Abstract
Background
The risk of venous thromboembolism (VTE) persists beyond hospitalization in surgical patients, yet post-hospital discharge chemoprophylaxis regimens are not common. The purpose of this study is to systematically review the literature regarding extended-duration (post-hospital discharge) venous thromboembolism chemoprophylaxis and to determine whether it is warranted in high-risk surgical patients, as determined by its safety and efficacy.
Method
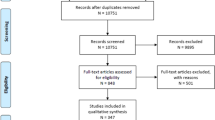
We searched four online databases for articles evaluating extended-duration (post-hospital discharge) VTE chemoprophylaxis regimens in surgical patients between the years January 2000 and February 2020. The Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) guidelines were used. GRADE methodology and the Cochrane Risk of Bias Assessment Tool for Randomized Controlled Trials were used to grade the quality of evidence and assess risk of bias.
Results
Nineteen studies with 10,544 patients were analyzed. The duration for extended-duration VTE chemoprophylaxis ranged from 7 to 42 days. In our study cohort, high-risk patients not prescribed extended-duration VTE chemoprophylaxis had a mean VTE incidence rate of 12.23%, while patients receiving 28–30 days of chemoprophylaxis had a mean VTE incidence rate of 4.37% (p = 0.006). The risk of bleeding events did not correlate with the duration of chemoprophylaxis.
Conclusion
Extended-duration VTE chemoprophylaxis in high-risk surgical patients decreased the incidence of thrombotic complications without increasing the risk of bleeding events. Further research is needed to establish guidelines for the optimal duration of VTE chemoprophylaxis in high-risk surgical patients.
Level of evidence
III.

Similar content being viewed by others
Change history
01 July 2020
In the original article, the authors’ affiliations and affiliation addresses are inaccurate. They are correct as reflected here.
References
Alshouimi RA et al (2019) The use of direct oral anticoagulants for extended duration thromboprophylaxis in medically ill patients: a systematic review and meta-analysis. J Thromb Thrombolysis 48:422–429
Agnelli G, Bolis G, Capussotti L, Scarpa RM, Tonelli F, Boniz-zoni E, Moia M, Parazzini F, Rossi R, Sonaglia F, Valarani B, Bianchini C, Gussoni G (2006) A clinical outcome-based prospective study on venous thromboembolism after cancer surgery: the @RISTOS project. Ann Surg 243(1):89–95
Faye AS et al (2019) Acute venous thromboembolism risk highest within 60 days after discharge from the hospital in patients with inflammatory bowel diseases. Clin Gastroenterol Hepatol. https://doi.org/10.1016/j.cgh.2019.07.028
Fagarasanu A et al (2016) Role of extended thromboprophylaxis after abdominal and pelvic surgery in cancer patients: A systematic review and meta-analysis. Ann Surg Oncol 23:1422–1430
Hull RD et al (2013) Venous thromboembolism in elderly high-risk medical patients: Time course of events and influence of risk factors. Clin Appl Thromb Hemost 19(4):357–362
Wright JD, Chen L, Jorge S et al (2016) Prescription of extended-duration thromboprophylaxis after high-risk, abdominopelvic cancer surgery. Gynecol Oncol 141(3):531–537
Raftopoulos I, Martindale C, Cronin A, Steinberg J (2008) The effect of extended post-discharge chemical thromboprophylaxis on venous thromboembolism rates after bariatric surgery: a prospective comparison trial. Surg Endosc 22(11):2384–2391
Eriksson BI, Lassen MR (2003) Duration of prophylaxis against venous thromboembolism with fondaparinux after hip fracture surgery. Arch Intern Med 163:1337–1342
Gilmartin CE, Cutts BA (2019) Ismail H. A cross-sectional study. Aust N Z J Obstet Gynaecol, Venous thromboembolism prophylaxis of the obese postpartum patient, pp 1–6
