Abstract
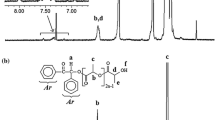
An A2B2-type miktoarm star copolymer with two linear poly(ε-caprolactone) (PCL) and two linear poly(cyclohexene oxide) (PCHO) arms was synthesized by using ring-opening polymerization (ROP), click chemistry, and photoinduced cationic polymerization, respectively. The ROP of ε-CL with a dihydroxy functional initiator, 3-cyclohexene-1,1-dimethanol, produced a well-defined two-arm PCL with a cyclohexene end-functional group, (PCL)2-CH. The subsequent bromination of (PCL)2-CH followed by azidation gave a diazido end-functionalized PCL, (PCL)2-(N3)2. Then, a well-defined two-arm PCL macrophotoinitiator with benzoin end-functional groups, (PCL)2-(PI)2, was prepared by the azide/alkyne click reaction of this (PCL)2-(N3)2 and PI-alkyne, a separately prepared benzoin photoinitiator bearing a terminal alkyne group. Solid data obtained from the 1H NMR, FT-IR, GPC, UV, and fluorescence spectroscopic studies proved the successful formation of the macrophotoinitiator having a core connected to two PCL chains and two benzoin photofunctional groups (PI). Finally, a novel A2B2-type miktoarm star copolymer was synthesized via photoinduced free radical-promoted cationic polymerization of cyclohexene oxide (CHO) monomer using (PCL)2-(PI)2 as photoinitiator. The obtained star copolymer consists of two PCL arms as A blocks and two PCHO arms as B blocks, (PCL)2-(PCHO)2, and was characterized by 1H NMR, FT-IR, and GPC measurements. The thermal behaviors of all the intermediate and final polymers were studied by thermogravimetric and differential thermal analysis (TG/DTA).













Similar content being viewed by others
References
Ren JM, McKenzie TG, Fu Q, Wong EHH, Xu J, An Z, Shanmugam S, Davis TP, Boyer C, Qiao GG (2016) Star polymers. Chem Rev 116:6743–6836
Hadjichristidis N (1999) Synthesis of miktoarm star (μ-star) polymers. J Polym Sci Part A Polym Chem 37:857–871
Kakkar A (2017) Miktoarm Star polymers: from basics of branched architecture to synthesis, self-assembly and applications. Royal Society of Chemistry, London
Shibuya Y, Nguyen HV-T, Johnson JA (2017) Mikto-brush-arm star polymers via cross-linking of dissimilar bottlebrushes: synthesis and solution morphologies. ACS Macro Lett 6:963–968
Kuckling D, Wycisk A (2013) Stimuli-responsive star polymers. J Polym Sci Part A Polym Chem 51:2980–2994
Mishra M, Kobayashi S (1999) Star and hyperbranched polymers. CRC Press, Boca Raton
Wu W, Wang W, Li J (2015) Star polymers: advances in biomedical applications. Prog Polym Sci 46:55–85
Khanna K, Varshney S, Kakkar A (2010) Miktoarm star polymers: advances in synthesis, self-assembly, and applications. Polym Chem 1:1171–1185
M-m Zhu, Song F, W-c Nie, X-l Wang, Y-z Wang (2016) A facile chemoenzymatic synthesis of amphiphilic miktoarm star copolymers from a sugar core and their potential for anticancer drug delivery. Polymer 93:159–166
Strandman S, Zarembo A, Darinskii AA, Laurinmäki P, Butcher SJ, Vuorimaa E, Lemmetyinen H, Tenhu H (2008) Effect of the number of arms on the association of amphiphilic star block copolymers. Macromolecules 41:8855–8864
Tunca U, Ozyurek Z, Erdogan T, Hizal G (2004) Novel miktofunctional initiator for the preparation of an ABC-type miktoarm star polymer via a combination of controlled polymerization techniques. J Polym Sci Part A Polym Chem 42:4228–4236
Yamazaki Y, Yokoyama A, Yokozawa T (2012) Three-arm star block copolymers of aromatic polyether and polystyrene from chain-growth condensation polymerization, atom transfer radical polymerization, and click reaction. J Polym Sci Part A Polym Chem 50:3648–3655
Gao H, Min K, Matyjaszewski K (2007) Synthesis of 3-arm star block copolymers by combination of “core-first” and “coupling-onto” methods using ATRP and click reactions. Macromol Chem Phys 208:1370–1378
Gao H, Matyjaszewski K (2006) Structural control in ATRP synthesis of star polymers using the arm-first method. Macromolecules 39:3154–3160
Altintas O, Yankul B, Hizal G, Tunca U (2006) A3-type star polymers via click chemistry. J Polym Sci Part A Polym Chem 44:6458–6465
Gao H, Matyjaszewski K (2008) Synthesis of low-polydispersity miktoarm star copolymers via a simple “arm-first” method: macromonomers as arm precursors. Macromolecules 41:4250–4257
Zhong L, Zhou Y, Yan D, Pan C (2008) Synthesis of a multi alternating-arm-containing dendritic star copolymer by RAFT and cationic ring-opening polymerization. Macromol Rapid Commun 29:1385–1391
Kolb HC, Finn M, Sharpless KB (2001) Click chemistry: diverse chemical function from a few good reactions. Angew Chem Int Ed 40:2004–2021
Rostovtsev VV, Green LG, Fokin VV, Sharpless KB (2002) A stepwise huisgen cycloaddition process: copper (I)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew Chem 114:2708–2711
