Abstract
We consider a class of epidemiological models that includes most well-known dynamics for directly transmitted diseases, and some reduced models for indirectly transmitted diseases. We then propose a simple observer that can be applied to models in this class. The error analysis of this observer leads to a non-autonomous error equation, and a new bound for fundamental matrices is also presented. We analyse and implement this observer in two examples: the classical SIR model, and a reduced Bailey-Dietz model for vector-borne diseases. In both cases we obtain arbitrary exponential convergence of the observer. For the latter model, we also applied the observer to recover the number of susceptible using dengue infection data from a district in the city of Rio de Janeiro.









Similar content being viewed by others
References
Abdelhedi A, Boutat D, Sbita L, Tami R (2014) Extended observer to estimate the spreading of contagious disease. In 2014 European control conference (ECC), pp 1843–1848. https://doi.org/10.1109/ECC.2014.6862172
Abdelhedi A, Boutat D, Sbita L, Tami R, Liu D-Y (2016) Observer design for a class of nonlinear piecewise systems. Application to an epidemic model with treatment. Math Biosci 271:128–135
Alonso-Quesada S, De la Sen M, Agarwal RP, Ibeas A (2012) An observer-based vaccination control law for an SEIR epidemic model based on feedback linearization techniques for nonlinear systems. Adv Differ Equ 161:32
Alvarez-Ramirez J, Meraz M, Velasco-Hernandez JX (2000) Feedback control of the chemotherapy of HIV. Int J Bifurc Chaos 10(09):2207–2219. https://doi.org/10.1142/S0218127400001377
Anderson RM, May RM (1991) Infectious diseases of humans: dynamics and control. Oxford Science Publications, Oxford
Arino J, van den Driessche P (2003) A multi-city epidemic model. Math Popul Stud 10(3):175–193. https://doi.org/10.1080/08898480306720
Aronna MS, Bliman P-A (2018) Interval observer for uncertain time-varying SIR-SI model of vector-borne disease. In Proceedings of European control conference. URL https://hal.inria.fr/hal-01493078
Audoly S, D’Angio L, Saccomani MP, Cobelli C (1998) Global identifiability of linear compartmental models-a computer algebra algorithm. IEEE Trans Biomed Eng 45(1):36–47
Bailey N (1975) The mathematical theory of infectious diseases and its applications. Griffin, London
Becker NG (1989) Analysis of infectious disease data, vol 33. CRC Press, Boca Raton
Besançon G (Ed) (2007) Nonlinear observers and applications. In Papers based on the presentations at the 28th Grenoble international summer school on control, Grenoble, France, September 2007. Lecture notes in control and information sciences 363. Berlin: Springer. xiii, 224 p
Bichara D, Cozic N, Iggidr A (2014) On the estimation of sequestered infected erythrocytes in Plasmodium falciparum malaria patients. Math Biosci Eng 11(4):741–759. ISSN 1547-1063. https://doi.org/10.3934/mbe.2014.11.741
Bjørnstad ON, Finkenstädt BF, Grenfell BT (2002) Dynamics of measles epidemics: estimating scaling of transmission rates using a time series sir model. Ecol Monogr 72(2):169–184
Bliman P-A, D’Avila Barros B (2017) Interval observers for SIR epidemic models subject to uncertain seasonality. In: Positive systems, volume 471 of Lecture Notes Control Inf Sci, pp 31–39. Springer, Cham
Bliman P-A, Efimov D, Ushirobira R (2018) A class of nonlinear adaptive observers for sir epidemic model. In Proceedings of European control conference
Capasso V, Capasso V (1993) Mathematical structures of epidemic systems, vol 88. Springer, Berlin
Cauchemez S, Ferguson NM (2008) Likelihood-based estimation of continuous-time epidemic models from time-series data: application to measles transmission in london. J R Soc Interface 5(25):885–897
Cauchemez S, Carrat F, Viboud C, Valleron A, Boelle P (2004) A bayesian mcmc approach to study transmission of influenza: application to household longitudinal data. Stat Med 23(22):3469–3487
Coppel W (1978) Dichotomies in Stability Theory, volume 629 of Lecture Notes in Mathematics. Springer-Verlag, Berlin
