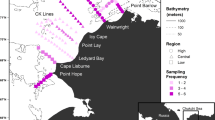
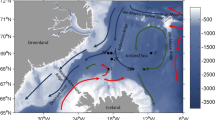
Abstract
Marine copepods of the genus Calanus have been extensively studied due to their central position in Arctic and Sub-Arctic food webs. Yet, our insight into how mortality in Calanus populations varies in time and space remains limited and restricts our ability to develop mechanistic understanding of the population dynamics of these key species. Based on 33 years of zooplankton data, collected in two Sub-Arctic fjords in Northern Norway, both at the onset of (October) and toward the end of winter (February), we estimated interannual variability in wintertime mortality rates for overwintering populations of Calanus spp. (C. finmarchicus and C. glacialis) and C. hyperboreus. Available hydrographical and meteorological information indicated that the influence of advection on the fjord basins were limited and was mainly associated with extreme weather conditions (strong winds or current). Estimated mortality of Calanus spp. was higher in the fjord where potential net-caught predators were more abundant (6.7 vs. 12.0 × 10−3 day−1), while mortality in the larger C. hyperboreus varied less between years and fjords (ca 4.5 × 10−3 day−1). This corroborates the view that predation is a key factor regulating loss from high-latitude Calanus populations in winter, but specific relationships between predators and Calanus mortality could not be established. These findings provide an outline of the relative potential of biological and physical mechanisms behind variability in Calanus mortality during overwintering. This approach highlights the need of integrative studies, combining advection and trophodynamics, in the study of fjord ecology.






Similar content being viewed by others
References
Aksnes DL, Magnesen T (1983) Distribution, development, and production of Calanus finmarchicus (gunnerus) in Lindåspollene, western Norway, 1979. Sarsia 68:195–207. https://doi.org/10.1080/00364827.1983.10420572
Aksnes DL, Ohman MD (1996) A vertical life table approach to zooplankton mortality estimation. Limnol Oceanogr 41:1461–1469. https://doi.org/10.4319/lo.1996.41.7.1461
Aksnes DL, Miller CB, Ohman MD, Wood SN (1997) Estimation techniques used in studies of copepod population dynamics—a review of underlying assumptions. Sarsia 82:279–296. https://doi.org/10.1080/00364827.1997.10413657
Arnkværn G, Daase M, Eiane K (2005) Dynamics of coexisting Calanus finmarchicus Calanus glacialis and Calanus hyperboreus populations in a high-Arctic fjord. Polar Biol 28:528–538. https://doi.org/10.1007/s00300-005-0715-8
Bagøien E, Kaartvedt S, Aksnes DL, Eiane K (2001) Vertical distribution and mortality of overwintering Calanus. Limnol Oceanogr 46:1494–1510
Browman HI, Yen J, Fields DM, St-Pierre J-F, Skiftesvik AB (2011) Fine-scale observations of the predatory behaviour of the carnivorous copepod Paraeuchaeta norvegica and the escape responses of their ichthyoplankton prey, Atlantic cod (Gadus morhua). Mar Biol 158:2653–2660. https://doi.org/10.1007/s00227-011-1763-y
Bucklin A, Kaartvedt S, Guarnieri M, Goswami U (2000) Population genetics of drifting (Calanus spp.) and resident (Acartia clausi) plankton in Norwegian fjords. J Plankton Res 22:1237–1251. https://doi.org/10.1093/plankt/22.7.1237
Carlotti F, Poggiale JC (2010) Towards methodological approaches to implement the zooplankton component in ‘‘end to end” food-web models. Prog Oceanogr 84:20–38
Choquet M et al (2017) Genetics redraws pelagic biogeography of Calanus. Biol Lett. https://doi.org/10.1098/rsbl.2017.0588