Espeel P, Du Prez FE (2015) “Click”-inspired chemistry in macromolecular science: matching recent progress and user expectations. Macromolecules 48:2–14
Akeroyd N, Klumperman B (2011) The combination of living radical polymerization and click chemistry for the synthesis of advanced macromolecular architectures. Eur Polym J 47:1207–1231
Degirmenci M, Sarac MA, Genli N (2014) Synthesis and characterization of mid-chain macrophotoinitiator of poly (ε-caprolactone) by combination of ROP and click chemistry. Polym Bull 71:1743–1755
Degirmenci M, Besli PA, Genli N (2014) Synthesis of a well-defined end-chain macrophotoinitiator of poly (ε-caprolactone) by combination of ring-opening polymerization and click chemistry. J Polym Res 21:540
Degirmenci M, Genli N (2009) Synthesis of well-defined telechelic macrophotoinitiator of polystyrene by combination of ATRP and click chemistry. Macromol Chem Phys 210:1617–1623
Uyar Z, Degirmenci M, Genli N, Yilmaz A (2017) Synthesis of well-defined bisbenzoin end-functionalized poly (ε-caprolactone) macrophotoinitiator by combination of ROP and click chemistry and its use in the synthesis of star copolymers by photoinduced free radical promoted cationic polymerization. Des Monomers Polym 20:42–53
Satoh Y, Miyachi K, Matsuno H, Isono T, Tajima K, Kakuchi T, Satoh T (2016) Synthesis of well-defined amphiphilic star-block and miktoarm star copolyethers via t-Bu-P4-catalyzed ring-opening polymerization of glycidyl ethers. Macromolecules 49:499–509
Isono T, Otsuka I, Kondo Y, Halila S, Fort S, Rochas C, Satoh T, Borsali R, Kakuchi T (2013) Sub-10 nm nano-organization in AB2-and AB3-type miktoarm star copolymers consisting of maltoheptaose and polycaprolactone. Macromolecules 46:1461–1469
Moquin A, Sharma A, Cui Y, Lau A, Maysinger D, Kakkar A (2015) Asymmetric AB3 miktoarm star polymers: synthesis, self-assembly, and study of micelle stability using AF4 for efficient drug delivery. Macromol Biosci 15:1744–1754
C-g Mu, X-d Fan, Tian W, Bai Y, Zhou X (2012) Miktoarm star polymers with poly (N-isopropylacrylamide) or poly (oligo (ethylene glycol) methacrylate) as building blocks: synthesis and comparison of thermally-responsive behaviors. Polym Chem 3:1137–1149
Qiu J, Zhang J, Yu F, Wei J, Ding L (2016) Novel ABC miktoarm star terpolyphosphoesters: facile construction and high-flame retardant property. J Polym Sci Part A Polym Chem 54:692–701
Lorenzo AT, Müller AJ, Priftis D, Pitsikalis M, Hadjichristidis N (2007) Synthesis and morphological characterization of miktoarm star copolymers (PCL)2(PS)2 of poly (ε-caprolactone) and polystyrene. J Polym Sci Part A Polym Chem 45:5387–5397
Gungor E, Hizal G, Tunca U (2008) A2B2 type miktoarm star copolymers via alkyne homocoupling reaction. J Polym Sci Part A Polym Chem 46:6703–6711
Zhang W, Zhang W, Zhu J, Zhang Z, Zhu X (2009) Controlled synthesis of pH-responsive amphiphilic A2B2 miktoarm star block copolymer by combination of SET-LRP and RAFT polymerization. J Polym Sci Part A Polym Chem 47:6908–6918
Liu R, Li Z, Yuan D, Meng C, Wu Q, Zhu F (2011) Synthesis and self-assembly of miktoarm star copolymers of (polyethylene)2–(polystyrene)2. Polymer 52:356–362
Uyar Z, Durgun M, Yavuz MS, Abaci MB, Arslan U, Degirmenci M (2017) Two-arm PCL and PLLA macrophotoinitiators with benzoin end-functional groups by combination of ROP and click chemistry and their use in the synthesis of A2B2 type miktoarm star copolymers. Polymer 123:153–168
Yaḡci Y, Kornowski A, Schnabel W (1992) N-alkoxy-pyridinium and N-alkoxy-quinolinium salts as initiators for cationic photopolymerizations. J Polym Sci Part A Polym Chem 30:1987–1991
Save M, Schappacher M, Soum A (2002) Controlled ring-opening polymerization of lactones and lactides initiated by lanthanum isopropoxide, 1. General aspects and kinetics. Macromol Chem Phys 203:889–899
Khazaei A, Zolfigol MA, Kolvari E, Koukabi N, Soltani H, Bayani LS (2010) Electrophilic bromination of alkenes, alkynes, and aromatic amines with iodic acid/potassium bromide under mild conditions. Synth Commun 40:2954–2962
Yaḡci Y, Borbely J, Schnabel W (1989) On the mechanism of acylphosphine oxide promoted cationic polymerization. Eur Polym J 25:129–131
Acknowledgements
The authors would like to thank Harran University, Scientific Research Council (HÜBAK) and Dicle University, Scientific Research Projects Coordinator (Project No: 13-MYO-125) for financial support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Uyar, Z., Genli, N., Cay, B. et al. Synthesis and characterization of an A2B2-type miktoarm star copolymer based on poly(ε-caprolactone) and poly(cyclohexene oxide). Polym. Bull. 76, 553–573 (2019). https://doi.org/10.1007/s00289-018-2396-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00289-018-2396-9