De la Sen M, Ibeas A, Alonso-Quesada S (2011) Observer-based vaccination strategy for a true mass action SEIR epidemic model with potential estimation of all the populations. Discr Dyn Nat Soc, pages Art. ID 743067, 19
Diaby M, Iggidr A, Sy M (2015) Observer design for a schistosomiasis model. Mathematical biosciences, 269:17–29. ISSN 0025-5564. http://www.sciencedirect.com/science/article/pii/S0025556415001625
Diekmann O, Heesterbeek J (2000) Mathematical epidemiology of infectious diseases: model building, analysis and interpretation. Wiley series in mathematical and computational biology. Wiley, Chichester
Dietz K (1975) Transmission and control of arbovirus diseases. In: Ludwig E, Cooke KL (eds) Epidemiology. SIAM, New York, pp 104–121
Drakeley C, Corran P, Coleman P, Tongren J, McDonald S, Carneiro I, Malima R, Lusingu J, Manjurano A, Nkya W et al (2005) Estimating medium-and long-term trends in malaria transmission by using serological markers of malaria exposure. Proc Nat Acad Sci USA 102(14):5108–5113
Esteva L, Vargas C (1998) Analysis of a dengue disease transmission model. Math Biosci 150(2):131–151
Finkenstädt BF, Grenfell BT (2000) Time series modelling of childhood diseases: a dynamical systems approach. J Roy Stat Soc: Ser C (Appl Stat) 49(2):187–205
Fisman D, Khoo E, Tuite A (2014) Early epidemic dynamics of the west african 2014 ebola outbreak:estimates derived with a simple two-parameter model. PLOS Curr. https://doi.org/10.1371/currents.outbreaks.89c0d3783f36958d96ebbae97348d571
Gauthier J, Hammouri H, Othman S (1992) A simple observer for nonlinear systems applications to bioreactors. IEEE Trans Autom Control 37(6):875–880
Gérardin P, Guernier V, Perrau J, Fianu A, Le Roux K, Grivard P, Michault A, De Lamballerie X, Flahault A, Favier F (2008) Estimating chikungunya prevalence in la reunion island outbreak by serosurveys: two methods for two critical times of the epidemic. BMC Infect Dis 8(1):1
Guiro A, Iggidr A, Ngom D, Touré H (2009) On the stock estimation for some fishery systems. Rev Fish Biol Fish 19(3):313–327
Guo H, Li MY, Shuai Z (2006) Global stability of the endemic equilibrium of multigroup SIR epidemic models. Can Appl Math Q 14(3):259–284
Guo H, Li MY, Shuai Z (2008) A graph-theoretic approach to the method of global Lyapunov functions. In: Proceedings of the American Mathematical Society, 136(8):2793–2802, 08. https://doi.org/10.2307/20535481. http://www.jstor.org/stable/20535481
Hethcote HW (2000) The mathematics of infectious diseases. SIAM Rev 42(4):599–653
Ibeas A, de la Sen M, Alonso-Quesada S, Zamani I (2015) Stability analysis and observer design for discrete-time SEIR epidemic models. Adv Differ Equ 122:21
Iggidr A (2004) Encyclopedia of Life Support Systems (EOLSS), chapter Controllability, Observability and Stability of Mathematical Models. UNESCO, Eolss Publishers, Oxford
Iggidr A, Sallet G, Tsanou B (2012) Global stability analysis of a metapopulation SIS epidemic model. Math Popul Stud 19(3):115–129. https://doi.org/10.1080/08898480.2012.693844
Iggidr A, Sallet G, Souza MO (2016) On the dynamics of a class of multi-group models for vector-borne diseases. J Math Anal Appl 441(2):723–743. ISSN 0022-247X. https://doi.org/10.1016/j.jmaa.2016.04.003. http://www.sciencedirect.com/science/article/pii/S0022247X16300324
Inaba H (1990) Threshold and stability results for an age-structured epidemic model. J Math Biol 28(4):411–434
Jacquez JA, Greif P (1985) Numerical parameter identifiability and estimability: integrating identifiability, estimability, and optimal sampling design. Math Biosci 77(1):201–227
Jacquez JA, et al. (1985) Compartmental analysis in biology and medicine. JSTOR
Joseph L, Gyorkos TW, Coupal L (1995) Bayesian estimation of disease prevalence and the parameters of diagnostic tests in the absence of a gold standard. Am J Epidemiol 141(3):263–272
Kågström B (1977) Bounds and perturbation bounds for the matrix exponential. BIT Numer Math 17(1):39–57. https://doi.org/10.1007/BF01932398
Keeling MJ, Rohani P (2008) Modeling infectious diseases in humans and animals. Princeton University Press, Princeton
