Abstract
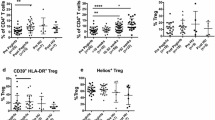
Polycythemia vera (PV) is a myeloproliferative neoplasm characterized by overproduction of red blood cells. We have performed a comprehensive characterization of blood immune cells for expression of naïve and memory receptors as well as β2m-associated and β2m-free MHC class I heavy chains, also known as closed and open conformers, respectively, in PV patients and age-matched controls (CTR). We show that the peripheral CD3+CD8+ T cell pool in PV patients is clearly divided into two discrete populations, a more granular CD3+CD8high T cell population enriched in effector-memory CD45RA+ T cells (CD8+ TEMRA) when compared to CTR (P < 0.001), and a less granular CD3+CD8int T cell population that is completely absent in the CTR group (78 vs. 0%, P < 0.001) and is a mixture of naïve (CD8+ TN) and CD8+ TEMRA cells expressing intermediate levels of CD28, i.e., CD3+CD8intCD28int. While the percentage of CD3+CD8int TN cells correlated positively with the number of erythrocytes, the percentage of CD3+CD8int TEMRA correlated negatively with the number of platelets. Finally, we report that PV patients’ lymphocytes and monocytes display lower levels of closed (W6/32+) MHC-I conformers at the cell surface while exhibiting increased amounts of open (HC-10+) MHC-I conformers. The implications of this distinctive immune signature are discussed.





Similar content being viewed by others
References
Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, Bloomfield CD, Cazzola M, Vardiman JW (2016) The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood 127(20):2391–2405
Cerquozzi S, Tefferi A (2015) Blast transformation and fibrotic progression in polycythemia vera and essential thrombocythemia: a literature review of incidence and risk factors. Blood Cancer J 5:e366
Vannucchi AM (2017) From leeches to personalized medicine: evolving concepts in the management of polycythemia vera. Haematologica 102(1):18–29
Patel AB, Vellore NA, Deininger MW (2016) New strategies in myeloproliferative neoplasms: the evolving genetic and therapeutic landscape. Clin Cancer Res 22(5):1037–1047
Kiladjian JJ, Cassinat B, Chevret S, Turlure P, Cambier N, Roussel M, Bellucci S, Grandchamp B, Chomienne C, Fenaux P (2008) Pegylated interferon-alfa-2a induces complete hematologic and molecular responses with low toxicity in polycythemia vera. Blood 112(8):3065–3072
Quintás-Cardama A, Abdel-Wahab O, Manshouri T, Kilpivaara O, Cortes J, Roupie AL, Zhang SJ, Harris D, Estrov Z, Kantarjian H, Levine RL, Verstovsek S (2013) Molecular analysis of patients with polycythemia vera or essential thrombocythemia receiving pegylated interferon α-2a. Blood 122(6):893–901
Eridani S, Fiorini GF, Batten E (1983) Peripheral blood lymphocyte subpopulations in polycythaemia and thrombocythaemia. Scand J Haematol 30(5):479–485
Wilde JT, Barnett D, Forrest MJ, Lawrence AC (1987) Peripheral blood lymphocyte subpopulations in patients with primary proliferative and secondary polycythaemia. J Clin Pathol 40(2):206–209
Rueda F, Remacha A, Martí F, Piñol G, Soler J, Guañabens C, Gimferrer E (1990) Different lymphocyte activity in patients with polycythaemia vera versus secondary polycythaemia and healthy blood donors. Acta Haematol 83(1):31–34