Ruff SM (2019) Practice patterns of VTE chemoprophylaxis after discharge following hepatic and pancreatic resections for cancer: A survey of hepatopancreatobiliary surgeons. J Thromb Thrombolysis 48(1):119–124
Lemke M et al (2016) Patient adherence and experience with extended use of prophylactic low-molecular-weight heparin following pancreas and liver resection. J Gastrointest Surg 20:1986–1996
Gould MK, Garcia DA, Wren SM et al (2012) Prevention of VTE in nonorthopedic surgical patients: Antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 141(2 Suppl):e227S–e277S
Falck-Ytter Y, Francis CW, Johanson NA, Curley C, Dahl OE, Schulman S et al (2012) Prevention of VTE in orthopedic surgery patients: Antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 141(2 Suppl):e278S–e325S
Zayed Y et al (2020) Extended duration of thromboprophylaxis for medically ill patients: a systematic review and meta-analysis of randomized controlled trials. Intern Med J 50(2):192–199
Muñoz-Atienza V et al (2018) Extended use of bemiparin as thromboprophylaxis during bariatric surgery: results of anti-factor Xa activity measurements. Surgery for Obesity and Related Diseases 14:354–360
Caprini JA (2011) Identification of patient venous thromboembolism risk across the continuum of care. Clin Appl Thromb Hemost 17:590–599
Kerwin AJ, Haut ER, Burns JB, Como JJ, Haider A, Stassen N, Dahm P (2012) The Eastern Association of the Surgery of Trauma approach to practice management guideline development using Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) methodology. J Trauma Acute Care Surg 73:S283–S287
Guyatt G, Oxman AD, Akl EA et al (2011) GRADE guidelines: 1. Introduction and GRADE evidence profiles and summary of findings tables. J Clin Epidemiol 64:383–394
Higgins JPT, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JAC (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomized trials. BMJ 343:d5928
Vedovati MC, Becattini C, Rondelli F et al (2014) A Randomized study on 1-week versus 4-week prophylaxis for venous thromboembolism after laparoscopic surgery for colorectal cancer. Ann Surg 259:665–669
Sakon M, Kobayashi T, Shimazui T (2010) Efficacy and safety of enoxaparin in Japanese patients undergoing curative abdominal or pelvic cancer surgery: results from a multicenter, randomized, open-label study. Thromb Res 125:e65–e70
Bergquivst D, Agnelli G, Cohen AT et al (2002) Duration of prophylaxis against venous thromboembolism with enoxaparin after surgery for cancer. N Engl J Med 346:975–980
Kakkar AJ, Brenner B, Dahl OE et al (2008) Extended duration rivaroxaban versus short-term enoxaparin for the prevention of venous thromboembolism after total hip arthroplasty: a double-blind, randomized controlled trial. Lancet 372:31–39
Kakkar VV, Balibrea JL, Martinez-Gonzales J et al (2010) Extended prophylaxis with bemiparin for the prevention of venous thromboembolism after abdominal or pelvic surgery for cancer: the CANBESURE randomized study. J Thromb Haemost 8:1223–1229
Kolb G, Bodamer I, Galster H et al (2003) Reduction of venous thromboembolism following prolonged prophylaxis with the low molecular weight heparin Certoparin after endoprothetic joint replacement or osteosynthesis of the lower limb in elderly patients. Thromb Haemost 90:1100–1105
Rasmussen MS, Jorgensen LN, Wille-Jorgensen P et al (2006) Prolonged prophylaxis with dalteparin to prevent late thromboembolic complications in patients undergoing major abdominal surgery: a multicenter randomized open-label study. J Thromb Haemost 4:2384–2390
Heit JA, Elliott CG, Trowbridge AA et al (2000) Ardeparin sodium for extended out-of-hospital prophylaxis against venous thromboembolism after total hip or knee replacement: a randomized, double-blind, placebo-controlled trial. Ann Int Med 132:853–861
Kukreja JEB, Levey HR, Scosyrev E et al (2015) Effectiveness and safety of extended-duration prophylaxis for venous thromboembolism in major urologic oncology surgery. Urologic Oncol Semin Orig Invest 33:387.e7–387.e16
