Abstract
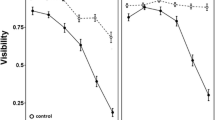
Understanding how long-term changes in environmental conditions influence the way that individuals cope with threats is essential in the context of behavioral adaptation to a rapidly changing world. However, little is known about the behavioral responses to predation risk for individuals that experienced different environmental conditions for extended periods of time, such as food levels and light conditions. In this experimental study, we tested whether previous long-term exposure to different food levels (low versus high) and light conditions (0-h light versus 8-h light) plays a significant role in shaping the antipredator response (i.e., the probability of emerging from the refuge and the distance moved) to stimuli from caged larval dragonflies, in larvae of the fire salamander (Salamandra salamandra). Specifically, we quantified behavioral differences in the response to predation risk in larval salamanders that were reared in the laboratory for 2 months under controlled food and light conditions. The results of this study showed that the interaction between food level and light conditions affected the antipredator behavior of the larvae. Fire salamander larvae maintained at low food levels and in 8-h light conditions emerged from the refuge with a higher probability (i.e., took more risk) than larvae maintained at high food levels and all other combinations of light conditions. Thus, our results highlight the complexity of antipredator responses, pointing attention to the fact that interactions among environmental factors are likely to determine the magnitude of antipredator response.
Significance statement
Few studies have investigated the role of multiple environmental factors on the expression of predator-induced behavioral responses. Specifically, because no study has so far investigated the risk-taking behavior in individuals exposed to long-term, contrasting food levels, and light conditions, we investigated this in amphibian larvae. We showed that environmental conditions interactively determined antipredator behavior. This highlights the importance of considering long-term environmental conditions experienced by an organism and their interactions when experimentally studying behavioral variation to adequately understand its expression in natural conditions.



Similar content being viewed by others
Data availability
The dataset analyzed during the current study is available in the KNB repository: https://knb.ecoinformatics.org/view/urn%3Auuid%3A7d01db76-0c1a-429c-a442-a8788e85f419.
References
Albrecht U, Oster H (2001) The circadian clock and behaviour. Behav Brain Res 125:89–91. https://doi.org/10.1016/S0166-4328(01)00288-1
Alford RA (1999) Ecology: resource use, competition, and predation. In: McDiarmid RW, Altig R (eds) Tadpoles: the biology of anuran larvae. University of Chicago Press, Chicago, pp 240–278
Altwegg R (2003) Hungry predators render predator-avoidance behavior in tadpoles ineffective. Oikos 100:311–316. https://doi.org/10.1034/j.1600-0706.2003.12206.x
Anderson JD, Graham RE (1967) Vertical migration and stratification of larval Ambystoma. Copeia 1967:371c374. https://doi.org/10.2307/1442127
Anholt BR, Werner EE (1995) Interaction between food availability and predation mortality mediated by adaptive behavior. Ecology 76:2230–2234. https://doi.org/10.2307/1941696
Anholt BR, Skelly DK, Werner EE (1996) Factors modifying antipredator behavior in larval toads. Herpetologica 52:301–313
Anholt BR, Werner E, Skelly DK (2000) Effect of food and predators on the activity of four larval ranid frogs. Ecology 81:3059–3521. https://doi.org/10.1890/0012-9658(2000)081[3509:EOFAPO]2.0.CO;2
Babbitt KJ, Tanner GW (1997) Effects of cover and predator identity on predation of Hyla squirella tadpoles. J Herpetol 31:128–130. https://doi.org/10.2307/1565342
Bates D, Mächler M, Bolker BM, Walker SC (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bonter DN, Zuckerberg B, Sedgwick CW, Hochachka WM (2013) Daily foraging patterns in free-living birds: exploring the predation-starvation trade-off. Proc R Soc B 280:20123087. https://doi.org/10.1098/rspb.2012.3087