Zhao WB, Li Y, Liu X, Zhang LY, Wang X (2008) Involvement of CD4+CD25+ regulatory T cells in the pathogenesis of polycythaemia vera. Chin Med J(Engl) 121(18):1781–1786
Riley CH, Jensen MK, Brimnes MK, Hasselbalch HC, Bjerrum OW, Straten PT, Svane IM (2011) Increase in circulating CD4+CD25+Foxp3+ T cells in patients with Philadelphia-negative chronic myeloproliferative neoplasms during treatment with IFN-α. Blood 118(8):2170–2173
Costa-Brito AR, Cardoso EM, Martinez J, Arosa FA (2015) The JAK2V617F mutation is associated with anomalies in the frequency of monocytes, B cells, CD8+ T cells and NK cells. Blood Cells Mol Dis 55(3):208–210
Sanchez C, Le Treut T, Baier C, Sébahoun G, Costello RT (2015) Distribution of lymphocyte subpopulations in patients with polycythemia vera. Hum Immunol 76(6):414–416
Riley CH, Brimnes MK, Hansen M, Jensen MK, Hasselbalch HC, Kjaer L, Straten PT, Svane IM (2016) Interferon-α induces marked alterations in circulating regulatory T cells, NK cell subsets, and dendritic cells in patients with JAK2V617F-positive essential thrombocythemia and polycythemia vera. Eur J Haematol 97(1):83–92
Kovacsovics-Bankowski M, Kelley TW, Efimova O, Kim SJ, Wilson A, Swierczek S, Prchal J (2016) Changes in peripheral blood lymphocytes in polycythemia vera and essential thrombocythemia patients treated with pegylated-interferon alpha and correlation with JAK2V617F allelic burden. Exp Hematol Oncol 5:28
Skov V, Riley CH, Thomassen M, Larsen TS, Jensen MK, Bjerrum OW, Kruse TA, Hasselbalch HC (2013) Whole blood transcriptional profiling reveals significant down-regulation of human leukocyte antigen class I and II genes in essential thrombocythemia, polycythemia vera and myelofibrosis. Leuk Lymphoma 54(10):2269–2273
Skov V, Riley CH, Thomassen M, Kjær L, Stauffer Larsen T, Bjerrum OW, Kruse TA, Hasselbalch HC (2017) The impact of interferon-alpha2 on HLA genes in patients with polycythemia vera and related neoplasms. Leuk Lymphoma 58(8):1914–1921
Vannucchi AM, Kiladjian JJ, Griesshammer M, Masszi T, Durrant S, Passamonti F, Harrison CN, Pane F, Zachee P, Mesa R, He S, Jones MM, Garrett W, Li J, Pirron U, Habr D, Verstovsek S (2015) Ruxolitinib versus standard therapy for the treatment of polycythemia vera. N Engl J Med 372(5):426–435
Massa M, Rosti V, Campanelli R, Fois G, Barosi G (2014) Rapid and long-lasting decrease of T-regulatory cells in patients with myelofibrosis treated with ruxolitinib. Leukemia 28(2):449–451
Schönberg K, Rudolph J, Vonnahme M, Parampalli Yajnanarayana S, Cornez I, Hejazi M, Manser AR, Uhrberg M, Verbeek W, Koschmieder S, Brümmendorf TH, Brossart P, Heine A, Wolf D (2015) JAK inhibition impairs NK cell function in myeloproliferative neoplasms. Cancer Res 75(11):2187–2199
Arosa FA, Santos SG, Powis SJ (2007) Open conformers: the hidden face of MHC-I molecules. Trends Immunol 28(3):115–123
Correia MP, Cardoso EM, Pereira CF, Neves R, Uhrberg M, Arosa FA (2009) Hepatocytes and IL-15: a favorable microenvironment for T cell survival and CD8+ T cell differentiation. J Immunol 182(10):6149–6159
Santos SG, Powis SJ, Arosa FA (2004) Misfolding of major histocompatibility complex class I molecules in activated T cells allows cis-interactions with receptors and signaling molecules and is associated with tyrosine phosphorylation. J Biol Chem 279(51):53062–53070
