Abstract
Purpose
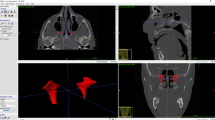
The pterygopalatine ganglion has yet not been identified on medical images in living humans. The primary aim of this study was to evaluate whether the pterygopalatine ganglion could be identified on 3 T MR imaging.
Methods
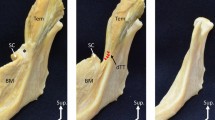
This study was performed on medical images of 20 Caucasian subjects on both sides (n = 40 ganglia) with an exploratory design. 3 T MR images were assessed by two physicians for the presence and size of the pterygopalatine ganglion. The distance from the pterygopalatine ganglion to four bony landmarks was registered from fused MR and CT images. In an equivalence analysis, the distances were compared to those obtained in an anatomical cadaveric study serving as historical controls (n = 50).
Results
A structure assumed to be the pterygopalatine ganglion was identified on MR images in all patients on both sides by both physicians. The mean size was depth 2.1 ± 0.5 mm, width 4.2 ± 1.1 mm and height 5.1 ± 1.4 mm, which is in accordance with formerly published data. Equivalence of the measurements on MR images and the historical controls was established, suggesting that the structure identified on the MR images is the pterygopalatine ganglion.
Conclusion
Our findings suggest that the pterygopalatine ganglion can be detected on 3 T MR images. Identification of the pterygopalatine ganglion may be important for image-guided interventions targeting the pterygopalatine ganglion, and has the potential to increase the efficacy, safety and reliability for these treatments.



Similar content being viewed by others
References
Ahn S, Park SH, Lee KH (2013) How to demonstrate similarity by using noninferiority and equivalence statistical testing in radiology research. Radiology 267:328–338. https://doi.org/10.1148/radiol.12120725
Alvernia JE, Spomar DG, Olivero WC (2007) A computed tomography scan and anatomical cadaveric study of the pterygopalatine ganglion for use in Gamma Knife treatment of cluster headache. J Neurosurg 107:805–808. https://doi.org/10.3171/jns-07/10/0805
Bratbak DF, Nordgard S, Stovner LJ, Linde M, Dodick DW, Aschehoug I, Folvik M, Tronvik E (2016) Pilot study of sphenopalatine injection of onabotulinumtoxinA for the treatment of intractable chronic migraine. Cephalalgia. https://doi.org/10.1177/0333102416648328
Bratbak DF, Nordgard S, Stovner LJ, Linde M, Folvik M, Bugten V, Tronvik E (2016) Pilot study of sphenopalatine injection of onabotulinumtoxinA for the treatment of intractable chronic cluster headache. Cephalalgia 36:503–509. https://doi.org/10.1177/0333102415597891
Cassano M, Marioni G, Russo L, Cassano P (2012) Sphenopalatine artery ligation with nerve resection in patients with vasomotor rhinitis and polyposis: a prospective, randomized, double-blind investigation. Acta Otolaryngol 132:525–532. https://doi.org/10.3109/00016489.2011.648272
Chang PC, Fischbein NJ, McCalmont TH, Kashani-Sabet M, Zettersten EM, Liu AY, Weissman JL (2004) Perineural spread of malignant melanoma of the head and neck: clinical and imaging features. AJNR Am J Neuroradiol 25:5–11
Kastler A, Cadel G, Comte A, Gory G, Piccand V, Tavernier L, Kastler B (2014) Alcohol percutaneous neurolysis of the sphenopalatine ganglion in the management of refractory cranio-facial pain. Neuroradiology 56:589–596. https://doi.org/10.1007/s00234-014-1354-y
Keller H (1980) Über Die Hintere Pfortenregion Der Fossa Pterygopalatina Und Die Lage Des Ganglion Pterygopalatinum. Doctoral Dissertation, Julius-Maximilans-Universitäts Würburg
Lang J, Keller H (1978) The posterior opening of the pterygopalatine fossa and the position of the pterygopalatine ganglion. Gegenbaurs Morphol Jahrb 124:207–214
Lovasova K, Sulla IJ, Bolekova A, Sulla I, Kluchova D (2013) Anatomical study of the roots of cranial parasympathetic ganglia: a contribution to medical education. Ann Anat 195:205–211. https://doi.org/10.1016/j.aanat.2013.01.011