Kermack WO, McKendrick AG (1927) A contribution to the mathematical theory of epidemics. In Proceedings of the Royal Society of London a: mathematical, physical and engineering sciences, volume 115, pp 700–721
Kuniya T (2011) Global stability analysis with a discretization approach for an age-structured multigroup SIR epidemic model. Nonlinear Anal Real World Appl 12(5):2640–2655
Lajmanovich A, Yorke J (1976) A deterministic model for gonorrhea in a nonhomogeneous population. Math Biosci 28:221–236
Lloyd AL (2001) The dependence of viral parameter estimates on the assumed viral life cycle: limitations of studies of viral load data. Proc Roy Soc Lond B Biol Sci 268(1469):847–854
Luenberger DG (1971) An introduction to observers. IEEE Trans Autom Control AC 16(6):596–602. http://search.ebscohost.com/login.aspx?direct=true&db=inh&AN=334765&lang=fr&site=ehost-live
Nishiura H (2006) Mathematical and statistical analyses of the spread of dengue. Dengue Bull 30:51–67
Pongsumpun P, Tang I (2003) Transmission of Dengue hemorrhagic fever in an age structured population. Math Comput Model 37(9–10):949–961
Reiner RC, Perkins TA, Barker CM, Niu T, Chaves LF, Ellis AM, George DB, Le Menach A, Pulliam JRC, Bisanzio D, Buckee C, Chiyaka C, Cummings DAT, Garcia AJ, Gatton ML, Gething PW, Hartley DM, Johnston G, Klein EY, Michael E, Lindsay SW, Lloyd AL, Pigott DM, Reisen WK, Ruktanonchai N, Singh BK, Tatem AJ, Kitron U, Hay SI, Scott TW, Smith DL (2013) A systematic review of mathematical models of mosquito-borne pathogen transmission: 1970–2010. J Roy Soc Interface. https://doi.org/10.1098/rsif.2012.0921
Rocha F, Aguiar M, Souza MO, Stollenwerk N (2013) Time-scale separation and center manifold analysis describing vector-borne disease dynamics. Int J Comput Math 90(10):2105–2125
Ross R (1911) The prevention of malaria. John Murray, London
Smith DL, Battle KE, Hay SI, Barker CM, Scott TW, McKenzie FE (2012) Ross, macdonald, and a theory for the dynamics and control of mosquito-transmitted pathogens. PLoS Pathog. https://doi.org/10.1371%2Fjournal.ppat.1002588
Smith HL (1995) Monotone dynamical systems: an introduction to the theory of competitive and cooperative systems. Mathematical surveys and monographs. American Mathematical Society (AMS), Providence
Souza MO (2014) Multiscale analysis for a vector-borne epidemic model. J Math Biol 68(5):1269–1293
Tami R, Boutat D, Zheng G (2013) Extended output depending normal form. Automatica 49(7):2192–2198. https://doi.org/10.1016/j.automatica.2013.03.025. http://www.sciencedirect.com/science/article/pii/S0005109813001945
Tami R, Boutat D, Zheng G, Kratz F (2014) Parameters and states estimation for dengue epidemic model. In 2014 European control conference (ECC), pp 528–533. https://doi.org/10.1109/ECC.2014.6862173
Toni T, Welch D, Strelkowa N, Ipsen A, Stumpf MP (2009) Approximate bayesian computation scheme for parameter inference and model selection in dynamical systems. J R Soc Interface 6(31):187–202
Towers S, Patterson-Lomba O, Castillo-Chavez C (2014) Temporal variations in the effective reproduction number of the 2014 West Africa Ebola outbreak. PLOS Curr Outbreaks
Velasco-Hernández JX, García JA, Kirschner D (2002) Remarks on modeling host-viral dynamics and treatment. InCastillo-Chavez CEEA (Ed) Mathematical approaches for emerging and reemerging infectious diseases: an introduction. In Proceedings of a tutorial introduction to epidemiology and immunology, pp 287–308. Springer. IMA Vol Math Appl 125
Wonham W (1979) Linear multivariable control: a geometric approach, 2nd edn. Springer, New York
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was partially funded by Inria and CAPES-Brazil in the framework of the program STIC AmSud (Project MOSTICAW). M. O. Souza was partially supported by CNPq Grants # 308113/2012-8, # 486395/2013-8 and # 309079/2015-2.
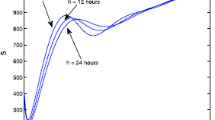
Appendix A: A glimpse at the data
Appendix A: A glimpse at the data
Rights and permissions
About this article
Cite this article
Iggidr, A., Souza, M.O. State estimators for some epidemiological systems. J. Math. Biol. 78, 225–256 (2019). https://doi.org/10.1007/s00285-018-1273-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00285-018-1273-3