Carlson BE, Newman JC, Langkilde T (2015) Food or fear: hunger modifies responses to injured conspecifics in tadpoles. Hydrobiologia 743:299–308. https://doi.org/10.1007/s10750-014-2048-5
Cressler CE, King AA, Werner EE (2010) Interactions between behavioral and life-history trade-offs in the evolution of integrated predator-defense plasticity. Am Nat 176:276–288. https://doi.org/10.1086/655425
Cupp PV (1994) Salamanders avoid chemical cues from predators. Anim Behav 48:232–235. https://doi.org/10.1006/anbe.1994.1231
da Silva Nunes V (1988) Vocalizations of treefrogs (Smilisca sila) in response to bat predation. Herpetologica 44:8–10
Delcourt J, Denoël M, Ylieff M, Poncin P (2013) Video multitracking of fish behaviour: a synthesis and future perspectives. Fish Fish 14:186–204. https://doi.org/10.1111/j.1467-2979.2012.00462.x
Ding GH, Lin ZH, Zhao LH, Fan XL, Wei L (2014) Effects of light intensity on activity in four sympatric anuran tadpoles. Zool Res 35:332–337. https://doi.org/10.13918/j.issn.2095-8137.2014.4.332
Erskine DJ, Hutchison VH (1982) Reduced thermal tolerance in an amphibian treated with melatonin. J Therm Biol 7:121–123
Fox J, Weisberg S (2011) An R companion to applied regression, 2nd edn. Sage, Thousand Oaks
Gern WA, Norris DO, Duvall D (1983) The effect of light and temperature on plasma melatonin in neotenic tiger salamanders (Ambystoma tigrinum). J Herpetol 17:228–234. https://doi.org/10.2307/1563824
Godin J-GJ, Crossman SL (1994) Hunger-dependent predator inspection and foraging behaviors in the threespine stickleback (Gasterosteus aculeatus) under predation risk. Behav Ecol Sociobiol 34:359–366. https://doi.org/10.1007/BF00197006
Heller R, Milinski M (1979) Optimal foraging of sticklebacks on swarming prey. Anim Behav 27:1127–1141. https://doi.org/10.1016/0003-3472(79)90061-7
Holomuzki JR (1986) Predator avoidance and diel patterns of microhabitat use by larval tiger salamanders. Ecology 67:737–748. https://doi.org/10.2307/1937697
Horat P, Semlitsch RD (1994) Effects of predation risk and hunger on the behaviour of two species of tadpoles. Behav Ecol Sociobiol 34:393–401. https://doi.org/10.1007/BF00167330
Hutchison VH, Black JJ, Erskine DJ (1979) Melatonin and chlorpromazine: thermal selection in the mud-puppy, Necturus maculosus. Life Sci 25:527–530. https://doi.org/10.1016/0024-3205(79)90565-4
Juszczyk W, Zakrzewski M (1981) External morphology of larval stages of the spotted salamander, Salamandra salamandra (L). Acta Biol Cracov Ser Zoo 23:127–135
Kats LB, Dill LM (1998) The scent of death: chemosensory assessment of predation risk by prey animals. Ecoscience 5:361–363. https://doi.org/10.1080/11956860.1998.11682468
Krause ET, Steinfartz S, Caspers BA (2011) Poor nutritional conditions during the early larval stage reduce risk-taking activities of fire salamander larvae (Salamandra salamandra). Ethology 117:416–421. https://doi.org/10.1111/j.1439-0310.2011.01886.x
Kurali A, Pásztor K, Hettyey A, Tóth Z (2018) Resource-dependent temporal changes in antipredator behavior of common toad (Bufo bufo) tadpoles. Behav Ecol Sociobiol 72:91. https://doi.org/10.1007/s00265-018-2503-9
Lawler SP (1989) Behavioural responses to predators and predation risk in four species of larval anurans. Anim Behav 38:1039–1047. https://doi.org/10.1016/S0003-3472(89)80142-3
Lienart GD, Mitchell MD, Ferrari MC, McCormick MI (2014) Temperature and food availability affect risk assessment in an ectotherm. Anim Behav 89:199–204. https://doi.org/10.1016/j.anbehav.2013.12.031
Lima SL (1998) Stress and decision making under the risk of predation: recent developments from behavioral, reproductive, and ecological perspectives. Adv Stud Behav 27:215–290. https://doi.org/10.1016/S0065-3454(08)60366-6
Lima SL, Dill LM (1990) Behavioral decisions made under the risk of predation: a review and prospectus. Can J Zool 68:619–640. https://doi.org/10.1139/z90-092
Manenti R, Ficetola GF, Marieni A, De Bernardi F (2011) Caves as breeding sites for Salamandra salamandra: habitat selection, larval development and conservation issues. North-West J Zool 7:304–309
Manenti R, Denoël M, Ficetola GF (2013) Foraging plasticity favours adaptation to new habitats in fire salamanders. Anim Behav 86:375–382. https://doi.org/10.1016/j.anbehav.2013.05.028