Campbell JP, Guy K, Cosgrove C, Florida-James GD, Simpson RJ (2008) Total lymphocyte CD8 expression is not a reliable marker of cytotoxic T-cell populations in human peripheral blood following an acute bout of high-intensity exercise. Brain Behav Immun 22(3):375–380
Arosa FA, Esgalhado AJ, Padrão CA, Cardoso EM (2017) Divide, conquer, and sense: CD8+CD28- T cells in perspective. Front Immunol 7:665
Vaidya R, Gangat N, Jimma T, Finke CM, Lasho TL, Pardanani A, Tefferi A (2012) Plasma cytokines in polycythemia vera: phenotypic correlates, prognostic relevance, and comparison with myelofibrosis. Am J Hematol 87(11):1003–1005
Hasselbalch HC, Bjørn ME (2015) MPNs as inflammatory diseases: the evidence, consequences, and perspectives. Mediat Inflamm 2015:102476
Hasselbalch HC (2013) Chronic inflammation as a promotor of mutagenesis in essential thrombocythemia, polycythemia vera and myelofibrosis. A human inflammation model for cancer development? Leuk Res 37(2):214–220
Mumprecht S, Schürch C, Scherrer S, Claus C, Ochsenbein AF (2010) Chronic myelogenous leukemia maintains specific CD8+ T cells through IL-7 signaling. Eur J Immunol 40(10):2720–2730
Riether C, Schürch CM, Ochsenbein AF (2015) Regulation of hematopoietic and leukemic stem cells by the immune system. Cell Death Differ 22(2):187–198
Vuddamalay Y, Attia M, Vicente R, Pomié C, Enault G, Leobon B, Joffre O, Romagnoli P, van Meerwijk JP (2016) Mouse and human CD8(+)CD28(low) regulatory T lymphocytes differentiate in the thymus. Immunology 148(2):187–196
Ryan KR, Evavold BD (1998) Persistence of peptide-induced CD4+ T cell anergy in vitro. J Exp Med 187(1):89–96
Holmström MO, Hjortsø MD, Ahmad SM, Met Ö, Martinenaite E, Riley C, Straten P, Svane IM, Hasselbalch HC, Andersen MH (2017) The JAK2V617F mutation is a target for specific T cells in the JAK2V617F-positive myeloproliferative neoplasms. Leukemia 31(2):495–498
Nocka KH, Ottman OG, Pelus LM (1989) The role of marrow accessory cell populations in the augmentation of human erythroid progenitor cell (BFU-E) proliferation by prostaglandin E. Leuk Res 13(7):527–534
Li S, Wang L, Zhao C, Li L, Peng J, Hou M (2007) CD8+ T cells suppress autologous megakaryocyte apoptosis in idiopathic thrombocytopenic purpura. Br J Haematol 139(4):605–611
Safeukui I, Gomez ND, Adelani AA, Burte F, Afolabi NK, Akondy R, Velazquez P, Holder A, Tewari R, Buffet P, Brown BJ, Shokunbi WA, Olaleye D, Sodeinde O, Kazura J, Ahmed R, Mohandas N, Fernandez-Reyes D, Haldar K (2015) Malaria induces anemia through CD8+ T cell-dependent parasite clearance and erythrocyte removal in the spleen. MBio 6(1):e02493-e025014
Qiu J, Liu X, Li X, Zhang X, Han P, Zhou H, Shao L, Hou Y, Min Y, Kong Z, Wang Y, Wei Y, Liu X, Ni H, Peng J, Hou M (2016) CD8(+) T cells induce platelet clearance in the liver via platelet desialylation in immune thrombocytopenia. Sci Rep 6:27445
Cassel A, Braester A, Quitt M, Froom P, Aghai E (1993) Soluble factors from peripheral blood T-cells of patients with polycythemia vera stimulate normal BFU-E. Leukemia 7(9):1370–1373
Geissler K, Ohler L, Födinger M, Kabrna E, Kollars M, Skoupy S, Lechner K (1998) Interleukin-10 inhibits erythropoietin-independent growth of erythroid bursts in patients with polycythemia vera. Blood 92(6):1967–1972
Ishii T, Zhao Y, Shi J, Sozer S, Hoffman R, Xu M (2007) T cells from patients with polycythemia vera elaborate growth factors which contribute to endogenous erythroid and megakaryocyte colony formation. Leukemia 21(12):2433–2441