Borkgren-Okonek MJ, Hart RW, Pantano JE, Rantis PC, Guske PJ, Kane JM, Gordon N, Sambol NC (2008) Enoxaparin thromboprophylaxis in gastric bypass patients: extended duration, dose stratification, and antifactor Xa activity. Surg Obes Rel Dis 4:625–631
Farrow NE, Aboagye JK, Lau BD et al (2018) The role of extended/outpatient venous thromboembolism prophylaxis after abdominal surgery for cancer or inflammatory bowel disease. J Patient Safety Risk Manage 23(1):19–26
Kanitra J, Holtrop J, Jawad A, Berri R (2019) Extended duration chemoprophylaxis for venous thromboembolism following abdominopelvic oncologic surgery. J Thromb Thrombolysis. https://doi.org/10.1007/s11239-019-02002-9
Cho SE, Delate T, Witt DM, Clark NP (2015) Thromboembolic and bleeding outcomes of extended duration low-intensity warfarin following elective total knee arthroplasty. Thromb Res 135:267–271
Schomburg J, Krishna S, Soubra A et al (2018) Extended outpatient chemoprophylaxis reduces venous thromboembolism after radical cystectomy. Urologic Oncol Semin Orig Invest 36:77.e9–77.e13
Schmeler KM, Langley G, Cain K et al (2013) Venous thromboembolism (VTE) rates following the implementation of extended duration prophylaxis for patients undergoing surgery for gynecologic malignancies. Gynecol Oncol 128(2):1–14
Marino PM, Horcajo RR, Grandal TG et al (2018) Thromboprophylaxis in gynecologic cancer surgery: Is extended prophylaxis with low molecular weight heparin justified? Eur J Obstet Gynecol Reprod Biol 230:90–95
Rattan R, Parreco J, Eidelson SA, Gold J, Dharmaraja A, Zakrison TL, Yeh D, Ginzburg E, Namias N (2018) Hidden burden of venous thromboembolism after trauma: A national analysis. J Trauma Acute Care Surg 85:899–906
Gangireddy C, Rectenwald JR, Upchurch GR et al (2007) Risk factors and clinical impact of postoperative symptomatic venous thromboembolism. J Vasc Surg 45:335–342
Huo MH, Muntz J (2009) Extended Thromboprophylaxis with low-molecular-weight heparins after hospital discharge in high-risk surgical and medical patients: a review. Clin Ther 31:1129–1141
Felder S, Rasmussen MS, King R, Sklow B, Kwaan M, Madoff R, Jensen C (2019) Prolonged thromboprophylaxis with low molecular weight heparin for abdominal or pelvic surgery (Review). Cochrane Database Syst Rev 8:CD004318
Rausa E, Kelly ME, Asti E et al (2018) Extended versus conventional thromboprophylaxis after major abdominal and pelvic surgery: systematic review and meta-analysis of randomized clinical trials. Surg 164:1234–1240
Naik R, Mandal I, Hampson A et al (2019) The role of extended VTE prophylaxis for major urological cancer operations. BJU Int 124(6):935–944
Forster R, Stewart M (2016) Anticoagulants (extended duration) for prevention of venous thromboembolism following total hip or knee replacement or hip fracture repair (Review). Cochrane Database Syst Rev 3:CD004179
Pow RE, Vale PR (2015) Thromboprophylaxis in patients undergoing total hip and knee arthroplasty: a review of current practices in an Australian teaching hospital. Int Med J 45(3):293–299
Author information
Authors and Affiliations
Contributions
Shaikh, Reddy, and Elkbuli were involved in the study conception and design. Shaikh, Reddy, and Elkbuli collected, interpreted, and analyzed the data. Shaikh and Reddy drafted the manuscript. Elkbuli, Shaikh, McKenney, and Reddy critically revised the manuscript. Shaikh, Elkbuli, Reddy, and McKenney were involved in the approval of the final version.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shaikh, S., Reddy, M., McKenney, M. et al. Is Extended-Duration (Post-Hospital Discharge) Venous Thromboembolism Chemoprophylaxis Safe and Efficacious in High-Risk Surgery Patients? A Systematic Review. World J Surg 44, 3363–3371 (2020). https://doi.org/10.1007/s00268-020-05638-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-020-05638-4