Oomen KP, Pameijer FA, Zwanenburg JJ, Hordijk GJ, De Ru JA, Bleys RL (2012) Improved depiction of pterygopalatine fossa anatomy using ultrahigh-resolution magnetic resonance imaging at 7 T. Sci World J 2012:7. https://doi.org/10.1100/2012/691095
Penteshina NA (1965) Morphology of the Pterygopalatine Ganglion. Zh Nevropat Psikhiat 65:1325–1330
Piagkou M, Demesticha T, Troupis T, Vlasis K, Skandalakis P, Makri A, Mazarakis A, Lappas D, Piagkos G, Johnson EO (2012) The pterygopalatine ganglion and its role in various pain syndromes: from anatomy to clinical practice. Pain Pract 12:399–412. https://doi.org/10.1111/j.1533-2500.2011.00507.x
Pipolo C, Bussone G, Leone M, Lozza P, Felisati G (2010) Sphenopalatine endoscopic ganglion block in cluster headache: a reevaluation of the procedure after 5 years. Neurol Sci 31(Suppl 1):S197–S199. https://doi.org/10.1007/s10072-010-0325-2
Rusu MC, Pop F, Curca GC, Podoleanu L, Voinea LM (2009) The pterygopalatine ganglion in humans: a morphological study. Ann Anat 191:196–202. https://doi.org/10.1016/j.aanat.2008.09.008
Vogel K (1930) Die Beziehungen des Ganglion Sphenopalatinum zu den von ihm ausgehenden Nervenstämmen. Z Hals- Nasen-Ohrenheilkd 25:485–502
Acknowledgements
The authors would like to acknowledge the contribution of research nurse Irina Aschehoug for her work during this trial and the kind assistance of Prof. Dr. Stefanie Kürten and Michael Christof at Institute of Anatomy and Cell Biology, University of Würzburg, Germany. Most importantly, we acknowledge the excellent work by Prof. Dr. Johannes Lang and Prof. Dr. Helmut Keller enabling us to perform this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by the Liaison Committee between the Central Norway Regional Health Authority and Norwegian University of Science and Technology (Grant number 12/9996); Joint Research Unit between St. Olavs Hospital and Norwegian University of Science and Technology (Grant number 9885); NTNU Discovery (Grant number 244278.
Conflict of interest
The results of this study may affect opinions on the feasibility of interventional treatments targeting the PPG. An intervention device for image-guided injections of pharmacological substances towards the PPG is developed at NTNU and St. Olavs Hospital, Trondheim University Hospital. These institutions may benefit financially of a commercialization of the device through future possible intellectual properties, this may include financial benefits to authors of this article. Dr. Bratbak is co-inventor of a proposed treatment targeting the PPG and the intervention device used to perform the treatment, both inventions patent pending, and may benefit financially of a commercialization of the proposed treatment through future possible intellectual properties. Dr. Folvik has nothing to disclose. Dr. Nordgård is co-inventor of a proposed treatment targeting the PPG and the intervention device used to perform the treatment, both inventions patent pending, and may benefit financially of a commercialization of the proposed treatment through future possible intellectual properties. Dr. Stovner has no relevant conflicts of interest. Dr. Dodick has consulted for Allergan, Amgen, Alder, CoLucid, Merck, eNeura, Eli Lilly & Company, Autonomic Technologies, Teva, Tonix, GBS, Dr. Reddy’s, and Xenon within the past 12 months. Dr. Matharu reports grants from Allergan, grants and part-sponsored educational events from electroCore, grants from Medtronic, grants from St Jude Medical, all outside the submitted work. Dr. Tronvik may benefit financially of a commercialization of a proposed treatment targeting the PPG and the intervention device used to perform the treatment through future possible intellectual properties.
Rights and permissions
About this article
Cite this article
Bratbak, D.F., Folvik, M., Nordgård, S. et al. Depicting the pterygopalatine ganglion on 3 Tesla magnetic resonance images. Surg Radiol Anat 40, 689–695 (2018). https://doi.org/10.1007/s00276-017-1960-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-017-1960-6