Conover R (1988) Comparative life histories in the genera Calanus and Neocalanus in high latitudes of the northern hemisphere. In: Biology of copepods. Springer, New York, pp 127–142
Daase M, Eiane K, Aksnes DL, Vogedes D (2008) Vertical distribution of Calanus spp. and Metridia longa at four Arctic locations Mar. Biol Res 4:193–207. https://doi.org/10.1080/17451000801907948
Daase M, Varpe Ø, Falk-Petersen S (2014) Non-consumptive mortality in copepods: occurrence of Calanus spp. carcasses in the Arctic Ocean during winter. J Plankton Res 36:129–144. https://doi.org/10.1093/plankt/fbt079
Eiane K, Aksnes DL, Ohman MD (1998) Advection and zooplankton fitness. Sarsia 83:87–93. https://doi.org/10.1080/00364827.1998.10413674
Eiane K, Aksnes DL, Ohman MD, Wood S, Martinussen MB (2002) Stage-specific mortality of Calanus spp. under different predation regimes. Limnol Oceanogr 47:636–645
Eliassen IK, Heggelund Y, Haakstad M (2001) A numerical study of the circulation in saltfjorden, saltstraumen and skjerstadfjorden. Cont Shelf Res 21:1669–1689. https://doi.org/10.1016/S0278-4343(01)00019-X
Espinasse B, Basedow SL, Tverberg V, Hattermann T, Eiane K (2016) A major Calanus finmarchicus overwintering population inside a deep fjord in northern Norway: implications for cod larvae recruitment success. J Plankton Res. https://doi.org/10.1093/plankt/fbw024
Falkenhaug T (1991) Prey composition and feeding rate of Sagitta elegans var. arctica (chaetognatha) in the Barents Sea in early summer. Polar Res 10:20. https://doi.org/10.3402/polar.v10i2.6761
Gentleman WC, Pepin P, Doucette S (2012) Estimating mortality: clarifying assumptions and sources of uncertainty in vertical methods. J Mar Syst 105–108:1–19. https://doi.org/10.1016/j.jmarsys.2012.05.006
Gislason A, Eiane K, Reynisson P (2006) Vertical distribution and mortality of Calanus finmarchicus during overwintering in oceanic waters southwest of Iceland. Mar Biol 150:1253–1263. https://doi.org/10.1007/s00227-006-0400-7
Greene CH, Landry MR (1985) Patterns of prey selection in the cruising calanoid predator euchaeta elongata. Ecology 66:1408–1416. https://doi.org/10.2307/1938003
Grodzins MA, Ruz PM, Keister JE (2016) Effects of oxygen depletion on field distributions and laboratory survival of the marine copepod Calanus pacificus. J Plankton Res 38:1412–1419. https://doi.org/10.1093/plankt/fbw063
Heath MR et al (2000) Comparative analysis of Calanus finmarchicus demography at locations around the Northeast Atlantic. ICES J Mar Sci 57:1562–1580. https://doi.org/10.1006/jmsc.2000.0950
Hirche HJ (1996) Diapause in the marine copepod Calanus finmarchicus—a review. Ophelia 44:129–143
Irigoien X, Harris RP, Head RN, Lindley JA, Harbour D (2000) Physiology and population structure of Calanus finmarchicus (Copepoda: Calanoida) during a Lagrangian tracer release experiment in the North Atlantic. J Plankton Res 22:205–221. https://doi.org/10.1093/plankt/22.2.205
Kara AB, Wallcraft AJ, Metzger EJ, Hurlburt HE, Fairall CW (2007) Wind stress drag coefficient over the global ocean. J Clim 20:5856–5864. https://doi.org/10.1175/2007JCLI1825.1
Kraft A, Berge J, Varpe Ø, Falk-Petersen S (2012) Feeding in Arctic darkness: mid-winter diet of the pelagic amphipods Themisto abyssorum and T. libellula. Mar Biol 160:241–248. https://doi.org/10.1007/s00227-012-2065-8
Kvile KØ, Stige LC, Prokopchuk I, Langangen Ø (2016) A statistical regression approach to estimate zooplankton mortality from spatiotemporal survey data. J Plankton Res 38:624–635. https://doi.org/10.1093/plankt/fbw028