Hansen TH, Lybarger L, Yu L, Mitaksov V, Fremont DH (2005) Recognition of open conformers of classical MHC by chaperones and monoclonal antibodies. Immunol Rev 207:100–111
Parham P, Barnstable CJ, Bodmer WF (1979) Use of a monoclonal antibody (W6/32) in structural studies of HLA-A,B,C, antigens. J Immunol 123(1):342–349
Ladasky JJ, Shum BP, Canavez F, Seuánez HN, Parham P (1999) Residue 3 of beta2-microglobulin affects binding of class I MHC molecules by the W6/32 antibody. Immunogenetics 49(4):312–320
Stam NJ, Spits H, Ploegh HL (1986) Monoclonal antibodies raised against denatured HLA-B locus heavy chains permit biochemical characterization of certain HLA-C locus products. J Immunol 137(7):2299–2306
Perosa F, Luccarelli G, Prete M, Favoino E, Ferrone S, Beta DF (2003) 2-microglobulin-free HLA class I heavy chain epitope mimicry by monoclonal antibody HC-10-specific peptide. J Immunol 171(4):1918–1926
Raine T, Brown D, Bowness P, Hill Gaston JS, Moffett A, Trowsdale J, Allen RL (2006) Consistent patterns of expression of HLA class I free heavy chains in healthy individuals and raised expression in spondyloarthropathy patients point to physiological and pathological roles. Rheumatology (Oxford) 45(11):1338–1344
Apps R, Gardner L, Hiby SE, Sharkey AM, Moffett A (2008) Conformation of human leucocyte antigen-C molecules at the surface of human trophoblast cells. Immunology 124(3):322–328
Schnabl E, Stockinger H, Majdic O, Gaugitsch H, Lindley IJ, Maurer D, Hajek-Rosenmayr A, Knapp W (1990) Activated human T lymphocytes express MHC class I heavy chains not associated with beta 2-microglobulin. J Exp Med 171(5):1431–1442
Ramnath N, Tan D, Li Q, Hylander BL, Bogner P, Ryes L, Ferrone S (2006) Is downregulation of MHC class I antigen expression in human non-small cell lung cancer associated with prolonged survival? Cancer Immunol Immunother 55(8):891–899
Demaria S, Schwab R, Gottesman SR, Bushkin Y (1994) Soluble beta 2-microglobulin-free class I heavy chains are released from the surface of activated and leukemia cells by a metalloprotease. J Biol Chem 269(9):6689–6694
Tsai WC, Chen CJ, Yen JH, Ou TT, Tsai JJ, Liu CS, Liu HW (2002) Free HLA class I heavy chain-carrying monocytes-a potential role in the pathogenesis of spondyloarthropathies. J Rheumatol 29(5):966–972
Lan CC, Tsai WC, Wu CS, Yu CL, Yu HS (2004) Psoriatic patients with arthropathy show significant expression of free HLA class I heavy chains on circulating monocytes: a potential role in the pathogenesis of psoriatic arthropathy. Br J Dermatol 151(1):24–31
Khan MA (2010) HLA and spondyloarthropathies. In: Narinder K. Mehra. The HLA complex in biology and medicine. New Delhi, India: Jayppee brothers medical publishers, pp 259–275 ISBN 978-81-8448-870-8
Hudson LE, Allen RL (2016) Leukocyte Ig-like receptors - a model for MHC class I disease associations. Front Immunol 7:281
Wong-Baeza I, Ridley A, Shaw J, Hatano H, Rysnik O, McHugh K, Piper C, Brackenbridge S, Fernandes R, Chan A, Bowness P, Kollnberger S (2013) KIR3DL2 binds to HLA-B27 dimers and free H chains more strongly than other HLA class I and promotes the expansion of T cells in ankylosing spondylitis. J Immunol 190(7):3216–3224
Cauli A, Piga M, Dessole G, Porru G, Floris A, Vacca A, Desogus E, La Nasa G, Mathieu A (2014) Killer-cell immunoglobulin-like receptors (KIR) and HLA-class I heavy chains in ankylosing spondylitis. Drug Dev Res 75(Suppl 1):S15–S19
