Abstract
Background
Recent clinical practice guidelines recommend that routine screening of MEN1 mutation carriers should start at the age of 5 years. The occurrence of clinically relevant MEN1 organ manifestations in children (≤18 years) was evaluated.
Methods
Two prospective collected databases of MEN1 patients (n = 166) who underwent annual screening were retrospectively analyzed for organ manifestations in MEN1 patients ≤18 years. The follow-up was based on the most recent screening examination until December 2015.
Results
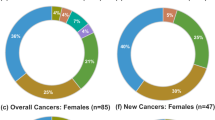
Twenty [11 females, 9 males, (12%)] of 166 MEN1 patients were diagnosed with at least one organ manifestation at age ≤18 years. The most frequent manifestation was mild asymptomatic pHPT (n = 9, 45%, age range 8–18 years). Eight (40%) young patients had pNENs (three non-functioning pNENs, five insulinomas, age range 9–18 years). All five insulinomas were diagnosed based on hypoglycemic symptoms. The other organ manifestations were asymptomatic pituitary adenomas in six patients (30%, age range 15–18 years) and a bronchial carcinoid in one 15-year-old patient. Only six (30%) patients ≤18 years had clinically relevant organ manifestations.
Conclusion
Symptomatic or severe manifestations in MEN1 patients rarely occur below the age of 16 years. With regard to psychological burden and cost-effectiveness, routine screening of asymptomatic MEN1 patients should be postponed at least until the age of 16 years.

Similar content being viewed by others
References
Chandrasekharappa SC, Guru SC, Manickam P et al (1997) Positional cloning of the gene for multiple endocrine neoplasia-type 1. Science 276(5311):404–407
Lourenco DM, Toledo RA, Coutinho FL et al (2007) The impact of clinical and genetic screenings on the management of the multiple endocrine neoplasia type 1. Clinics (Sao Paulo) 62(4):465–476
Thakker RV, Newey PJ, Walls GV et al (2012) Clinical practice guidelines for multiple endocrine neoplasia type 1 (MEN1). J Clin Endocrinol Metab 97(9):2990–3011
Marini F (2015) Genetic test in multiple endocrine neoplasia type 1 syndrome: an evolving story. World J Exp Med 5(2):124
Trump D, Farren B, Wooding C et al (1996) Clinical studies of multiple endocrine neoplasia type 1 (MEN1). QJM 89(9):653–669
Waldmann J, Fendrich V, Habbe N et al (2009) Screening of patients with multiple endocrine neoplasia type 1 (MEN-1): a critical analysis of its value. World J Surg 33(6):1208–1218. doi: 10.1007/s00268-009-9983-8
Geerdink EAM, van der Luijt RB, Lips CJM (2003) Do patients with multiple endocrine neoplasia syndrome type 1 benefit from periodical screening? Eur J Endocrinol 149(6):577–582
Goudet P, Murat A, Binquet C et al (2010) Risk factors and causes of death in MEN1 disease. A GTE (Groupe d’Etude des Tumeurs Endocrines) cohort study among 758 patients. World J Surg 34(2):249–255. doi: 10.1007/s00268-009-0290-1
Ramundo V, Milone F, Severino R et al (2011) Clinical and prognostic implications of the genetic diagnosis of hereditary NET syndromes in asymptomatic patients. Horm Metab Res 43(11):794–800
Goudet P, Dalac A, Le Bras M et al (2015) MEN1 disease occurring before 21 years old: a 160-patient cohort study from the Groupe d’étude des Tumeurs Endocrines. J Clin Endocrinol Metab 100(4):1568–1577
Yates CJ, Newey PJ, Thakker RV (2015) Challenges and controversies in management of pancreatic neuroendocrine tumours in patients with MEN1. Lancet Diabetes Endocrinol 3(11):895–905
Bartsch DK, Albers MB, Lopez CL et al (2015) Bronchopulmonary neuroendocrine neoplasms and their precursor lesions in multiple endocrine neoplasia type 1. Neuroendocrinology 103(3-4):240–247
Langer P, Kann PH, Fendrich V et al (2004) Prospective evaluation of imaging procedures for the detection of pancreaticoduodenal endocrine tumors in patients with multiple endocrine neoplasia type 1. World J Surg 28(12):1317–1322. doi: 10.1007/s00268-004-7642-7
Schaaf L, Pickel J, Zinner K et al (2007) Developing effective screening strategies in multiple endocrine neoplasia type 1 (MEN 1) on the basis of clinical and sequencing data of German patients with MEN 1. Exp Clin Endocrinol Diabetes 115(8):509–517
Machens A, Schaaf L, Karges W et al (2007) Age-related penetrance of endocrine tumours in multiple endocrine neoplasia type 1 (MEN1): a multicentre study of 258 gene carriers. Clin Endocrinol (Oxf) 67(4):613–622
Giusti F, Marini F, Brandi ML (2005) Multiple Endocrine Neoplasia Type 1. In: Pagon RA, Adam MP, Ardinger HH, Wallace SE, Amemiya A, Bean LJH, Bird TD, Ledbetter N, Mefford HC, Smith RJH, Stephens K (eds). GeneReviews® [Internet]. Seattle (WA), University of Washington, Seattle; 1993-2017. Available from: https://www.ncbi.nlm.nih.gov/books/NBK1538/
Clerici T, Schmid C, Komminoth P et al (2001) 10 Swiss kindreds with multiple endocrine neoplasia type 1: assessment of screening methods. Swiss Med Wkly 131(25–26):381–386
Bosman FT, Carneiro F, Hruban RHTN (2010) World Health Organization classification of tumours of the digestive system. IARC, Lyon
