Abstract
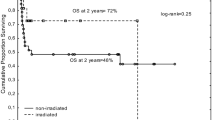
In the context of discussions on the reproducibility of clinical studies, we reanalyzed a prospective randomized study on the role of splenic irradiation as adjunct to the conditioning for hematopoietic stem cell transplantation (HSCT) for chronic myeloid leukemia (CML). Between 1986 and 1989, a total of 229 patients with CML were randomized; of these, 225 (98 %; 112 with, 113 without splenic irradiation) could be identified in the database and their survival updated. Results confirmed the early findings with no significant differences in all measured endpoints (overall survival at 25 years: 42.7 %, 32.0–52.4 % vs 52.9 %, 43.2–62.6 %; p = 0.355, log rank test). Additional splenic irradiation failed to reduce relapse incidence. It did not increase non-relapse mortality nor the risk of late secondary malignancies. Comforting are the long-term results from this predefined consecutive cohort of patients: more than 60 % were alive at plus 25 years when they were transplanted with a low European Society for Blood and Marrow Transplantation (EBMT) risk sore. This needs to be considered today when treatment options are discussed for patients who failed initial tyrosine kinase inhibitor therapy and have an available low risk HLA-identical donor.

Similar content being viewed by others
References
Goldman JM, Baughan AS, McCarthy DM et al (1982) Marrow transplantation for patients in the chronic phase of chronic granulocytic leukaemia. Lancet 2(8299):623–625
McGlave PB, Arthur DC, Kim TH, Ramsay NK, Hurd DD, Kersey J (1982) Successful allogeneic bone-marrow transplantation for patients in the accelerated phase of chronic granulocytic leukaemia. Lancet 2(8299):625–627
Gratwohl A, Goldman J, Gluckman E, Zwaan F (1985) Effect of splenectomy before bone-marrow transplantation on survival in chronic granulocytic leukaemia. Lancet 2(8467):1290–1291
Gratwohl A, Hermans J, von Biezen A et al (1992) No advantage for patients who receive splenic irradiation before bone marrow transplantation for chronic myeloid leukaemia: results of a prospective randomized study. Bone Marrow Transplant 10:147–152
Gratwohl A, Hermans J, van Biezen A et al (1996) Splenic irradiation before bone marrow transplantation for chronic myeloid leukaemia. Chronic Leukaemia Working Party of the European Group for Blood and Marrow Transplantation (EBMT). Br J Haematol 95:494–500
O’Brien SG, Guilhot F, Larson RA et al (2003) IRIS Investigators. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med 348:994–1004
Baccarani M, Deininger MW, Rosti G et al (2013) European LeukemiaNet recommendations for the management of chronic myeloid leukemia. Blood 122:872–884
Ito T, Akagi K, Kondo T, Kawabata H, Ichinohe T, Takaori-Kondo A (2012) Splenic irradiation as a component of a reduced-intensity conditioning regimen for hematopoietic stem cell transplantation in myelofibrosis with massive splenomegaly. Tohoku J Exp Med 228:295–299
Gratwohl A, Pfirrmann M, Zander A et al (2015) Long-term outcome of patients with newly diagnosed chronic myeloid leukemia: a randomized comparison of stem cell transplantation with drug treatment. Leukemia. doi:10.1038/leu.2015.281
Barrett AJ, Ito S (2015) The role of stem cell transplantation for chronic myelogenous leukemia in the 21st century. Blood 125(21):3230–3235
Gratwohl A, Baldomero H, Aljurf M et al (2010) Hematopoietic stem cell transplantation: a global perspective. JAMA 303:1617–1624
Pasquini MC (2012) Hematopoietic cell transplantation for chronic myeloid leukemia in developing countries: perspectives from Latin America in the post-tyrosine kinase inhibitor era. Hematology 17(Suppl 1):S79–S82
Ebrahim S, Sohani ZN, Montoya L et al (2014) Reanalyses of randomized clinical trial data. JAMA 312:1024–1032
Gratwohl A (2012) The EBMT risk score. Bone Marrow Transplant 47:749–756
Barbui T, Björkholm M, Gratwohl A (2012) Optimizing investigator-led oncology research in Europe. Haematologica 97(6):800–804
Taylor J, Patrick H, Lyratzopoulos G et al (2014) Methodological challenges in evaluating the value of registers. Int J Technol Assess Health Care 30:28–33
Acknowledgments
The authors would like to thank all participating patients and transplant teams.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflicts of interest
There are no conflicts of interest to declare.