Ohman MD (2012) Estimation of mortality for stage-structured zooplankton populations: what is to be done? J Mar Syst 93:4–10. https://doi.org/10.1016/j.jmarsys.2011.05.008
Østvedt OJ (1955) Zooplankton investigations from weathership M in the Norwegian Sea, 1948–49 Hvalråd Skrift 40:1–93
Pepin P (2013) Distribution and feeding of Benthosema glaciale in the western Labrador Sea: Fish–zooplankton interaction and the consequence to calanoid copepod populations. Deep Sea Res Pt I 75:119–134. https://doi.org/10.1016/j.dsr.2013.01.012
Pond S, Pickard GL (2013) Introductory dynamical oceanography, 2nd edn. Elsevier, pp 349
Rullyanto A, Jónasdóttir SH, Visser AW (2015) Advective loss of overwintering Calanus finmarchicus from the faroe-shetland channel. Deep Sea Res Pages 1(98):76–82. https://doi.org/10.1016/j.dsr.2014.12.009
Skagseth, Ø., K. F. Drinkwater, and E. Terrile (2011), Wind- and buoyancyinduced transport of the Norwegian Coastal Current in the Barents Sea. J Geophys Res 116:C08007. https://doi.org/10.1029/2011JC006996
Skardhamar J, Reigstad M, Carroll J, Eiane K, Wexels Riser C, Slagstad D (2011) Effects of mortality changes on biomass and production in Calanus spp. populations. Aquat Biol 12:129–145
Skreslet S (1994) Årsvariasjoner i fiskefaunaen på reketrålfelt i Salten. vol 1994
Skreslet S, Olsen K, Chelak M, Eiane K (2015) NE Atlantic zooplankton wintering in fjord habitats responds to hemispheric climate. J Plankton Res 37:773–789. https://doi.org/10.1093/plankt/fbv032
Smolina I et al (2014) Genome- and transcriptome-assisted development of nuclear insertion/deletion markers for Calanus species (Copepoda: Calanoida) identification. Mol Ecol Resour 14:1072–1079. https://doi.org/10.1111/1755-0998.12241
Thor P, Nielsen TG, Tiselius P (2008) Mortality rates of epipelagic copepods in the post-spring bloom period in Disko Bay, western Greenland. Mar Ecol Prog Ser 359:151–160
Visbeck M (2002) The ocean’s role in Atlantic climate variability. Science 297:2223–2224. https://doi.org/10.1126/science.1074029
Runge JA, Franks PJS, Gentleman WC, Megrey BA, Rose KA, Werner FE, Zakardjian B (2005) Diagnosis and prediction of variability in secondary production and fish recruitment processes: developments in physical-biological modeling. In: Robinson AR, Brink KH (eds) The global coastal ocean: multiscale interdisciplinary processes. Harvard University Press, Cambridge, pp 413–473
Yen J (1985) Selective predation by the carnivorous marine copepod Euchaeta elongata: laboratory measurements of predation rates verified by field observations of temporal and spatial feeding patterns1,2. Limnol Oceanogr 30:577–597. https://doi.org/10.4319/lo.1985.30.3.0577
Acknowledgements
We would like to thank all the crew members and technical assistants who helped in the logistic, sampling and processing the data. More specifically, we are grateful to Morten Krogstad, who has faithfully performed field-sampling routines, analyzed water samples and secured the data files and to Liv Johansen, Kjell Johansen, Robert Nicolaisen, Marit Reigstad, Odd Arne Schistad and the crews of R/V ‘Raud’ and R/V ‘Oscar Sund’. This study is a contribution of the ARCTOS LoVe MarinEco project, made possible through the support of Statoil.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Espinasse, B., Tverberg, V., Kristensen, J.A. et al. Winter mortality in Calanus populations in two northern Norwegian fjords from 1984 to 2016. Polar Biol 41, 1405–1415 (2018). https://doi.org/10.1007/s00300-018-2294-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-018-2294-5