Assa-Kunik E, Fishman D, Kellman-Pressman S, Tsory S, Elhyany S, Baharir O, Segal S (2003) Alterations in the expression of MHC class I glycoproteins by B16BL6 melanoma cells modulate insulin receptor-regulated signal transduction and augment resistance to apoptosis. J Immunol 171(6):2945–2952
Chang CC, Ferrone S (2007) Immune selective pressure and HLA class I antigen defects in malignant lesions. Cancer Immunol Immunother 56(2):227–236
Smith C, Santi M, Rajan B, Rushing EJ, Choi MR, Rood BR, Cornelison R, MacDonald TJ, Vukmanovic S (2009) A novel role of HLA class I in the pathology of medulloblastoma. J Transl Med 7:59
Smith C, Santi M, Rushing EJ, Cornelison R, MacDonald TJ, Vukmanovic S (2011) Characterization of signaling function and expression of HLA class I molecules in medulloblastoma. J Neuro-Oncol 103(2):197–206
Fishman D, Elhyany S, Segal S (2004) Non-immune functions of MHC class I glycoproteins in normal and malignant cells. Folia Biol (Praha) 50(2):35–42
Bowness P, Caplan S, Edidin M (2009) MHC molecules lead many lives. Workshop on MHC class I molecules at the interface between Biology & Medicine. EMBO Rep 10(1):30–34
Plaisier CL, O'Brien S, Bernard B, Reynolds S, Simon Z, Toledo CM, Ding Y, Reiss DJ, Paddison PJ, Baliga NS (2016) Causal mechanistic regulatory network for glioblastoma deciphered using systems genetics network analysis. Cell Syst 3(2):172–186
Acknowledgements
The authors wish to thank Dr. Isabel Lobo and collaborators from Centro do Sangue e Transplantação de Coimbra (CST-C) for providing buffy-coats of regular blood donors for this study. We also thank Dr. Simon Powis (Biomedical Sciences Research Complex, University of St Andrews, St. Andrews, Scotland) for kindly providing the HC-10 monoclonal antibody. EMC, AJE, and FAA were supported by FEDER funds through the POCI–COMPETE 2020—Program Competitiveness and Internationalization in Axis I—strengthening research, technological development, and innovation (Project No. 007491), through project CENTRO-01-0145-FEDER-000013, and National Funds by FCT—Foundation for Science and Technology (Project UID/Multi/00709).
Author information
Authors and Affiliations
Contributions
EMC and FAA designed the experiments, analyzed data, and wrote the manuscript; EMC and AJE performed the experiments and analyzed data; LP collaborated in PV patient sample collection and clinical data analysis; MS collected clinical data; VPN collected blood samples from controls; JM contributed with blood samples from PV and SP patients and controls; MAVP recruited age-matched controls; HS recruited PV patients and collected clinical data. All authors read and agreed with the submission of the manuscript.
Corresponding author
Ethics declarations
The Ethic Committees of the three Hospitals approved the study and each subject gave written informed consent prior to enrolment in the study according to the Declaration of Helsinki. All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Cardoso, E.M., Esgalhado, A.J., Patrão, L. et al. Distinctive CD8+ T cell and MHC class I signatures in polycythemia vera patients. Ann Hematol 97, 1563–1575 (2018). https://doi.org/10.1007/s00277-018-3332-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-018-3332-7