (2011) Existing general population models inaccurately predict lung cancer risk in patients referred for surgical evaluation. Ann Thorac Surg 91:227–33
Soardi GA, Perandini S, Motton M, Montemezzi S (2015) Assessing probability of malignancy in solid solitary pulmonary nodules with a new Bayesian calculator: improving diagnostic accuracy by means of expanded and updated features. Eur Radiol 25:155–162
Al-Ameri A, Malhotra P, Thygesen H, Plant PK, Vaidyanathan S, Karthik S, Callister ME (2015) Risk of malignancy in pulmonary nodules: a validation study of four prediction models. Lung Cancer
Acknowledgments
The scientific guarantor of this publication is Simone Perandini. The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article. The authors state that this work has not received any funding. One of the authors has significant statistical expertise (S.P.). Institutional Review Board approval was not required because the research involved collection and analysis of existing data. Written informed consent was not required for this study because the research involved collection and analysis of existing data. Data and diagnostic specimens were recorded by the investigator in such a manner that subjects cannot be identified. Methodology: retrospective, diagnostic or prognostic study, multicentre study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Perandini, S., Soardi, G.A., Larici, A.R. et al. Multicenter external validation of two malignancy risk prediction models in patients undergoing 18F-FDG-PET for solitary pulmonary nodule evaluation. Eur Radiol 27, 2042–2046 (2017). https://doi.org/10.1007/s00330-016-4580-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-016-4580-3