Ballard HS, Fame B, Hartsock RJ (1964) Familial multiple endocrine adenoma-peptic ulcer complex. Medicine (Baltimore) 43:481–516
Gonçalves TD, Toledo RA, Sekiya T et al (2013) Penetrance of functioning and nonfunctioning pancreatic neuroendocrine tumors in multiple endocrine neoplasia type 1 in the second decade of life. J Clin Endocrinol Metab 99(1):E89–E96
Lopez CL, Waldmann J, Fendrich V et al (2011) Long-term results of surgery for pancreatic neuroendocrine neoplasms in patients with MEN1. Langenbeck’s Arch Surg 396(8):1187–1196
Triponez F, Dosseh D, Goudet P et al (2006) Epidemiology data on 108 MEN 1 patients from the GTE with isolated nonfunctioning tumors of the pancreas. Ann Surg 243(2):265–272
Doherty GM, Olson JA, Frisella MM et al (1998) Lethality of multiple endocrine neoplasia type I. World J Surg 22(6):581–587. doi: 10.1007/s002689900438
Tamagno G, Carlo E, Martini C et al (2004) The early diagnosis of multiple endocrine neoplasia type 1 (MEN 1): a case report. J Endocrinol Invest 27(9):878–882
Newey PJ, Jeyabalan J, Walls GV et al (2009) Asymptomatic children with multiple endocrine neoplasia type 1 mutations may harbor nonfunctioning pancreatic neuroendocrine tumors. J Clin Endocrinol Metab 94(10):3640–3646
Fabbri HC, de Mello MP, Soardi FC et al (2010) Long-term follow-up of an 8-year- old boy with insulinoma as the first manifestation of a familial form of multiple endocrine neoplasia type 1. Arq Bras Endocrinol Metabol 54(8):754–760
Crippa S, Zerbi A, Boninsegna L et al (2012) Surgical management of insulinomas: short- and long-term outcomes after enucleations and pancreatic resections. Arch Surg 147(3):261–266
Giudici F, Nesi G, Brandi ML et al (2012) Surgical management of insulinomas in multiple endocrine neoplasia type 1. Pancreas 41(4):547–553
Schussheim DH, Skarulis MC, Agarwal SK, Simonds WF, Burns AL, Spiegel AM, Marx SJ (2001) Multiple endocrine neoplasia type 1: new clinical and basic findings. Trends Endocrinol Metab 12(4):173–178
Vezzosi D, Cardot-Bauters C, Bouscaren N et al (2015) Long-term results of the surgical management of insulinoma patients with MEN1: a Groupe d’étude des Tumeurs Endocrines (GTE) retrospective study. Eur J Endocrinol 172(3):309–319
Gibril F, Schumann M, Pace A et al (2004) Multiple endocrine neoplasia type 1 and zollinger-ellison syndrome. Medicine (Baltimore) 83(1):43–83
Capella C, Riva C, Leutner M et al (1995) Pituitary lesions in multiple endocrine neoplasia syndrome (MENS) type 1. Pathol Res Pract 191(4):345–347
Horvath A, Stratakis CA (2008) Clinical and molecular genetics of acromegaly: MEN1, Carney complex, McCune-Albright syndrome, familial acromegaly and genetic defects in sporadic tumors. Rev Endocr Metab Disord 9(1):1–11
Pinna G, Orgiana G, Carcassi C et al (2004) A novel germline mutation of MEN 1 gene in a patient with acromegaly and multiple endocrine tumors. J Endocrinol Invest 27(6):577–582
Sahdev A, Jager R (2000) Bilateral pituitary adenomas occurring with multiple endocrine neoplasia type one. AJNR 21(6):1067–1069
Burgess JR, Shepherd JJ, Parameswaran V et al (1996) Spectrum of pituitary disease in multiple endocrine neoplasia type 1 (MEN 1): clinical, biochemical, and radiological features of pituitary disease in a large MEN 1 kindred. J Clin Endocrinol Metab 81(7):2642–2646
Vergès B, Boureille F, Goudet P et al (2002) Pituitary disease in MEN type 1 (MEN1): data from the France-Belgium MEN1 multicenter study. J Clin Endocrinol Metab 87(2):457–465
Sachithanandan N, Harle RA, Burgess JR (2005) Bronchopulmonary carcinoid in multiple endocrine neoplasia type 1. Cancer 103(3):509–515
Gatta-Cherifi B, Chabre O, Murat A et al (2012) Adrenal involvement in MEN1. Analysis of 715 cases from the Groupe d’etude des Tumeurs Endocrines database. Eur J Endocrinol 166(2):269–279
Waldmann J, Bartsch DK, Kann PH et al (2007) Adrenal involvement in multiple endocrine neoplasia type 1: results of 7 years prospective screening. Langenbecks Arch Surg. 392(4):437–443
Schaefer S, Shipotko M, Meyer S et al (2008) Natural course of small adrenal lesions in multiple endocrine neoplasia type 1: an endoscopic ultrasound imaging study. Eur J Endocrinol 158(5):699–704
Stromsvik N, Nordin K, Berglund G et al (2007) Living with multiple endocrine neoplasia type 1: decent care-insufficient medical and genetic information: a qualitative study of MEN 1 patients in a Swedish hospital. J Genet Couns 16(1):105–117
Acknowledgements
We are grateful to all patients that participated in the study.
Funding
This research did not receive any specific grant from any funding agency in the public, commercial or nonprofit sector.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research/data reported.
Rights and permissions
About this article
Cite this article
Manoharan, J., Raue, F., Lopez, C.L. et al. Is Routine Screening of Young Asymptomatic MEN1 Patients Necessary?. World J Surg 41, 2026–2032 (2017). https://doi.org/10.1007/s00268-017-3992-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-017-3992-9