Manenti R, Melotto A, Denoël M, Ficetola GF (2016) Amphibian breeding in refuge habitats have larvae with stronger antipredatory responses. Anim Behav 118:115–121. https://doi.org/10.1016/j.anbehav.2016.06.006
Mangel M, Clark CW (1986) Towards a unified foraging theory. Ecology 67:1127–1138. https://doi.org/10.2307/1938669
Martín J, López P (1999) When to come out from a refuge: risk-sensitive and state-dependent decisions in an alpine lizard. Behav Ecol 10:487–492. https://doi.org/10.1093/beheco/10.5.487
McNamara JM, Houston AI (1986) The common currency for behavioral decisions. Am Nat 127:358–378
Nonacs P, Dill LM (1990) Mortality risk versus food quality trade-offs in a common currency: patch preferences in ants. Ecology 71:1886–1892. https://doi.org/10.2307/1937596
Oswald P, Tunnat BA, Hahn LG, Caspers BA (2020) There is no place like home: larval habitat type and size affect risk-taking behaviour in fire salamander larvae (Salamandra salamandra). Ethology 126:914–921. https://doi.org/10.1111/eth.13070
Poulsen SB, Svendsen JC, Jensen LF, Schulz C, Jäger-Kleinicke T, Schwarten H (2010) Effects of food deprivation on refuge use and dispersal in juvenile North Sea houting Coregonus oxyrinchus under experimental conditions. J Fish Biol 77:1702–1708. https://doi.org/10.1111/j.1095-8649.2010.02772.x
R Core Team (2020) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, https://www.R-project.org/
Rand AS, Bridarolli ME, Dries L, Ryan MJ (1997) Light levels influence female choice in Túngara frogs: predation risk assessment? Copeia 1997:447–450. https://doi.org/10.2307/1447770
Schneider C, Rasband W, Eliceiri K (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Methods 9:671–675. https://doi.org/10.1038/nmeth.2089
Semlitsch RD (1987) Interactions between fish and salamander larvae. Oecologia 72:482–486. https://doi.org/10.1007/BF00378972
Semlitsch RD, Reyer HU (1992) Modification of anti-predator behaviour in tadpoles by environmental conditioning. J Anim Ecol 61:353–360. https://doi.org/10.2307/5327
Sih A (1980) Optimal behavior: can foragers balance two conflicting demands? Science 210:1041–1043. https://doi.org/10.1126/science.210.4473.1041
Sih A (1987) Predators and prey lifestyles: an evolutionary and ecological overview. In: Kerfoot WC, Sih A (eds) Predation: direct and indirect impacts on aquatic communities. University Press of New England, Hanover, pp 203–224
Sih A, Petranka JW, Kats LB (1988) The dynamics of prey refuge use: a model and tests with sunfish and salamander larvae. Am Nat 132:463–483. https://doi.org/10.1086/284865
Sih A, Kats LB, Moore RD (1992) Effects of predatory sunfish on the density, drift and refuge use of stream salamander larvae. Ecology 73:1418–1430. https://doi.org/10.2307/1940687
Sogard SM, Olla BL (1996) Food deprivation affects vertical distribution and activity of a marine fish in a thermal gradient: potential energy-conserving mechanisms. Mar Ecol Prog Ser 133:43–55. https://doi.org/10.3354/meps133043
Sredl MJ, Collins JP (1992) The interaction of predation, competition, and habitat complexity in structuring an amphibian community. Copeia 1992:607–614. https://doi.org/10.2307/1446138
Taylor JT (1984) Comparative evidence for competition between the salamanders Ambystoma gracile and Taricha granulosa. Copeia 1984:672–683. https://doi.org/10.2307/1445148
Teplitsky C, Plénet S, Joly P (2003) Tadpoles’ responses to risk of fish introduction. Oecologia 134:270–277. https://doi.org/10.1007/s00442-002-1106-2
Thiesmeier B (2004) Der Feuersalamander. Laurenti-Verlag, Bielefeld
Tvardíková K, Fuchs R (2011) Do birds behave according to dynamic risk assessment theory? A feeder experiment. Behav Ecol Sociobiol 65:727–733. https://doi.org/10.1007/s00265-010-1075-0
Van Buskirk J, Schmidt BR (2000) Predator-induced phenotypic plasticity in larval newts: trade-offs, selection, and variation in nature. Ecology 81:3009–3028. https://doi.org/10.1890/0012-9658(2000)081[3009:PIPPIL]2.0.CO;2
Vanecek J (1998) Cellular mechanisms of melatonin action. Physiol Rev 78:687–721. https://doi.org/10.1152/physrev.1998.78.3.687