Funding
The study was funded by the Chronic Malignancies Working Party of the European Society for Blood and Marrow Transplantation EBMT. EBMT is supported by grants from the corporate members: Amgen Europe, ViroPharma Europe, Celegene International SARL, Genzyme Europe B.V., Gilead Sciences Europe Ltd., MiltenylBiotec GmbH, Schering-Plough International Inc., Bristol Myers Squibb, CaridiaBCT Europe NV, Cephalon Europe, F. Hoffmann-La Roche Ltd, Fresenius Biotech GmbH, Therakos Inc., Alexion Europe, Chugai Sanofi – Aventis, Merck Sharp and Dohme, Novartis, Pfizer, Pierre Fabre Médicament.
Appendix
Appendix
List of participating centers with past (ref Gratwohl et al. 1996) and current principal investigators.
Basel: Kantonsspital (today University Hospital; B. Speck, A. Gratwohl, J. Passweg); Berlin: University Hospital Rudolf Virchow (W. Siegert, R. Arnold); Birmingham: Queen Elizabeth Hospital (I.M. Franklin, C. Craddock); Brussels: Insitut J. Bordet (L. Debusscher, D. Bron), Hôpital Universitaire St. Luc (A. Ferrant, X. Poiré); Caen: Centre Hospitalier (X.Troussard, O. Reman); Copenhagen: University Hospital (N. Jacobsen, H. Sengeloev); Córdoba: Hospital Reina Sofia (A. Torrez-Gomez, P. Gomez Garcia); Créteil: HôpitalMondor (J.-P. Vernant, C. Cordonnier); Dublin: St. James’ Hospital (S.R. McCann, P. Browne); Edinburgh: Royal Infirmary (A.C. Parker, A.J.M. Broom); Genova: Ospedale San Martino (A. Bacigalupo, F. Frassoni, A. Bacigalupo); Hannover: Medizinische Hochschule (H. Link, A. Ganser); Helsinki: University of Helsinki Hospital (T. Ruutu, L. Volin); Huddinge: Karolinska Institute (B. Lönnqvist, G. Gahrton, O. Ringden, P. Ljungman); Innsbruck: University Hospital (D. Niederwieser, G. Gastl); Kiel: Christian Albrechts University Hospital (N. Schmitz, M. Gramatzki); Leiden: University Hospital (F. Zwaan, W. Fibbe, J.H. Veelken); Lisbon: Ins. Portuges Oncologia (M. Abecasis, M. Abecasis); London: Harley Street clinic (P.J. Gravett), Royal Marsden Hospital (R. Powles, G. Helenglass, M. Potter), Royal Free Hospital (H.G. Prentice, S. McKinnon); Munich: Ludwig Maximilian University Hospital (H.J. Kolb, W.Hiddemann, J. Tischer); Nijmegen: Sint Radboud Ziekenhuis (T. deWitte, T. Schattenberg, N. Schaap); Paris: Centre Hospitalier St. Antoine (N.C. Gorin, L.Fouillard, M. Mohty); Pessac: Centre Hospitalier (J. Reiffers, N. Millpied); Riyadh: King Faisal Hospital (P. Ernst, M. Aljurf); Rome: UniversitadegliStudi di Roma La Sapienza (W. Arcese, P. de Fabritiis, R. Foa); Santander: Hospital Nacional “Marques de Valdecilla” (A. Iriondo, C. Richard Espiga); Vienna: Allgemeines Krankenhaus der Stadt Wien (W. Hinterberger, P. Kahls)
Rights and permissions
About this article
Cite this article
Gratwohl, A., Iacobelli, S., Bootsman, N. et al. Splenic irradiation before hematopoietic stem cell transplantation for chronic myeloid leukemia: long-term follow-up of a prospective randomized study. Ann Hematol 95, 967–972 (2016). https://doi.org/10.1007/s00277-016-2638-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-016-2638-6