Werner EE, Anholt BR (1993) Ecological consequences of the tradeoff between growth and mortality rates mediated by foraging activity. Am Nat 142:242–272. https://doi.org/10.1086/285537
Whitham J, Mathis AJ (2000) Effects of hunger and predation risk on foraging behavior of graybelly salamanders, Eurycea multiplicata. J Chem Ecol 26:1659–1665. https://doi.org/10.1023/A:1005590913680
Winandy L, Colin M, Denoël M (2016) Temporal habitat shift of a polymorphic newt species under predation risk. Behav Ecol 27:1025–1032. https://doi.org/10.1093/beheco/arw008
Wise SE, Buchanan BW (2006) Influence of artificial illumination on the nocturnal behavior and physiology of salamanders. In: Rich C, Tongcore T (eds) Ecological Consequences of Artificial Night Lighting. Island Press, Washington DC, pp 221–251
Wise SE, Jaeger RJ (1998) The influence of tail autotomy on agonistic behaviour in a territorial salamander. Anim Behav 55:1707–1716. https://doi.org/10.1006/anbe.1997.0720
Zhou XQ, Niu CJ, Li QF, Ma HF (1998) The effect of light intensity on daily food consumption and specific growth rate of the juvenile soft-shelled turtle, Trionyx sinensis. Acta Zool Sinica 44:157–161
Acknowledgments
We are grateful to the following master, PhD students, and volunteers for their invaluable help in rearing the fire salamander larvae: Sabina Vlad (Ochiană), Elena Șușter, Alexandra Telea, Anca Soare, Sebastian Topliceanu, Roberto Festuccia, Ştefan Cătălin Baba, Augustin Nae, Ionuț Popa, Valerică Toma, Marius Robu, and Dragos Bălăşoiu. We highly appreciate the invaluable advice offered by Diana Szèkely and Dan Cogălniceanu, which greatly improved our experimental design. We thank both reviewers for their constructive comments on the manuscript.
Funding
The collaboration between Ovidius University Constanța, Institute of Speleology of the Romanian Academy and University of Liège was supported by a grant from Wallonie-Bruxelles International (W.B.I.) and Executive Unit for Financing Higher Education, Research, Development and Innovation CCCDI-UEFISCDI (ANCS), project number 105BM/2017. RIB was additionally supported by UEFISCDI through grant PN-II-RU-TE-2014-4-1536, and grant PN-III-P1-1.1-TE-2019-1233. MD is a Research Director at Fonds de la Recherche Scientifique – FNRS (Belgium). FS was partly supported by the project ANTREPRENORDOC, in the framework of Human Resources Development Operational Programme 2014-2020, financed from the European Social Fund under the contract number 36355/23.05.2019 HRD OP /380/6/13 – SMIS Code: 123847, and by grant PN-III-1.2-PCCDI-2017-0721 (INTERASPA).
Author information
Authors and Affiliations
Contributions
RIB, FS, and MD conceived the idea of the study. RIB, FS, and BRS analyzed the data. RIB and MD wrote the manuscript with input from FS and BRS. MD supervised the work. RP and IN contributed to the design and carrying out the behavioral experiments.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
Because this study was only observational and not experimental with regards of laboratory research on animals, it did not need specific ethical approval. The collection, captive maintenance, and observation procedure of the fire salamander larvae complied with all relevant guidelines, notably the Directive 2010/63/UE of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. Romanian Speleological Heritage Commission issued environmental permit no 78/10.02.2016. Both 8-h and 0-h light experimental conditions used to rear the fire salamander larvae mimicked natural situations, as this species is naturally present in these two situations in the wild as they can be present in caves (Manenti et al. 2011). No larva died during the experiment.
Additional information
Communicated by A. Taylor Baugh
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
ESM 1
(DOCX 29.0 kb)
Rights and permissions
About this article
Cite this article
Băncilă, R.I., Plăiaşu, R., Stănescu, F. et al. Food level and light conditions affect the antipredator behavior in larvae of a stream-breeding amphibian. Behav Ecol Sociobiol 75, 36 (2021). https://doi.org/10.1007/s00265-021-02966-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00265-021-02966-w